Abstract
Background: Households represent important settings for transmission of influenza and other respiratory viruses. Current influenza diagnosis and treatment relies upon patient visits to healthcare facilities, which may lead to under‐diagnosis and treatment delays. This study aimed to assess the feasibility of an at‐home approach to influenza diagnosis and treatment via home testing, telehealth care, and rapid antiviral home delivery.
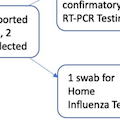
Methods: We conducted a pilot interventional study of remote influenza diagnosis and treatment in Seattle‐area households with children during the 2019‐2020 influenza season using pre‐positioned nasal swabs and home influenza tests. Home monitoring for respiratory symptoms occurred weekly; if symptoms were reported within 48 hours of onset, participants collected mid‐nasal swabs and used a rapid home‐based influenza immunoassay. An additional home‐collected swab was returned to a laboratory for confirmatory influenza RT‐PCR testing. Baloxavir antiviral treatment was prescribed and delivered to symptomatic and age‐eligible participants, following a telehealth encounter.
Results: 124 households comprising 481 individuals self‐monitored for respiratory symptoms, with 58 home tests administered. 12 home tests were positive for influenza, of which eight were true positives confirmed by RT‐PCR. The sensitivity and specificity of the home influenza test were 72.7% and 96.2%, respectively. There were eight home deliveries of Baloxavir, with 7 (87.5%) occurring within 3 hours of prescription and all within 48 hours of symptom onset.
Conclusions: We demonstrate the feasibility of self‐testing combined with rapid home delivery of influenza antiviral treatment. This approach may be an important control strategy for influenza epidemics and pandemics.