SARS-CoV-2 evolution in light of Omicron
Trevor Bedford (@trvrb)
Fred Hutchinson Cancer Research Center / Howard Hughes Medical Institute
15 Mar 2022
Viruses and Vaccines Seminar Series
Brotman Baty Institute
Slides at: bedford.io/talks
1. Emergence of variants of concern
2. Assessing adaptive evolution
3. Variant transmission dynamics
4. Omicron emergence and spread
5. Future perspectives
Emergence of variants of concern
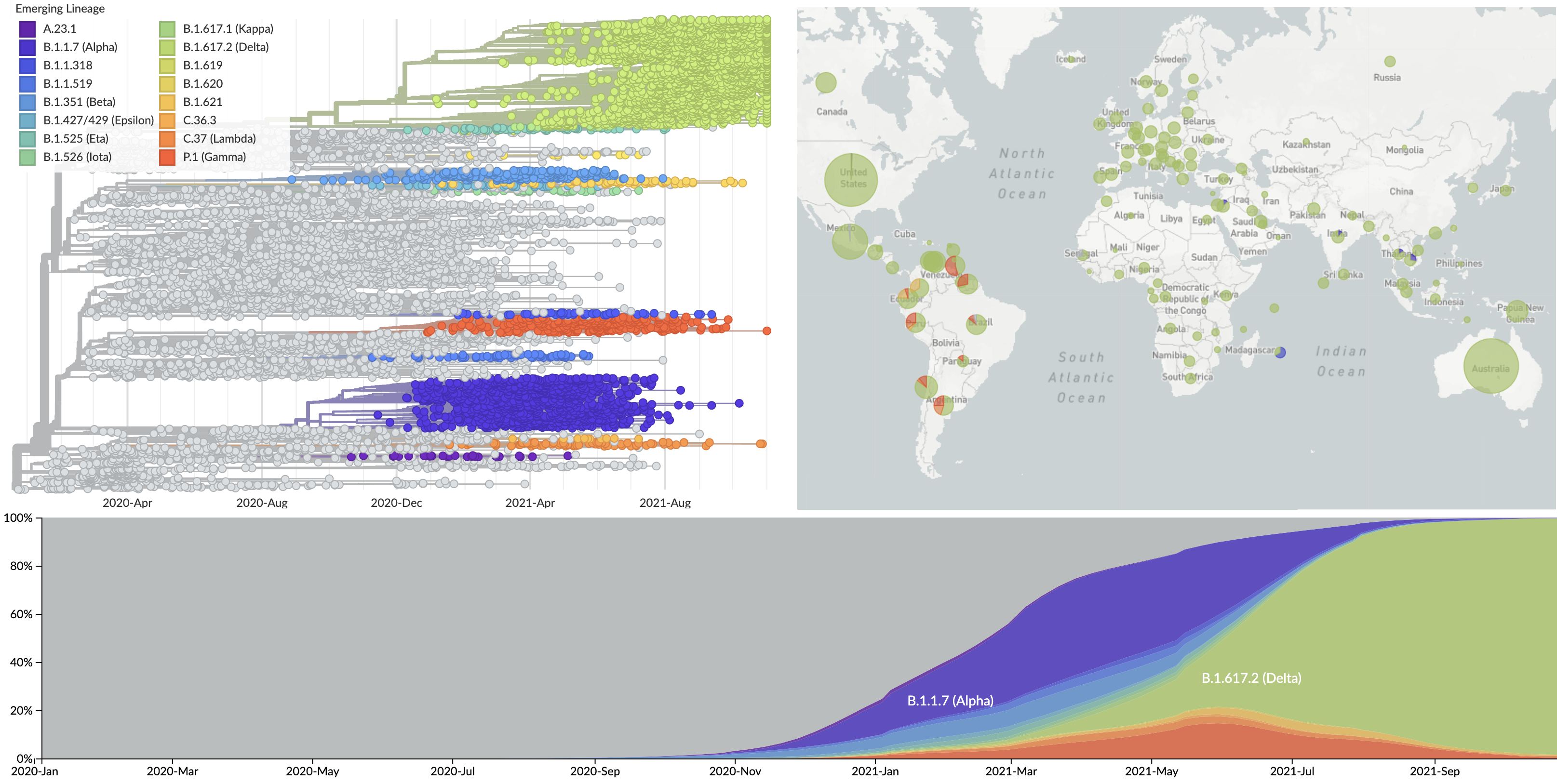
Over 9.2M SARS-CoV-2 genomes shared to GISAID and evolution tracked in real-time at nextstrain.org
![]() Richard Neher,
Richard Neher,
![]() Emma Hodcroft,
Emma Hodcroft,
![]() James Hadfield,
James Hadfield,
![]() Thomas Sibley,
Thomas Sibley,
![]() John Huddleston,
John Huddleston,
![]() Ivan Aksamentov,
Ivan Aksamentov,
![]() Moira Zuber,
Moira Zuber,
![]() Jover Lee,
Jover Lee,
![]() Cassia Wagner,
Cassia Wagner,
![]() Denisse Sequeira,
Denisse Sequeira,
![]() Cornelius Roemer,
Cornelius Roemer,
![]() Victor Lin,
Victor Lin,
![]() Jennifer Chang
Jennifer Chang
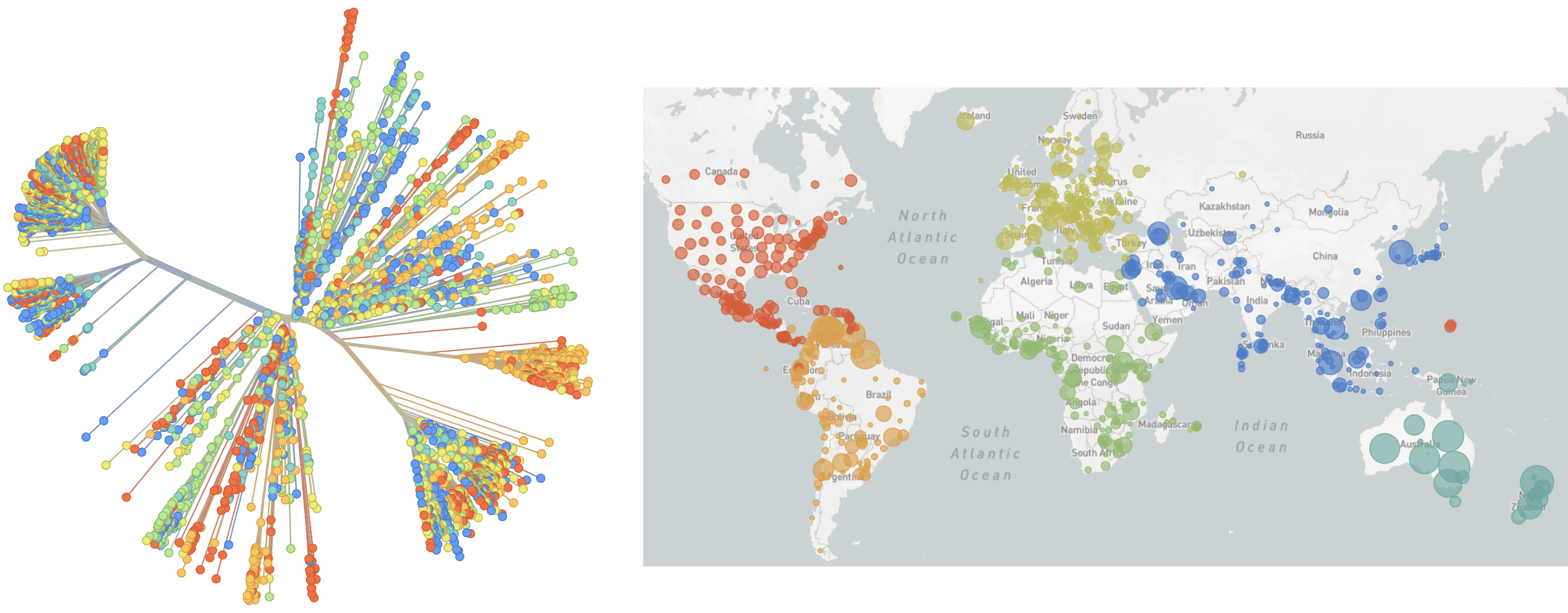
SARS-CoV-2 lineages establish globally in February and March
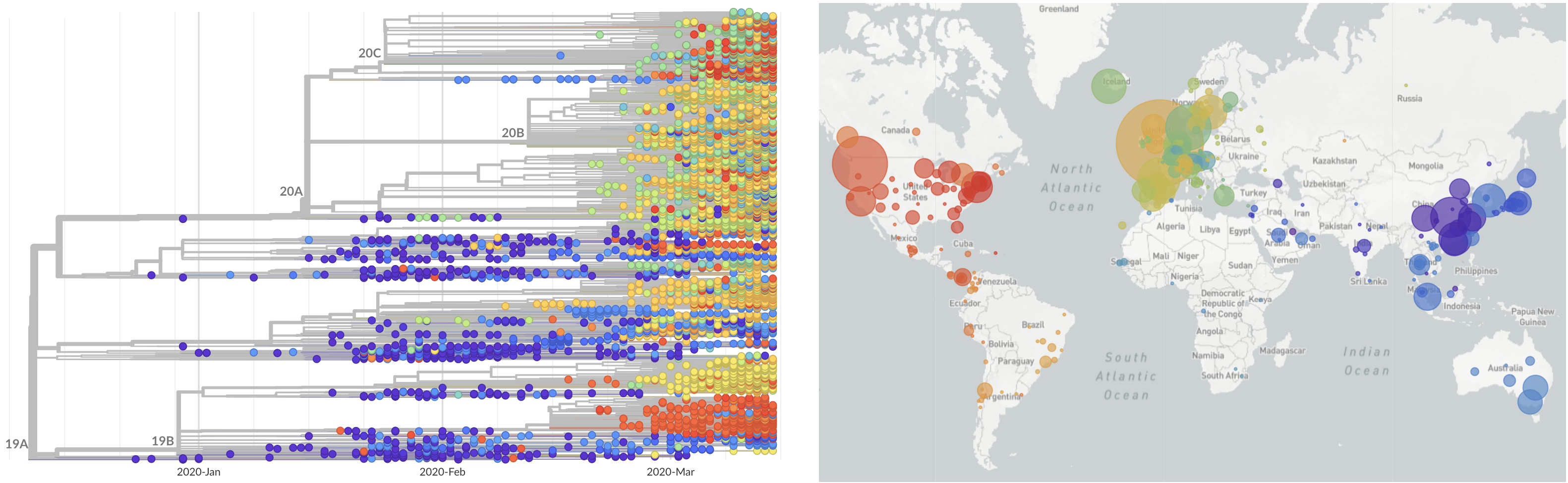
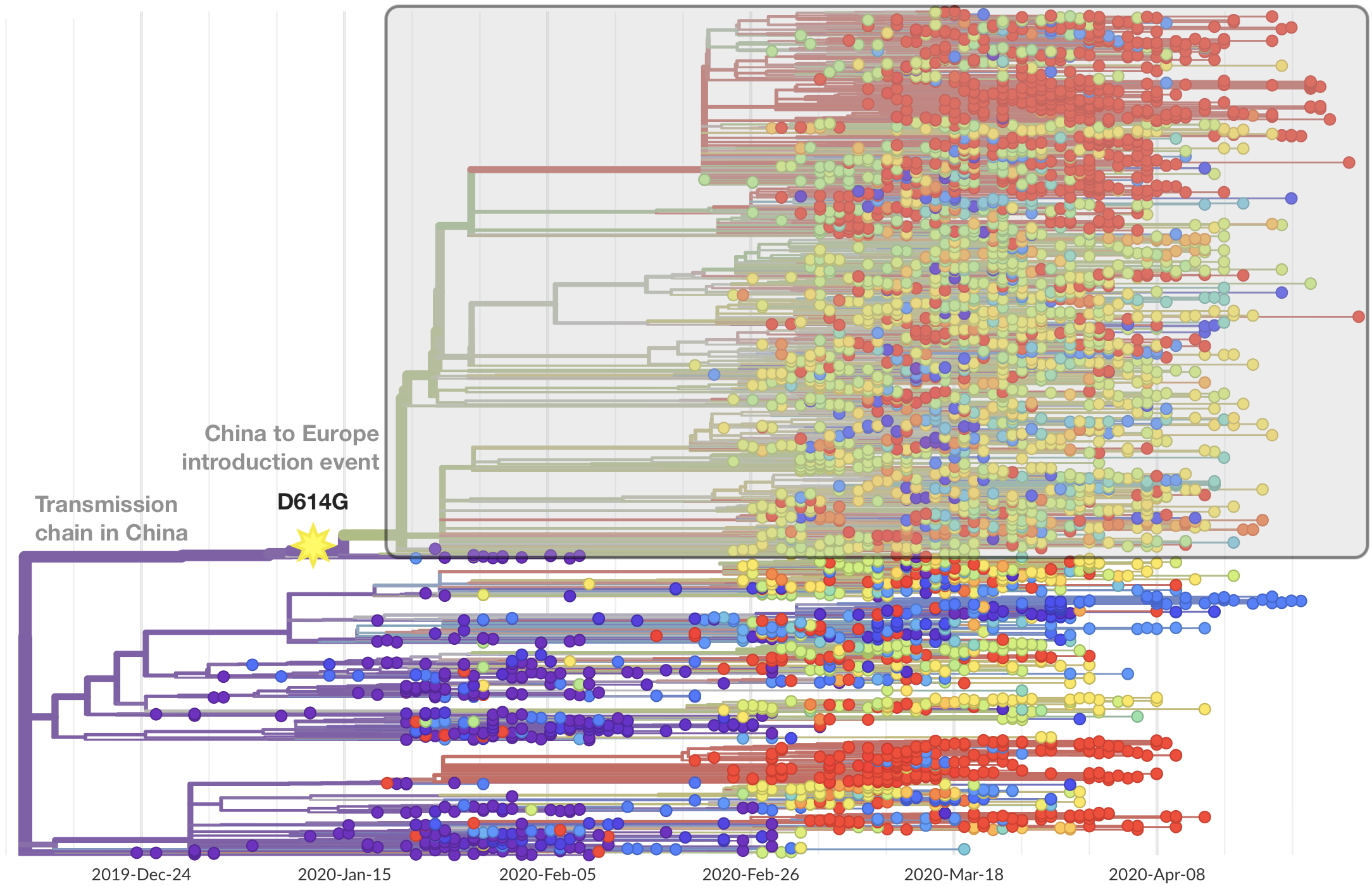
Limited early mutations like spike D614G spread globally during initial wave
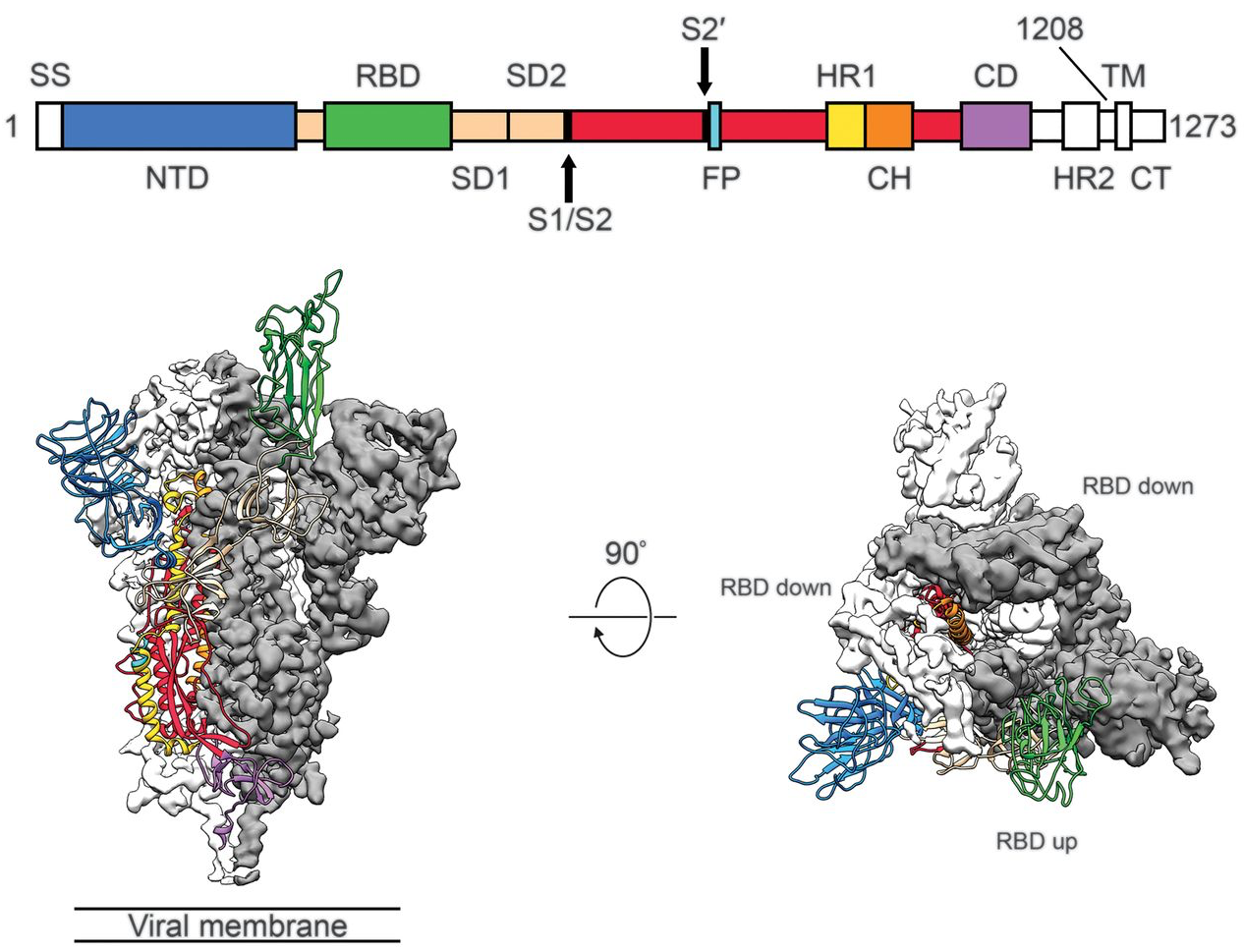
Spike protein is critical for cell invasion by the virus and is the primary target for human immune response
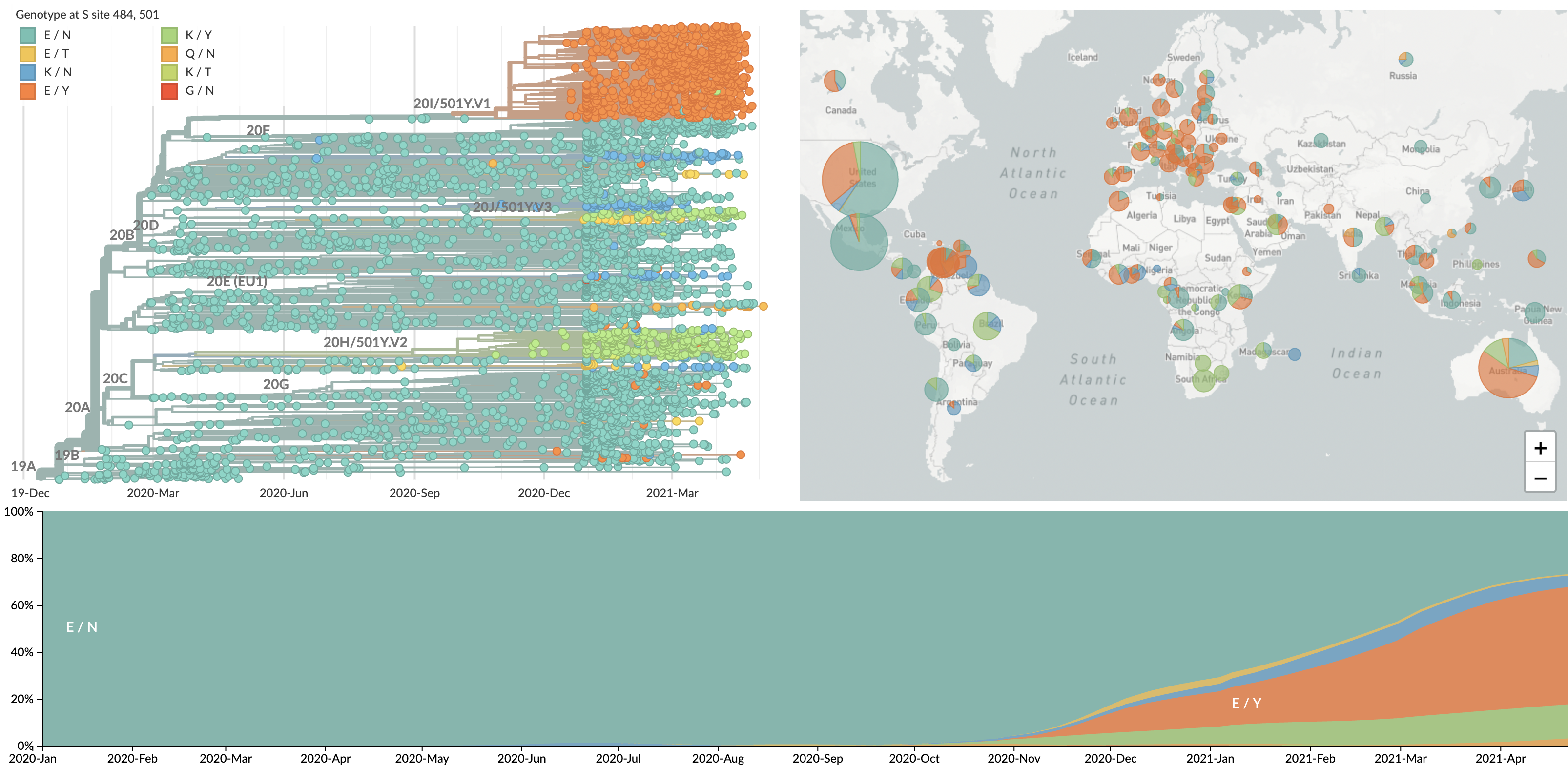
Repeated emergence of 484K and 501Y across the world
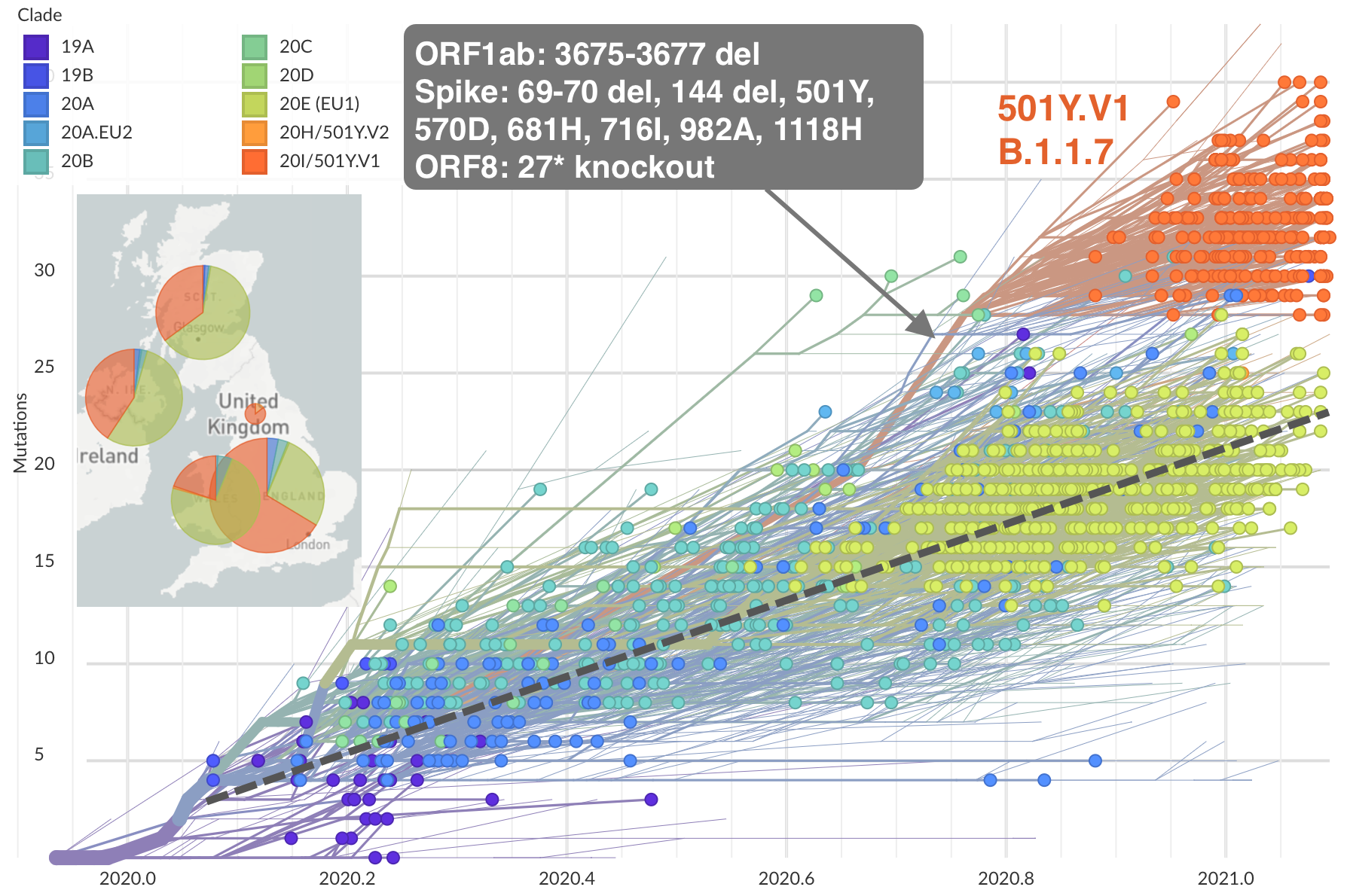
Emergence of Alpha (B.1.1.7) in the UK
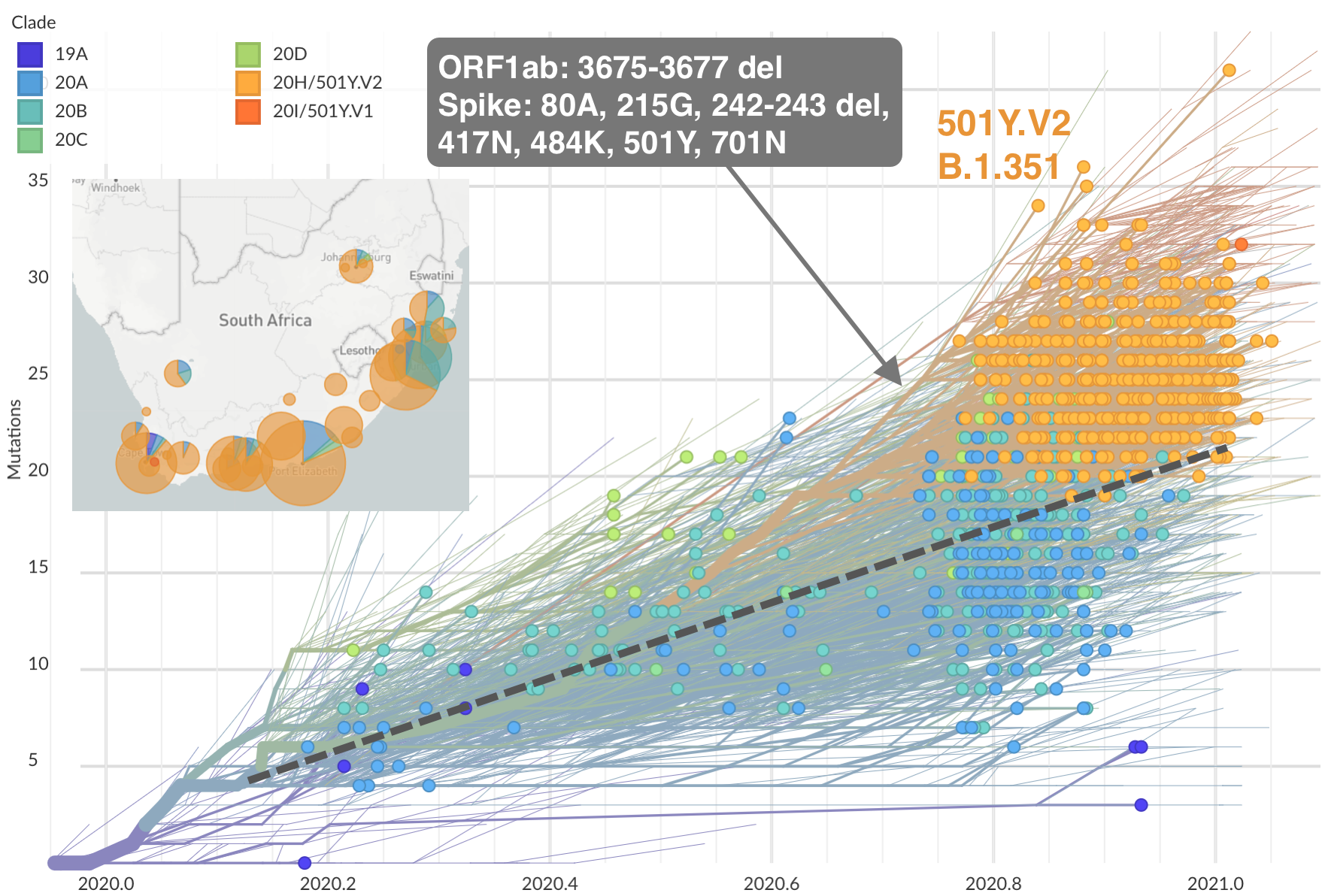
Emergence of Beta (B.1.351) in the South Africa
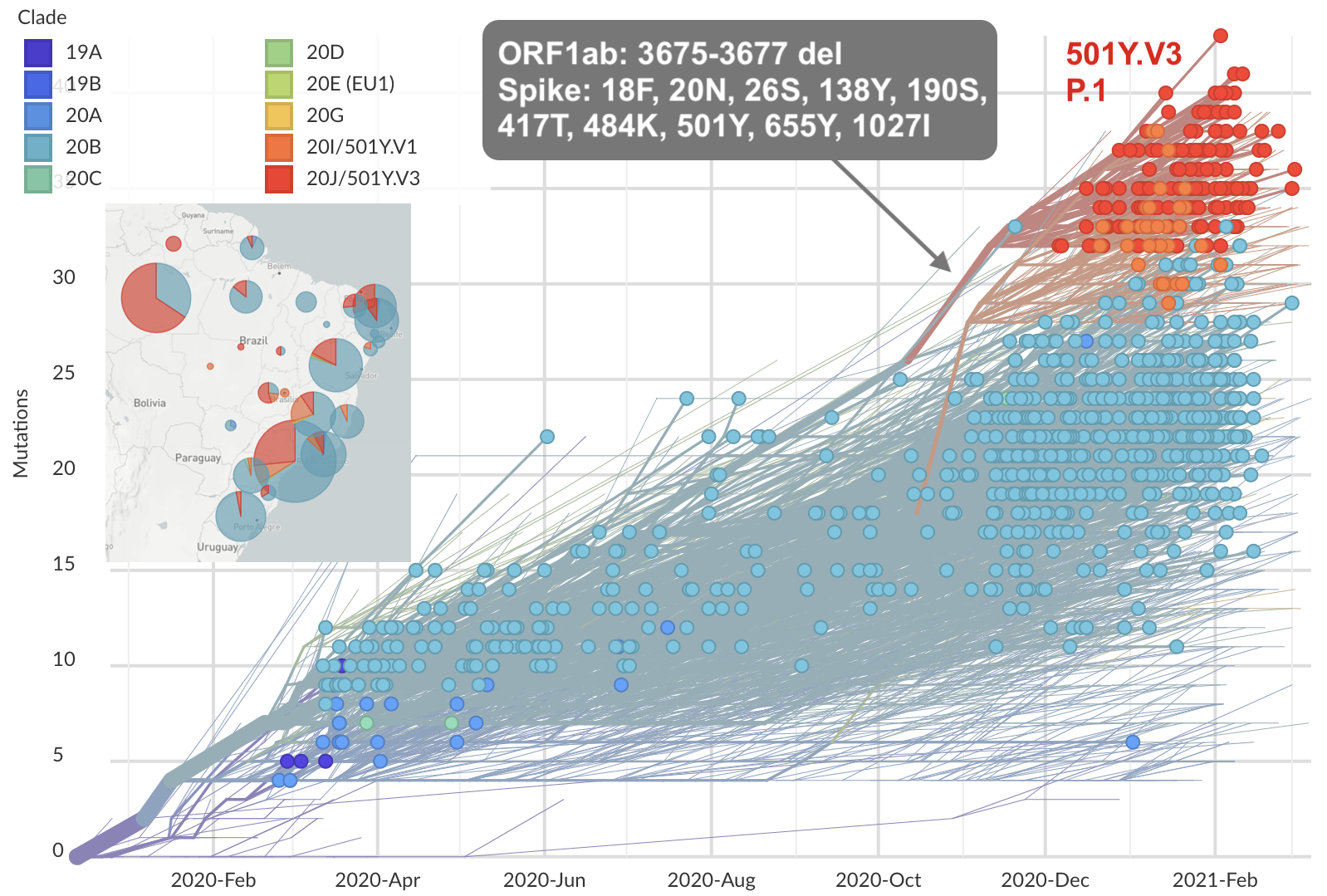
Emergence of Gamma (P.1) in the Brazil
Spread of VOC / VOI lineages across the world
Delta outcompeting other variants and seemed poised to sweep
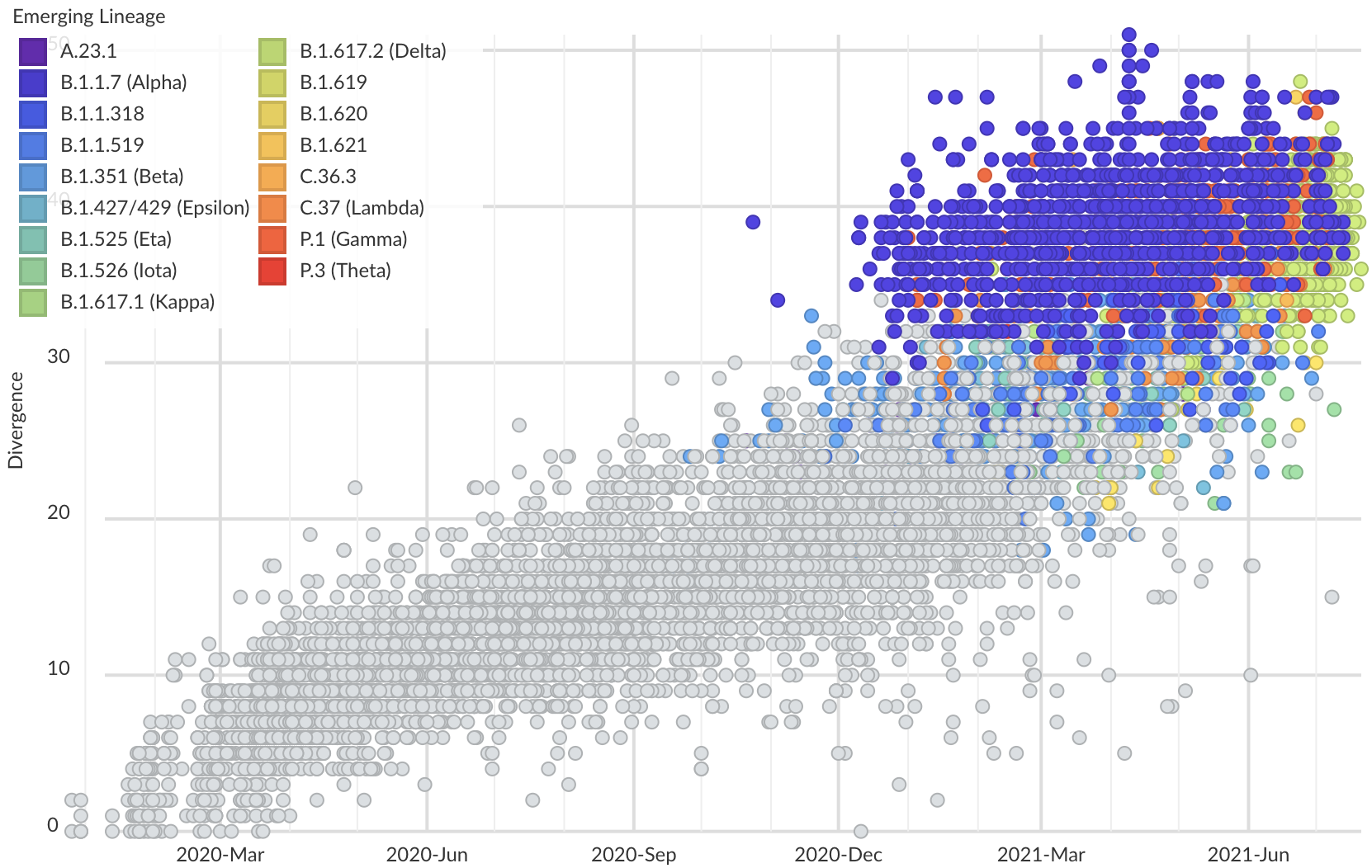
Variants show excess mutations across the genome
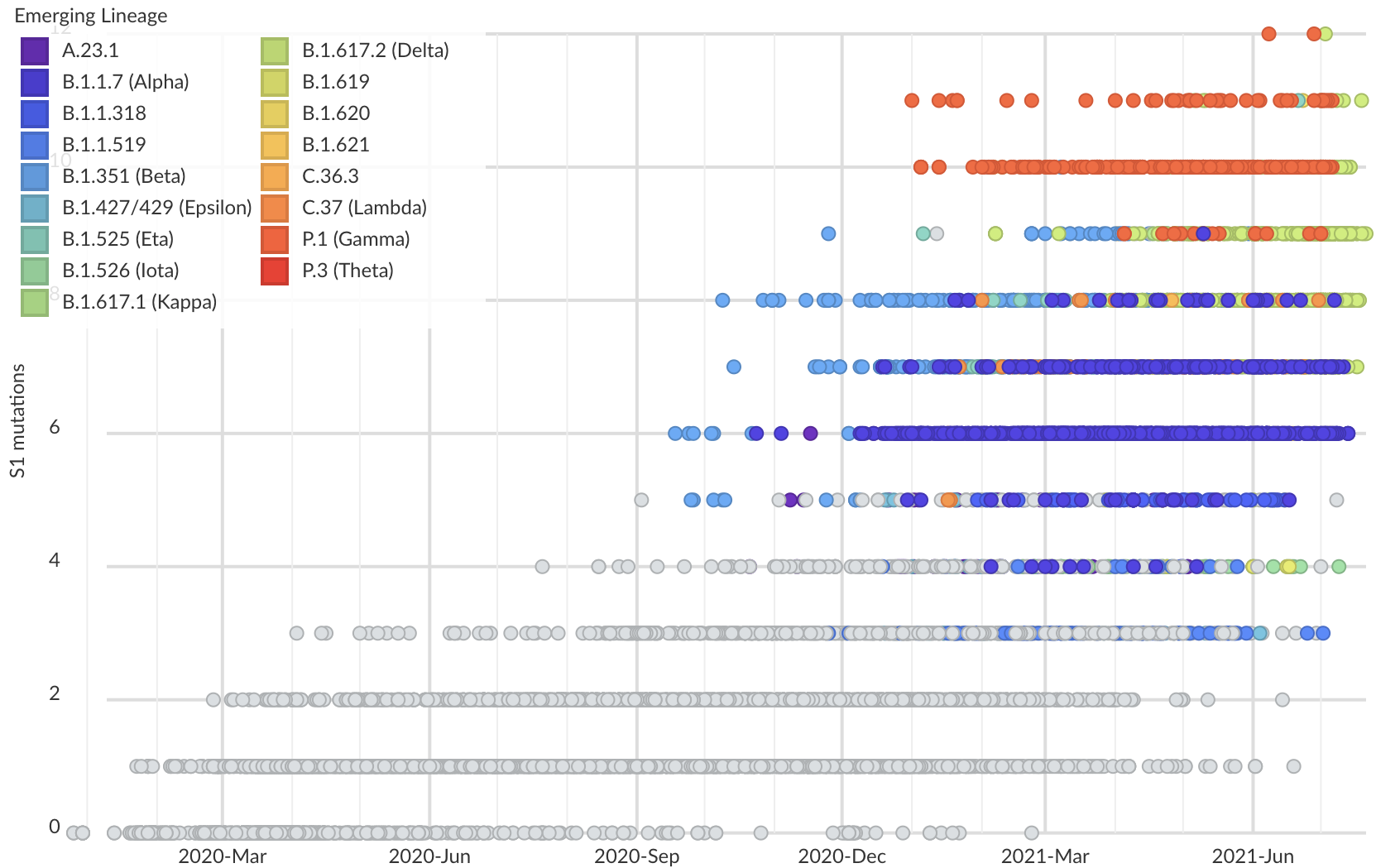
But show most substantial excess in the S1 domain of spike
Assessing adaptive evolution
Rapid and parallel adaptive mutations in spike S1 drive clade success in SARS-CoV-2
 Katie Kistler,
Katie Kistler,
 John Huddleston
John Huddleston
Phylogeny of 10k genomes equitably sampled in space and time
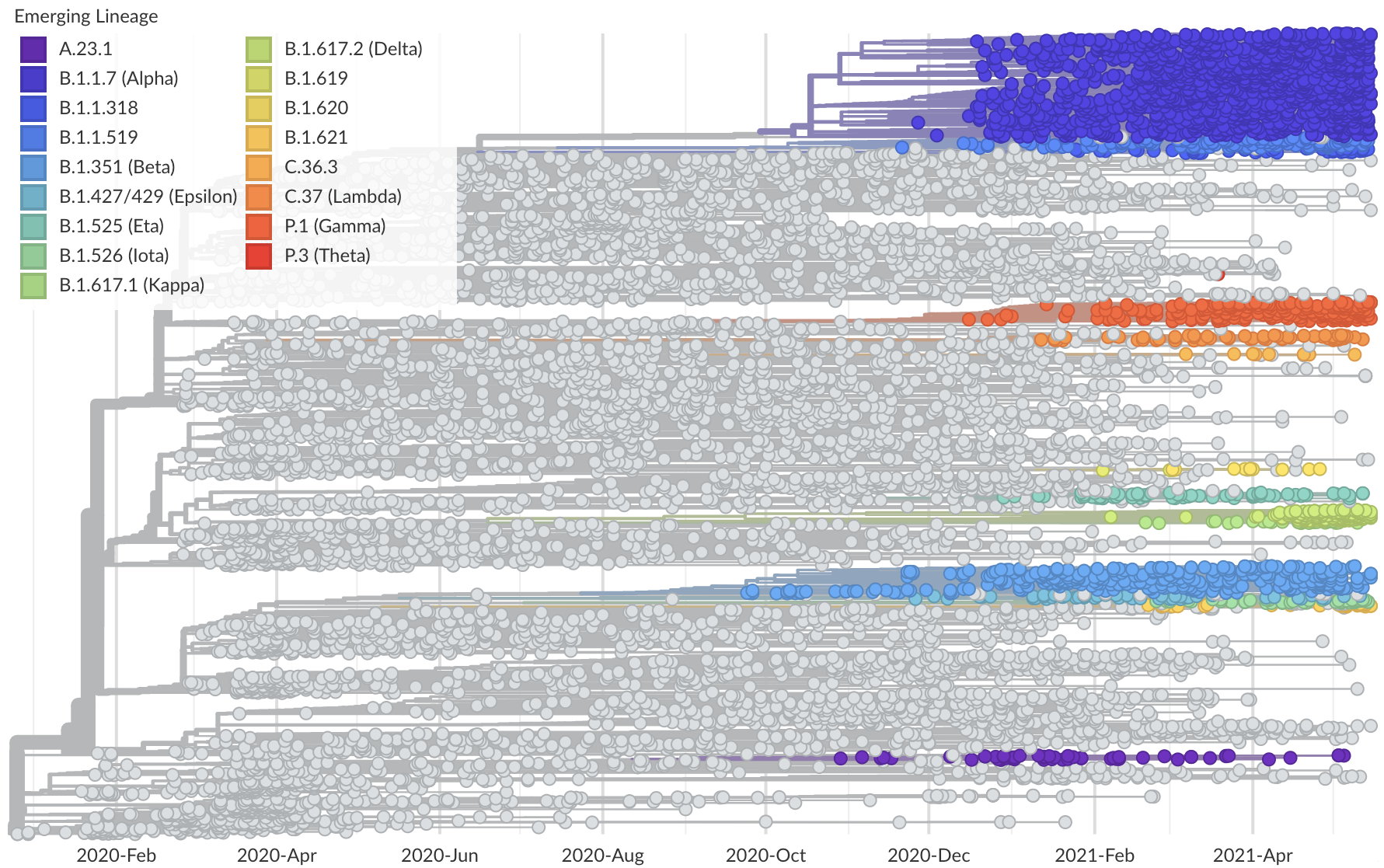
Measure clade growth as a proxy for viral fitness
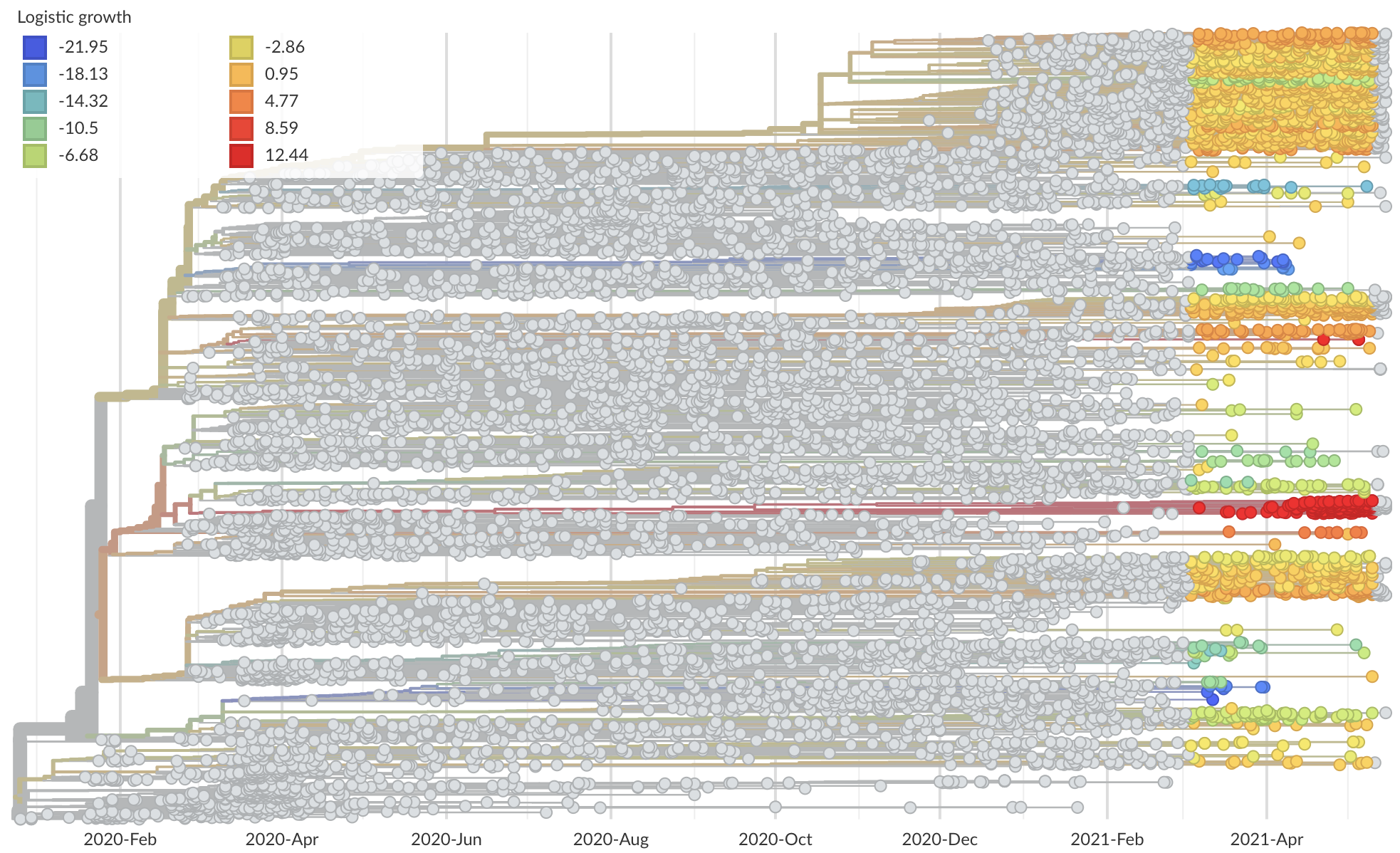
Clades with more S1 nonsynonymous mutations grow faster
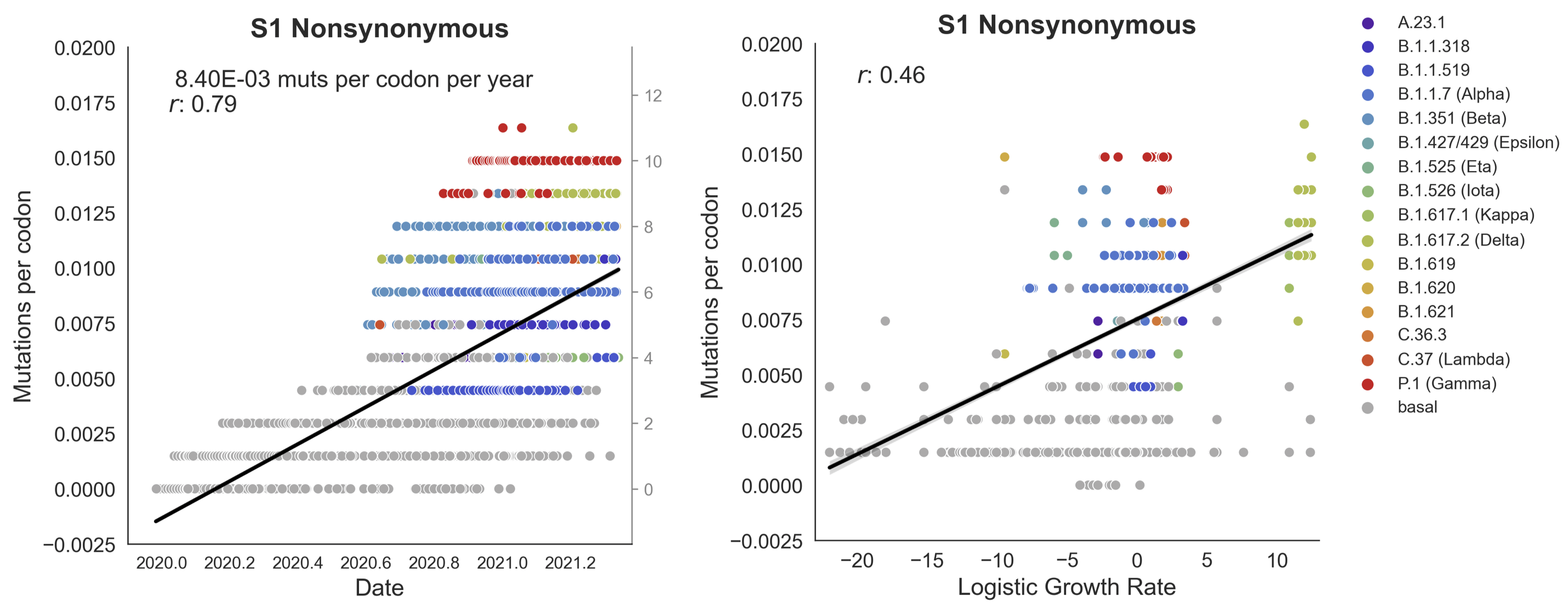
S1 is quickly evolving and highly correlated with clade growth
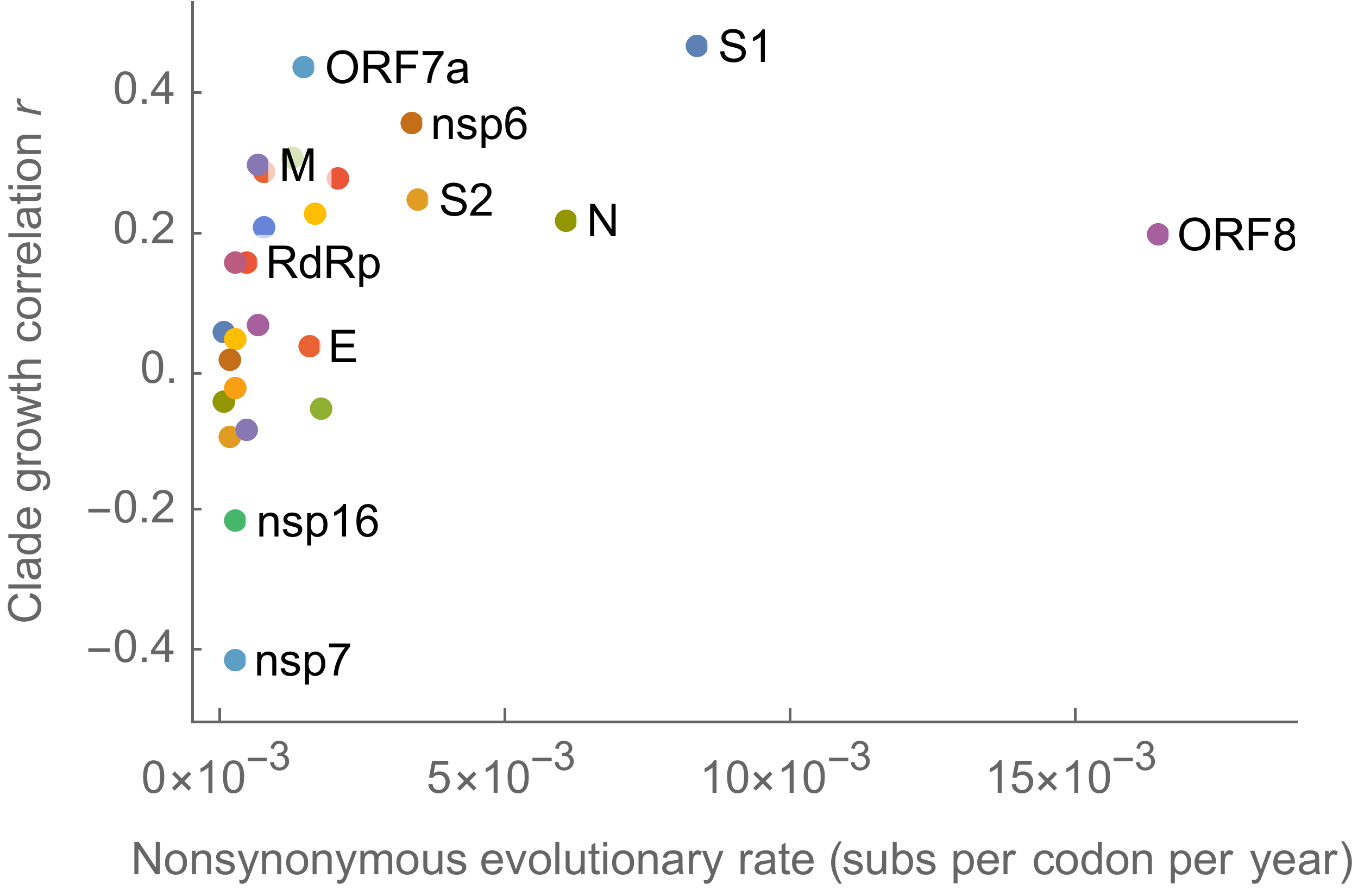
dN/dS to root further highlights adaptive evolution
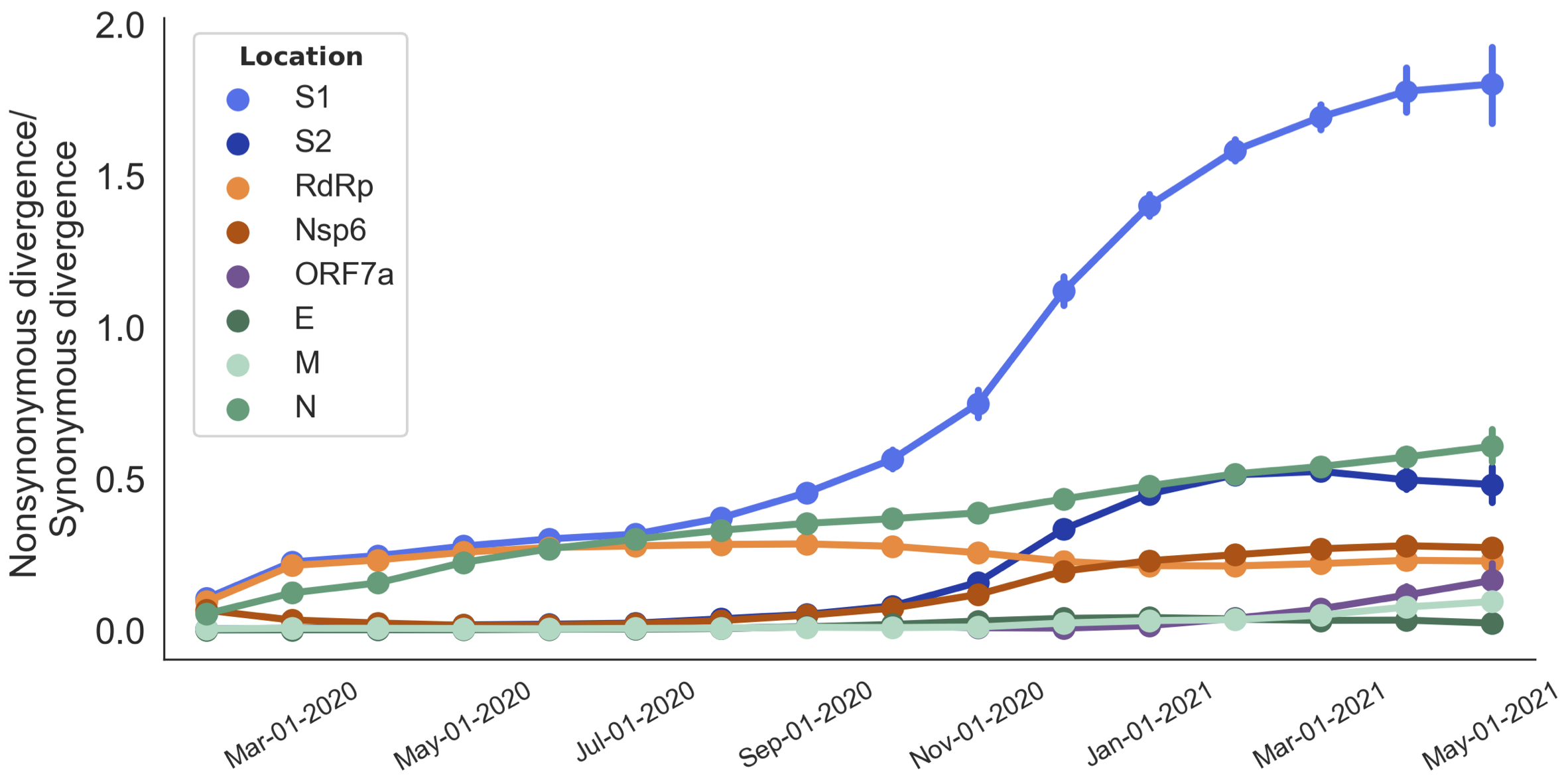
Rapid pace of adaptive evolution relative to H3N2 influenza
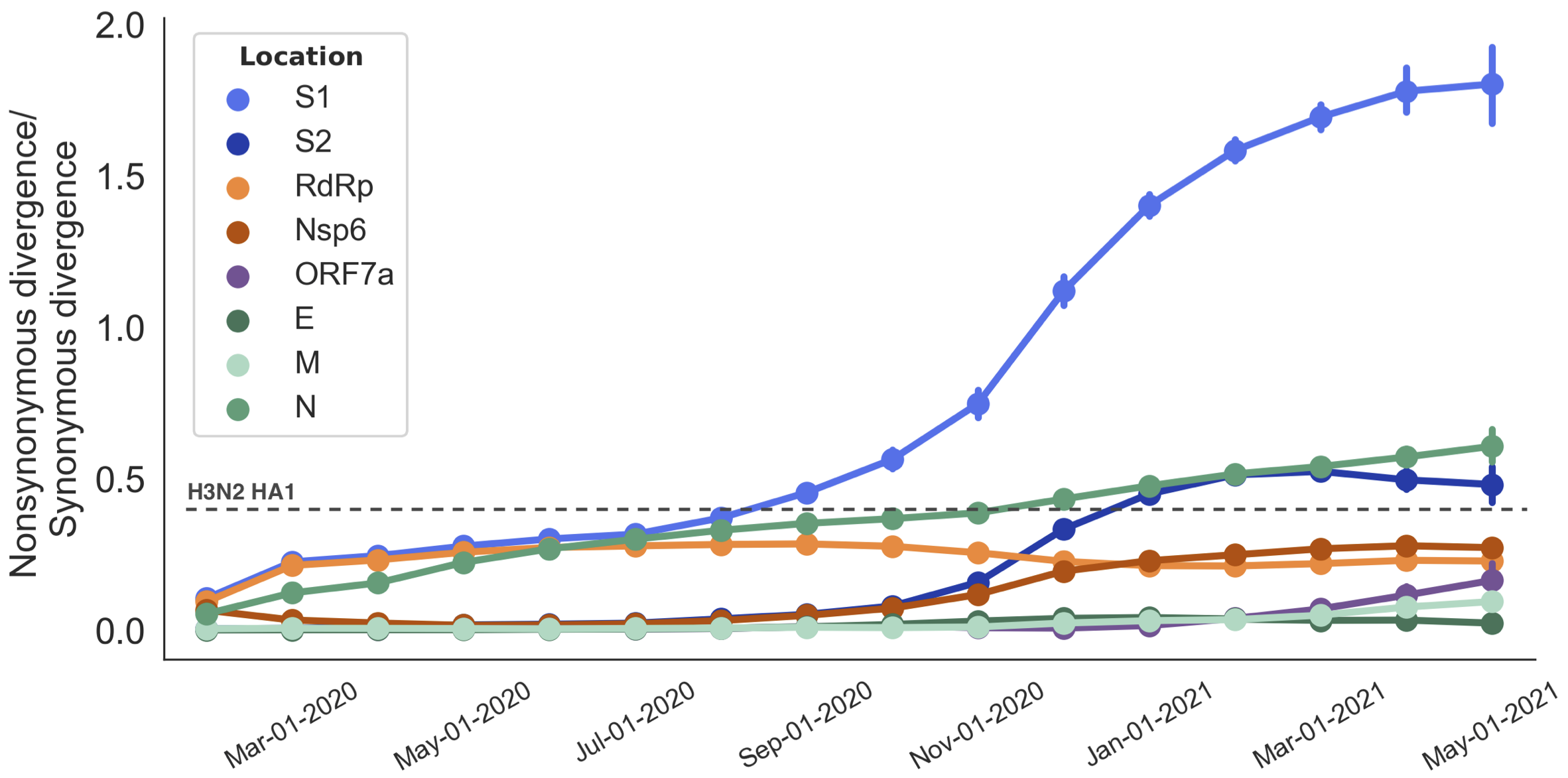
Mutations in S1 arising via within-host pressures result increase viral fitness and are enriched in the viral population by natural selection
Although selection has not been primarily for antigenic drift, observed level of adaptability suggests its potential
Variant transmission dynamics
Multiple approaches to modeling fitness differences between circulating variants
Multinomial logistic regression models work well on frequencies
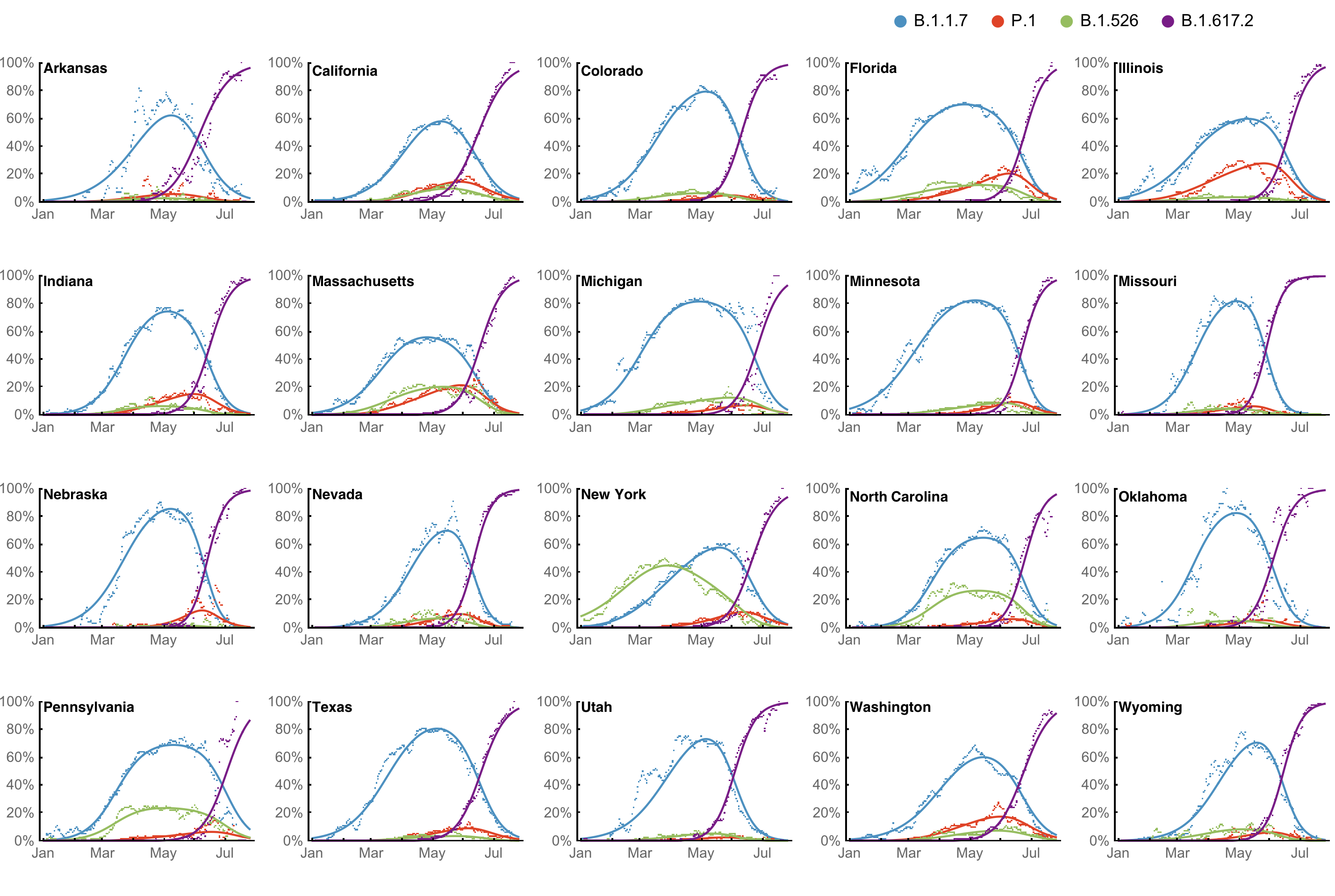
However, frequency of a variant may rise while cases fall
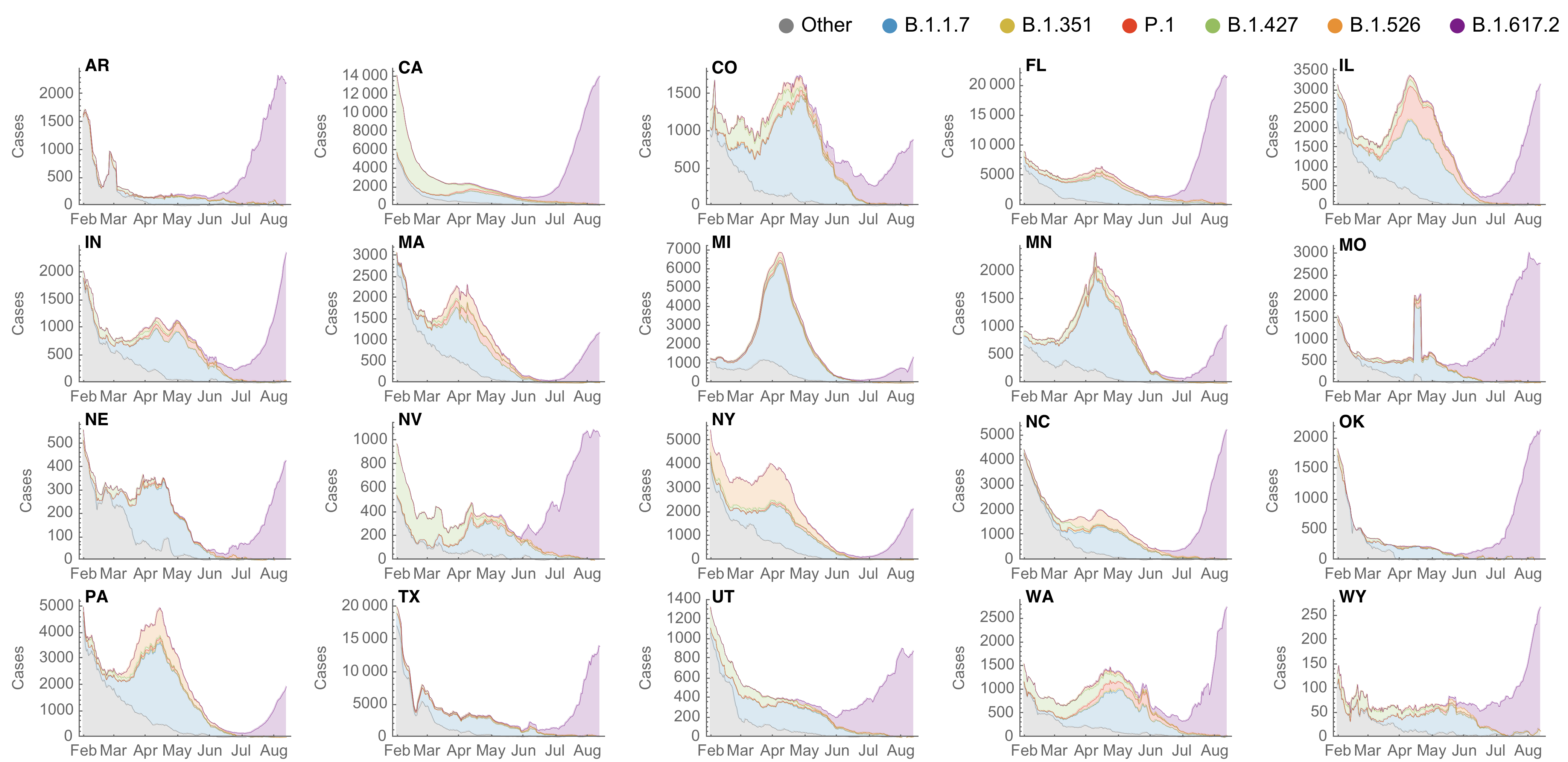
SARS-CoV-2 variant dynamics across US states show consistent differences in transmission rates
 Marlin Figgins
Marlin Figgins
Estimation of variant-specific Rt through time using state-level data
State-level case counts are partitioned based on frequencies of sequenced cases
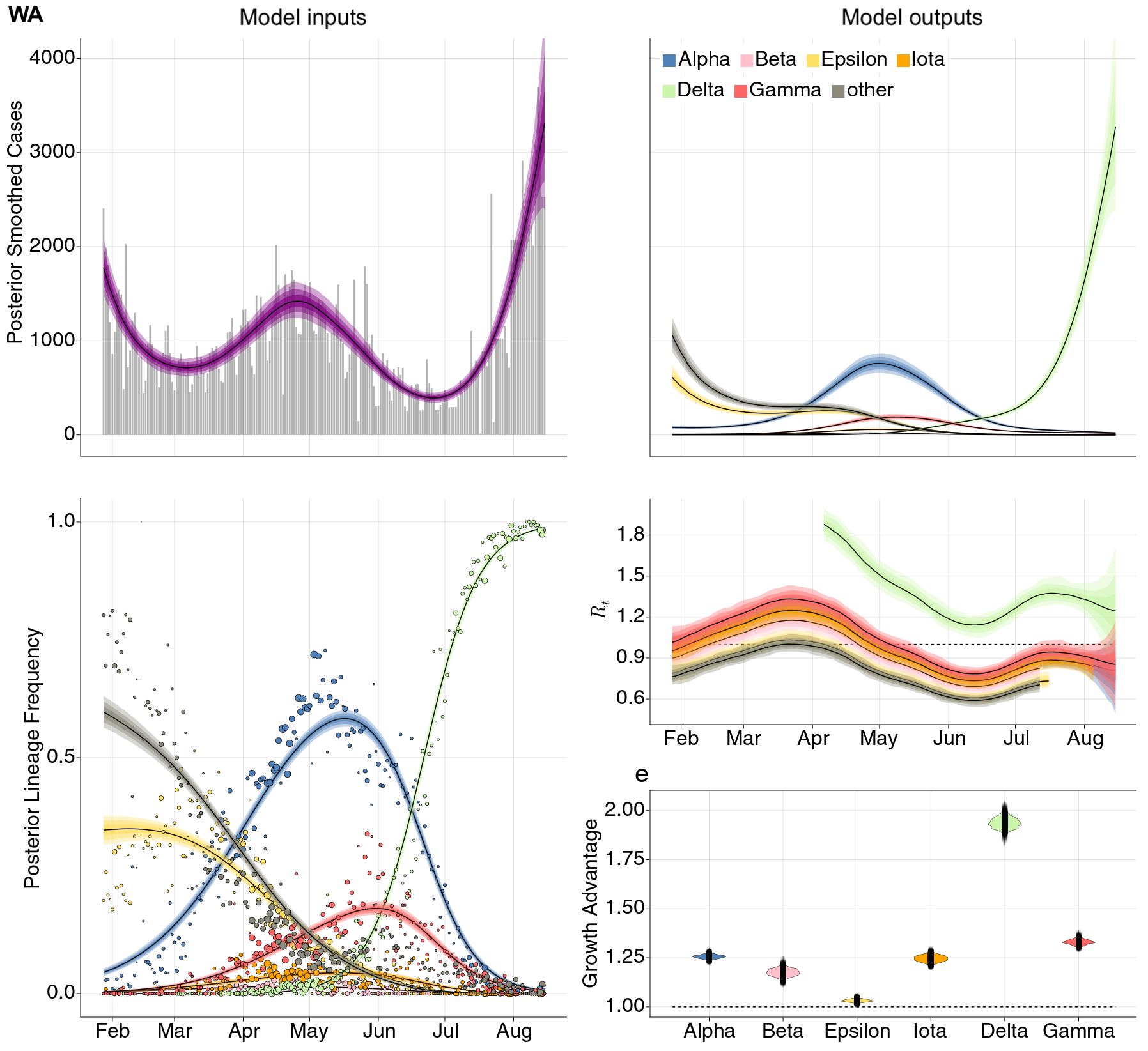
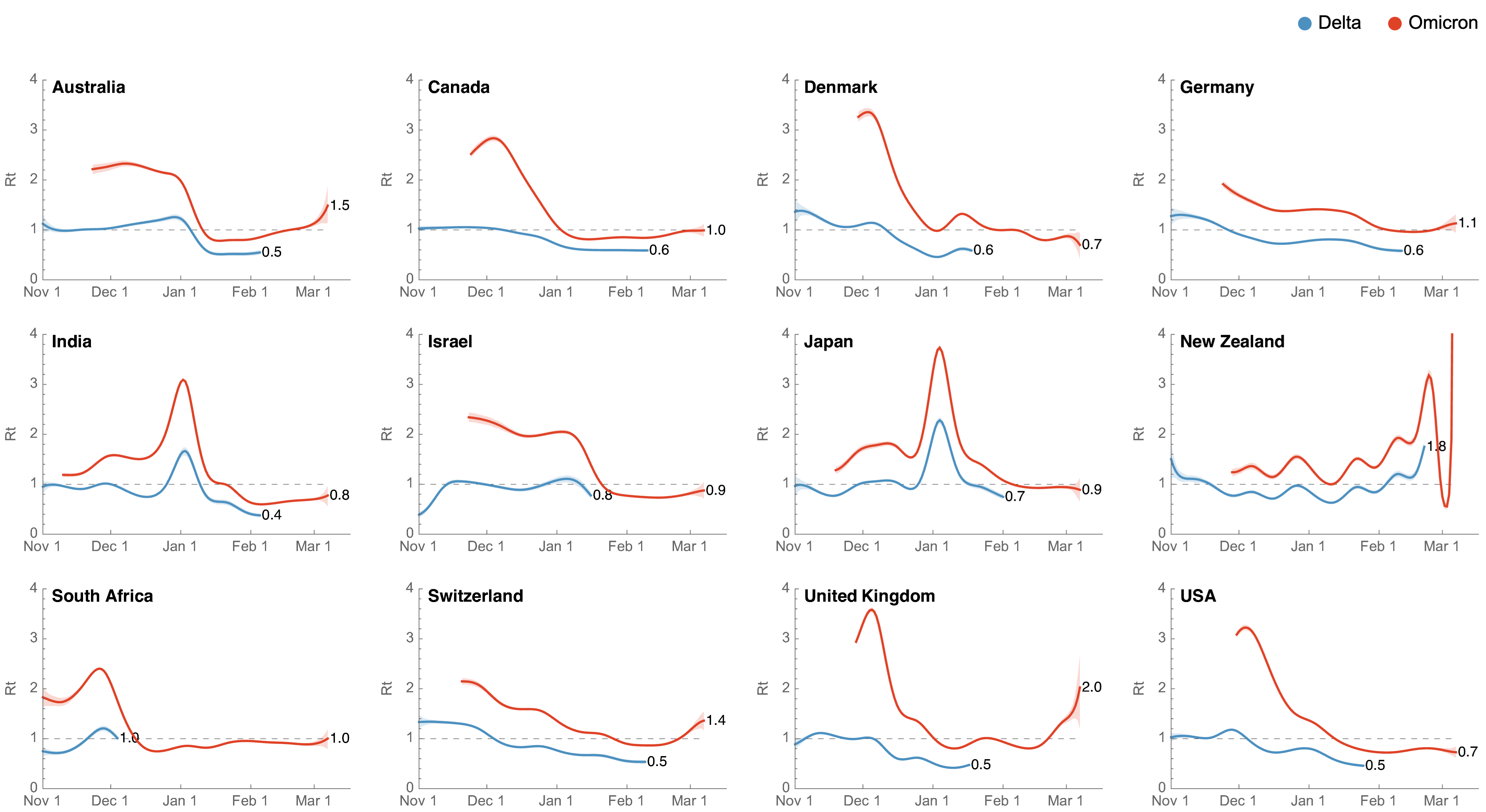
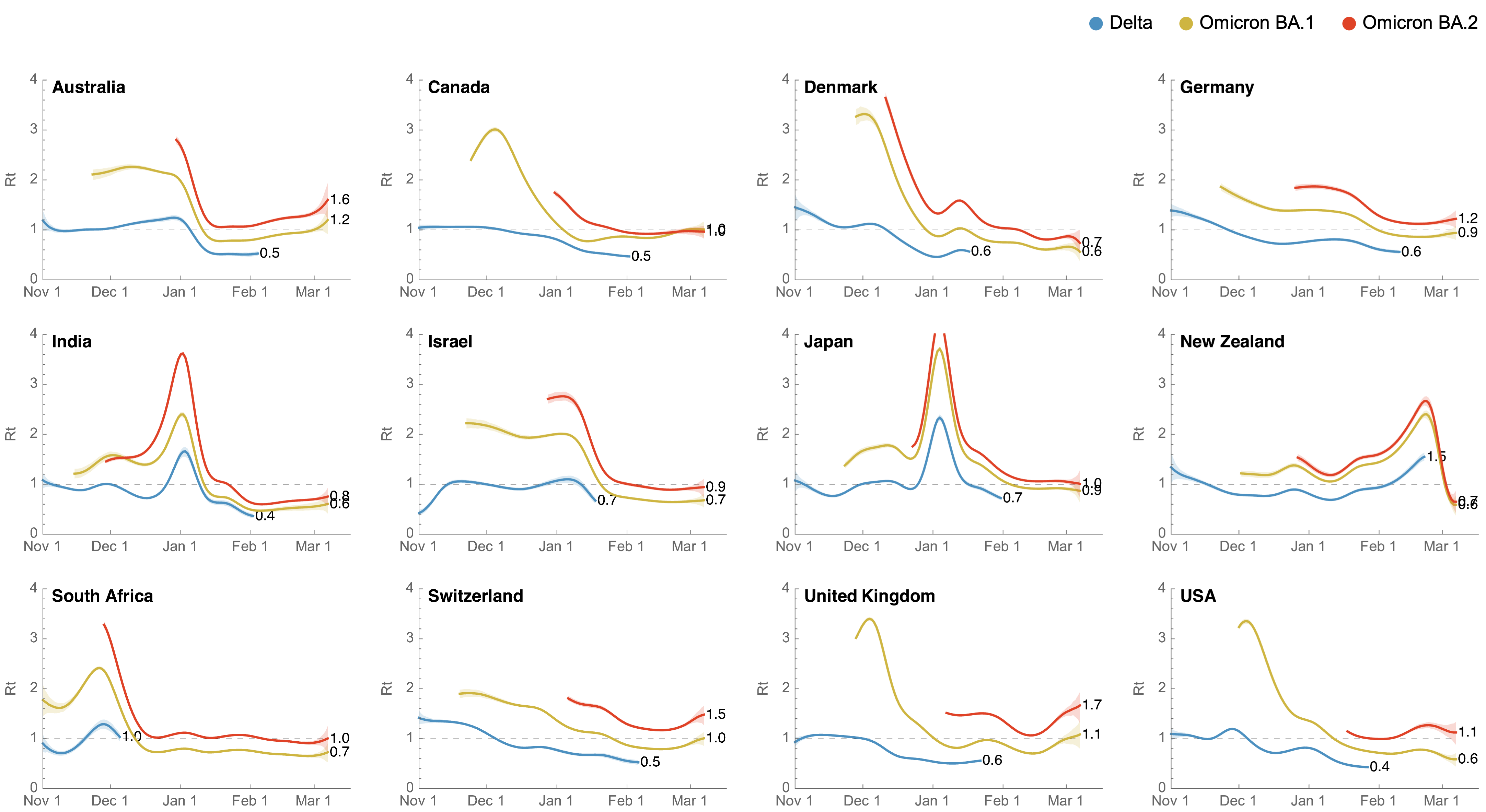
Differences in intrinsic Rt across variants, but all trending downwards
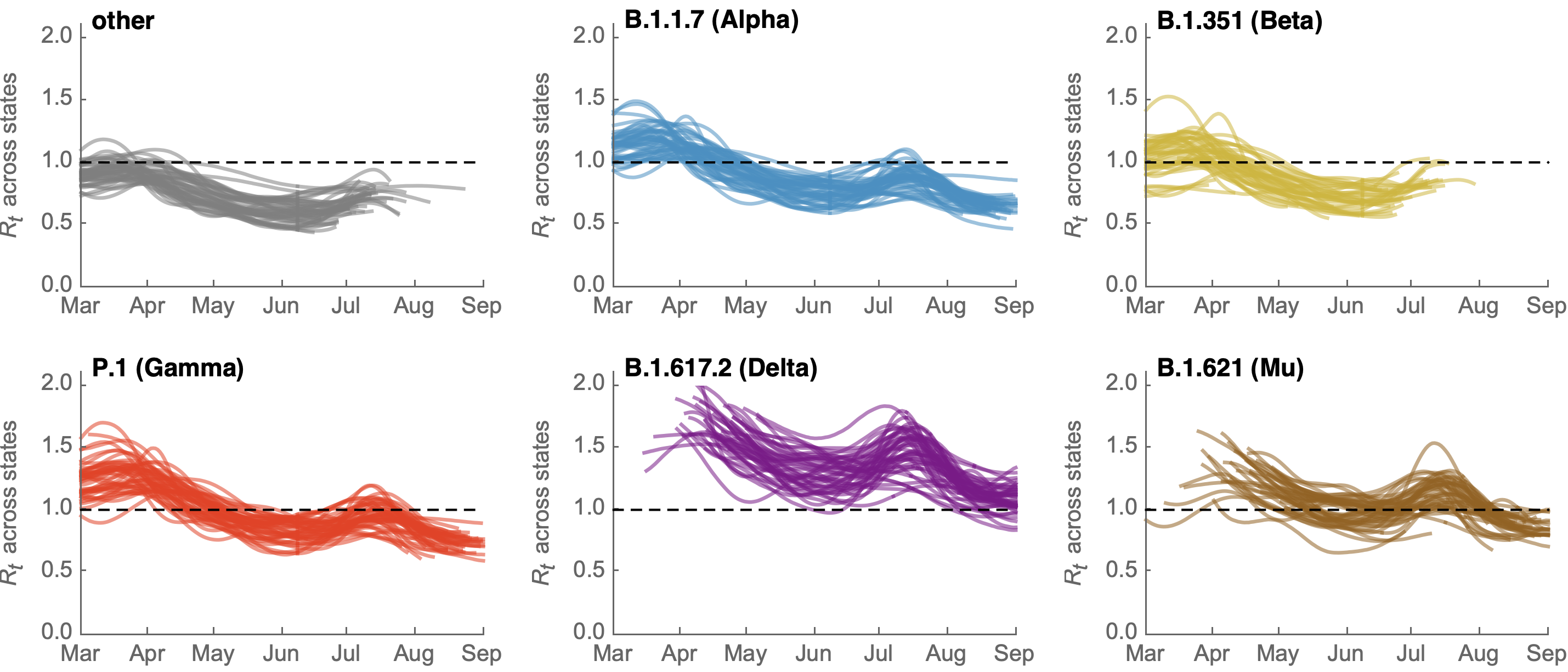
Consistent differences in variant-specific transmission rate across states
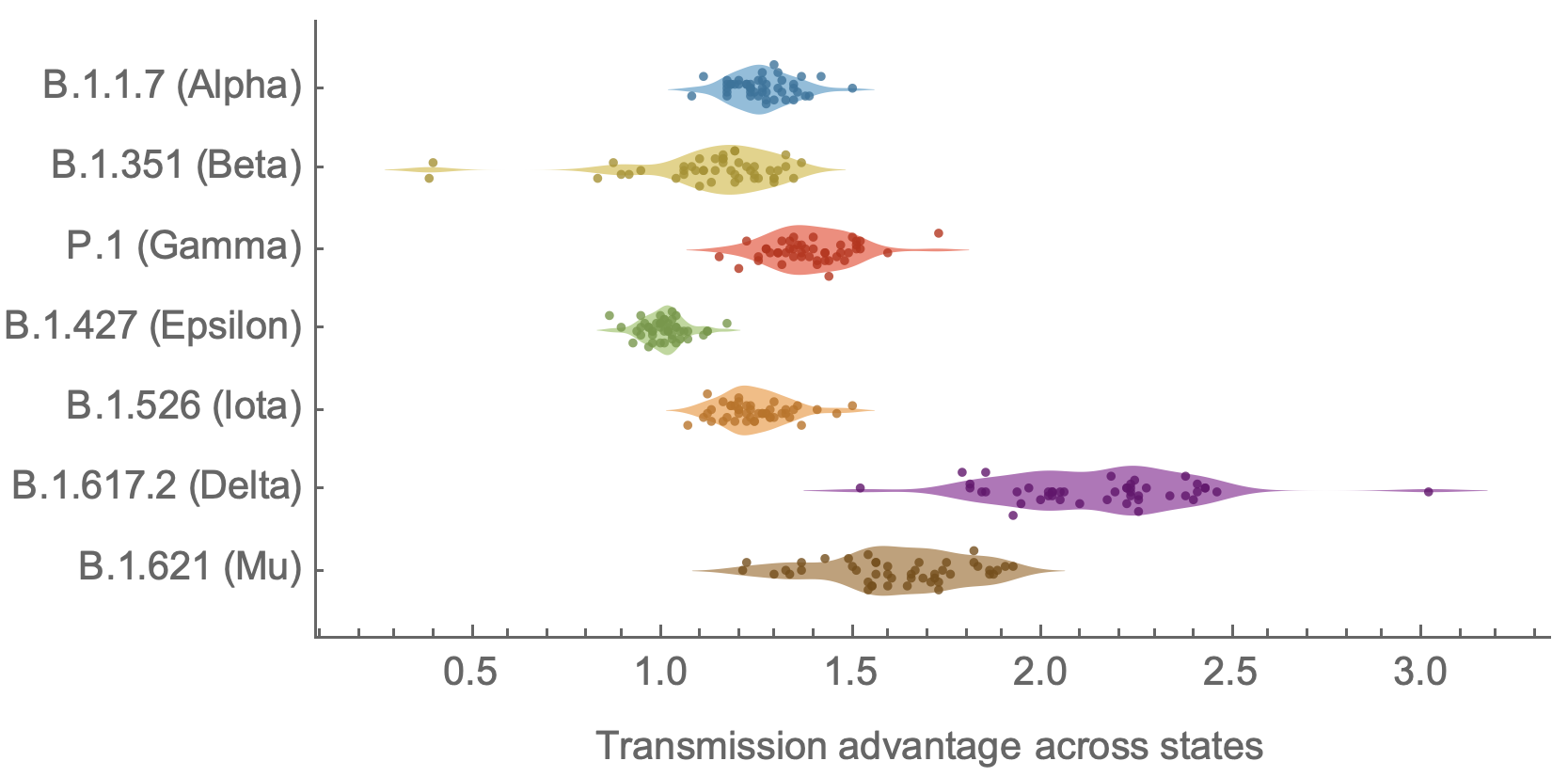
Future work would ideally tie together granular empirical estimates of viral fitness from frequency data together with mutational and phenotypic predictors to learn what is driving variant success
Omicron emergence and spread
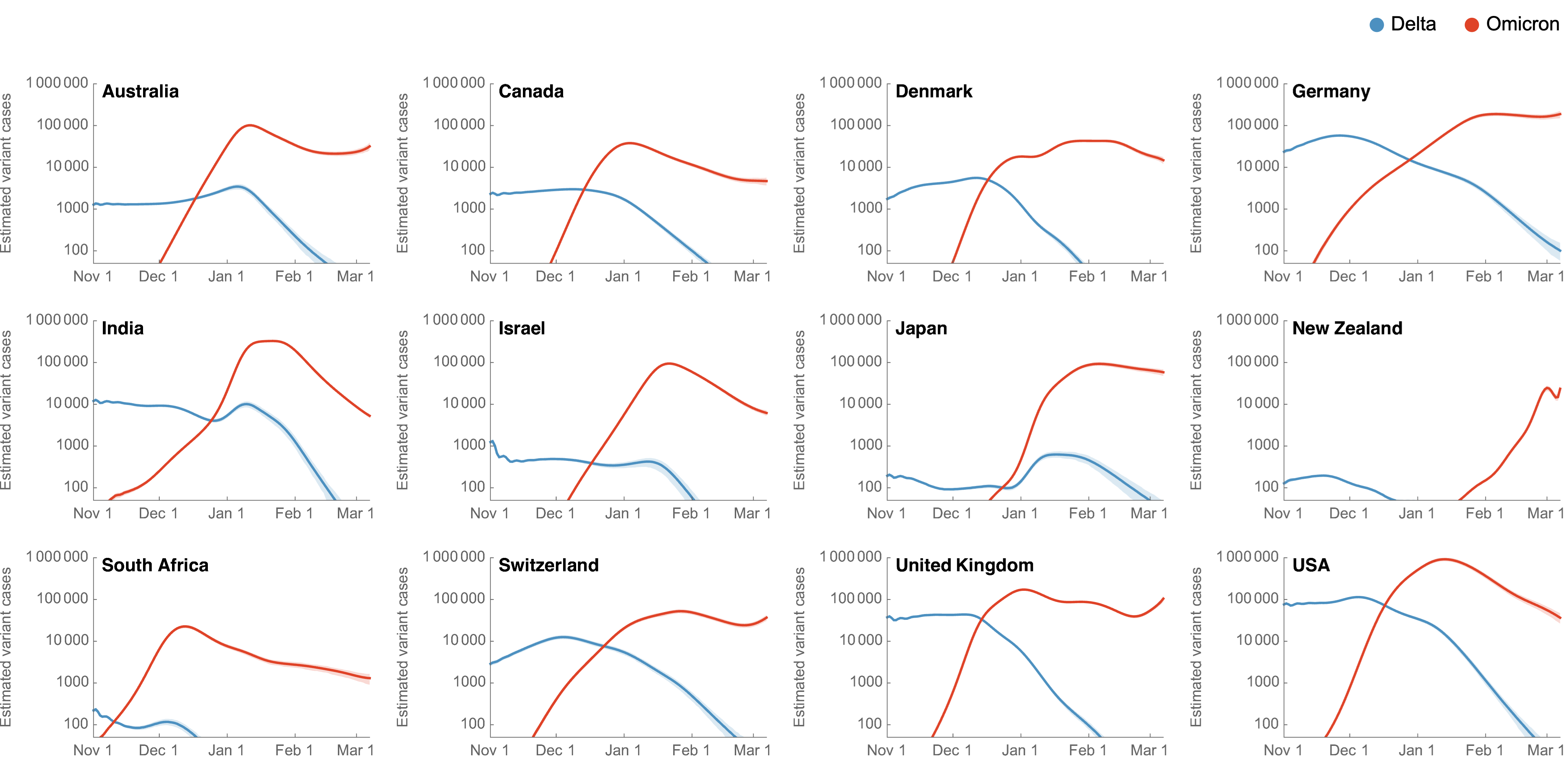
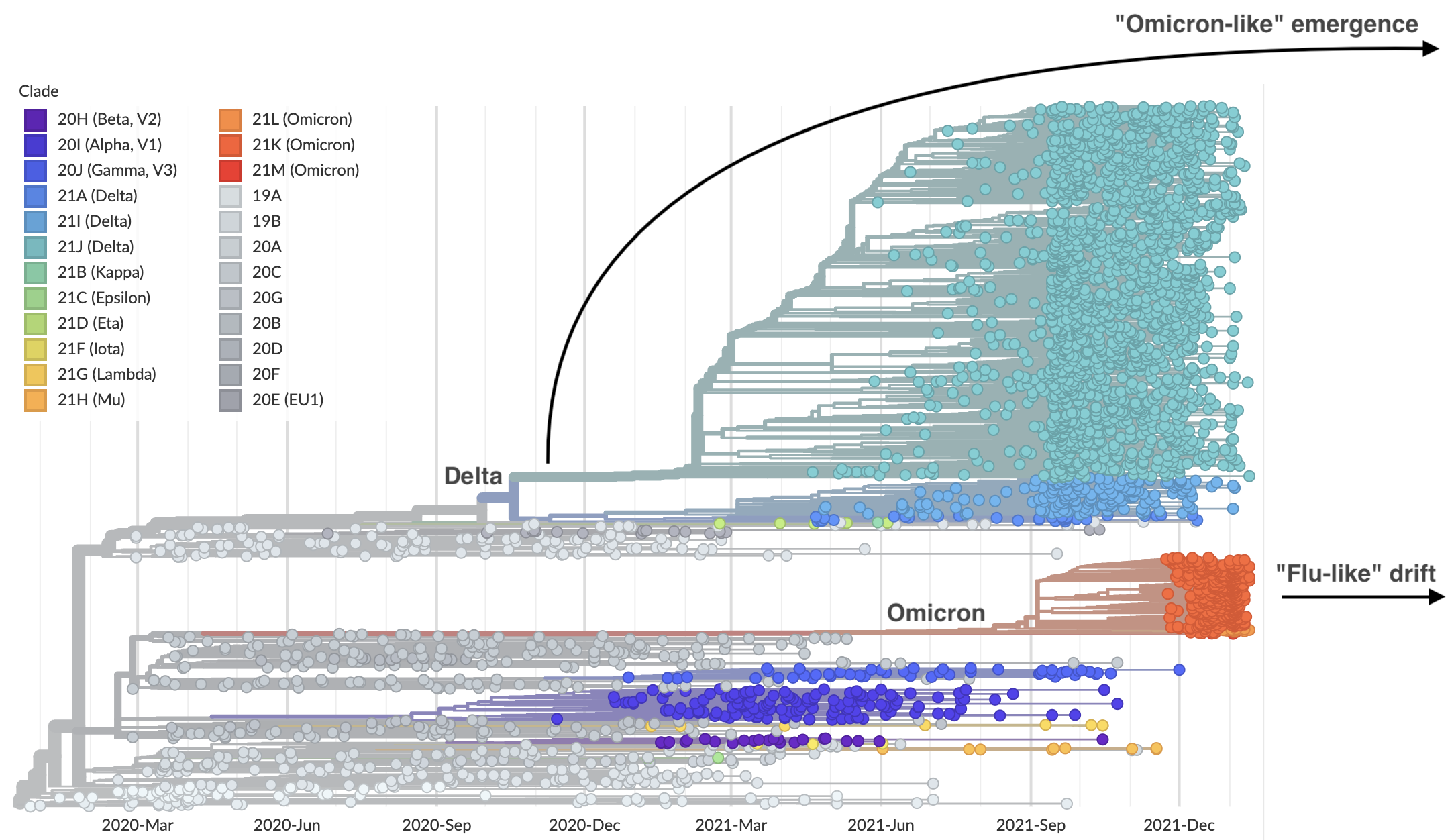
Genetic relationships of SARS-CoV-2 sampled up to Mar 2022
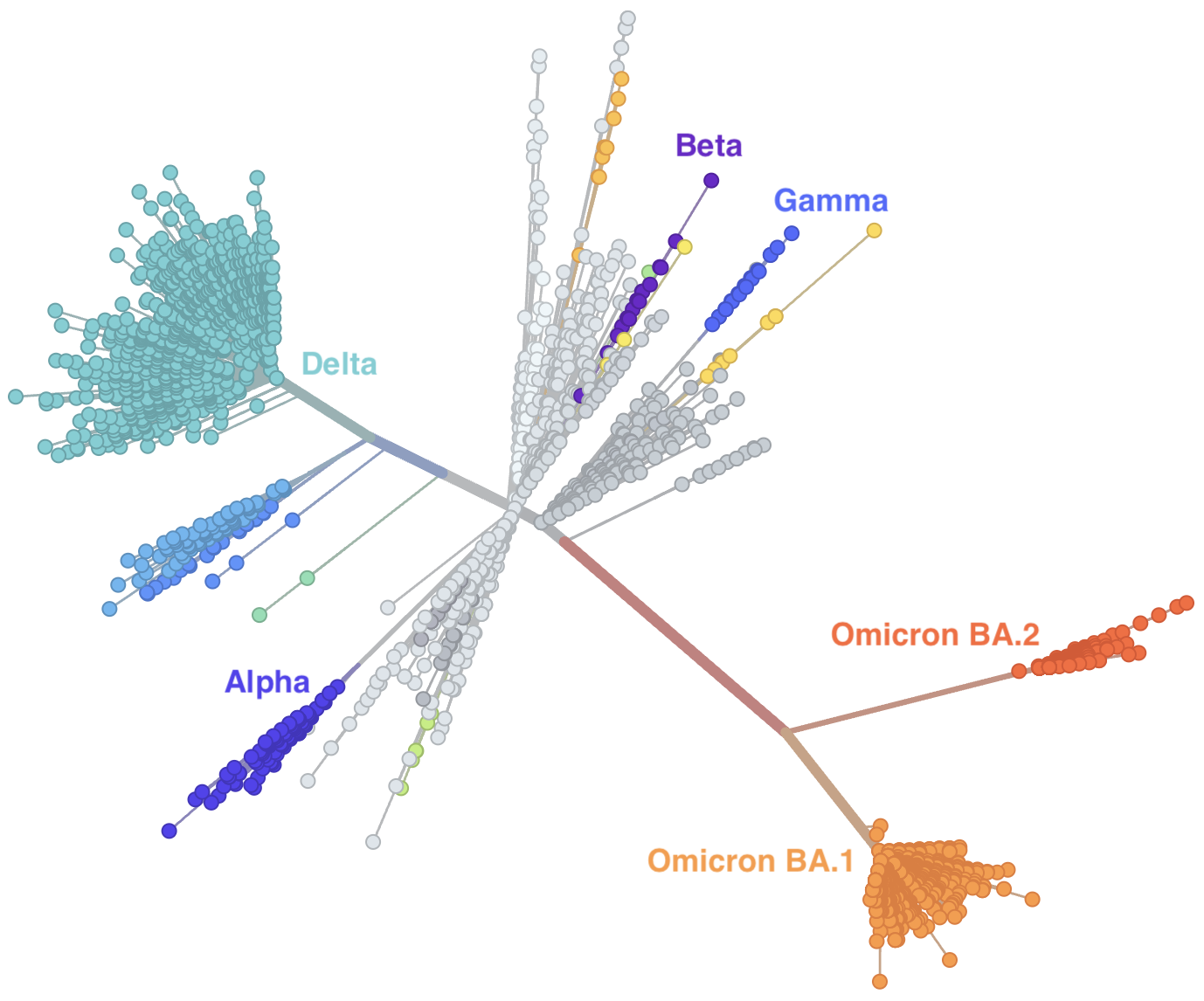
Rapid displacement of existing diversity by Omicron
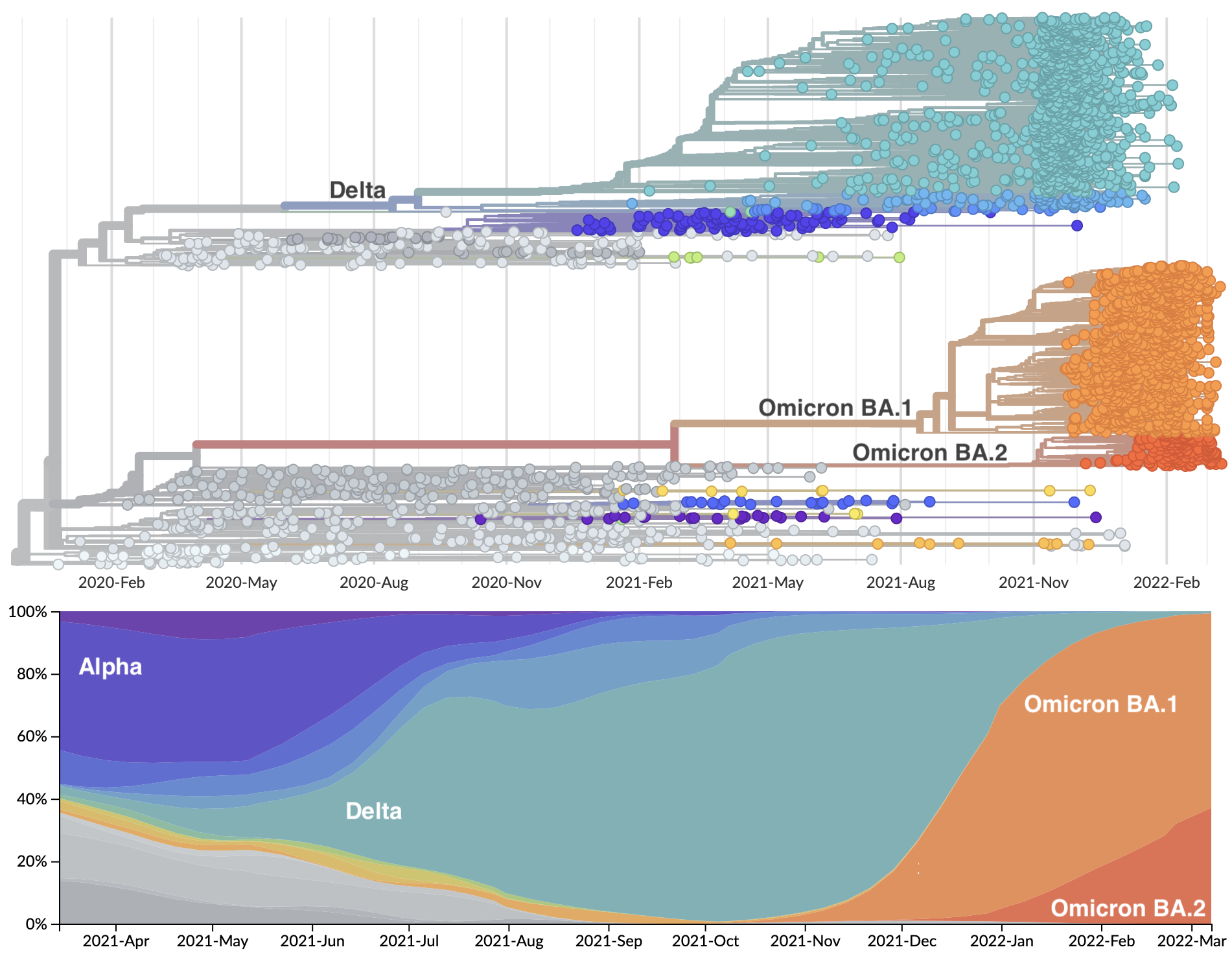
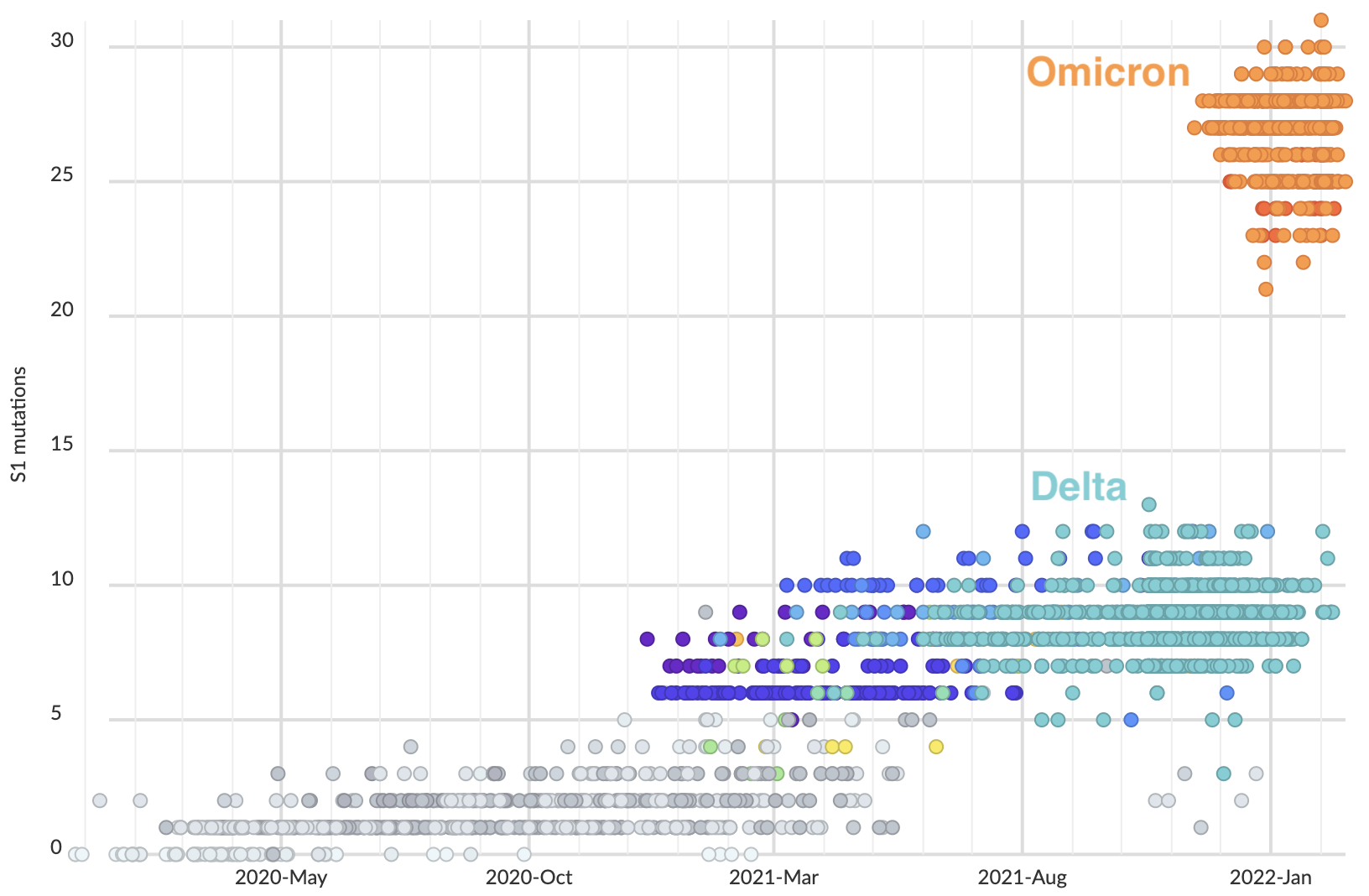
Omicron viruses possess huge excess of mutations in S1
Primary hypotheses for Omicron's origin
-
(More likely) Evolution during chronic infection in an immunocompromised individual
- Supported by large excess of S1 mutations, but paucity of non-spike mutations
- Hard to explain simultaneous BA.1, BA.2 and BA.3 sublineages otherwise
-
(Less likely) Human to animal spillover mid-2020 and spillback in late 2021
- Mutational spectra and particular mutations such as 493R and 498R suggest rodent intermediary (Wei et al)
S1 evolved at a rate of 12 amino acid changes per year in 2021
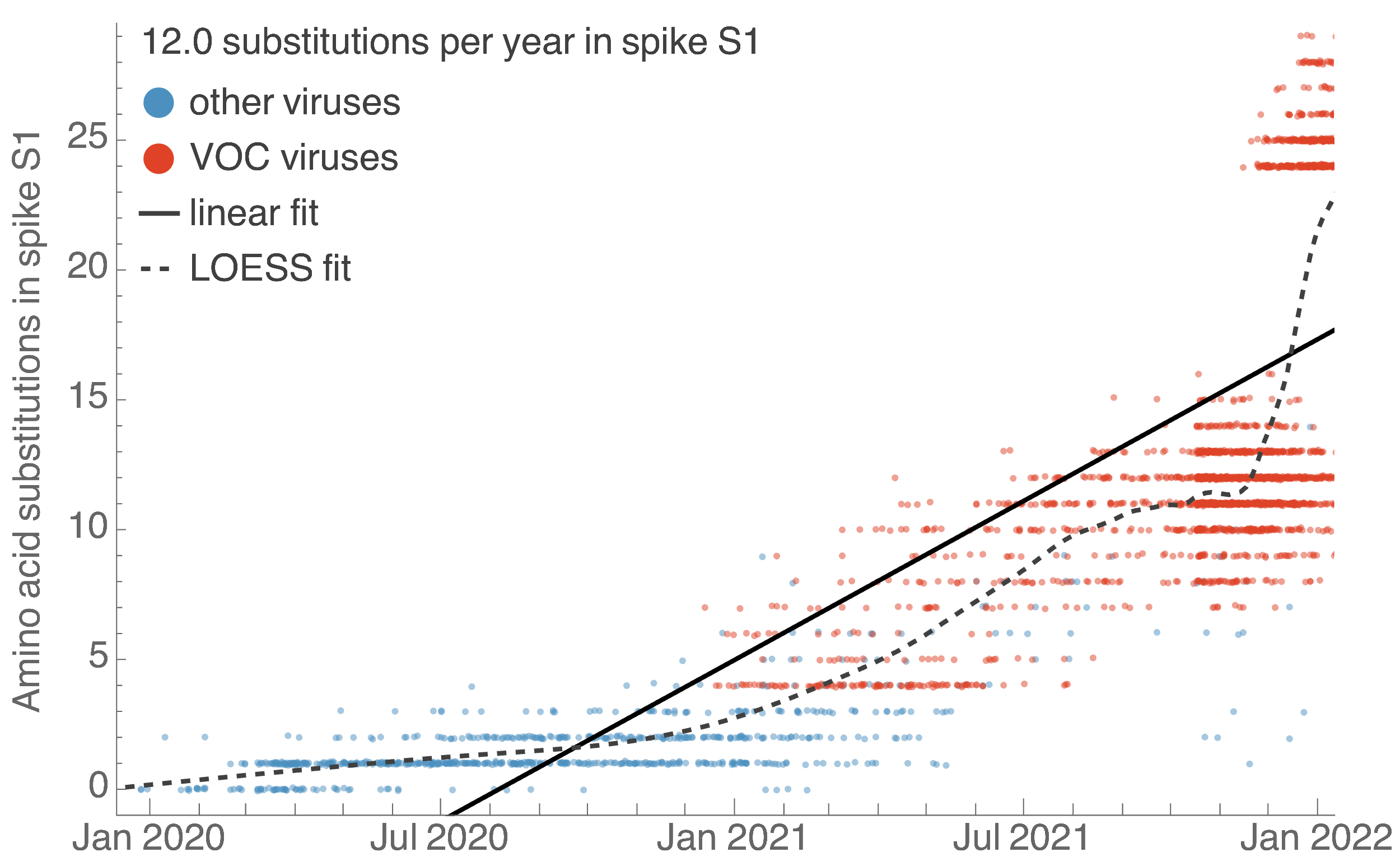
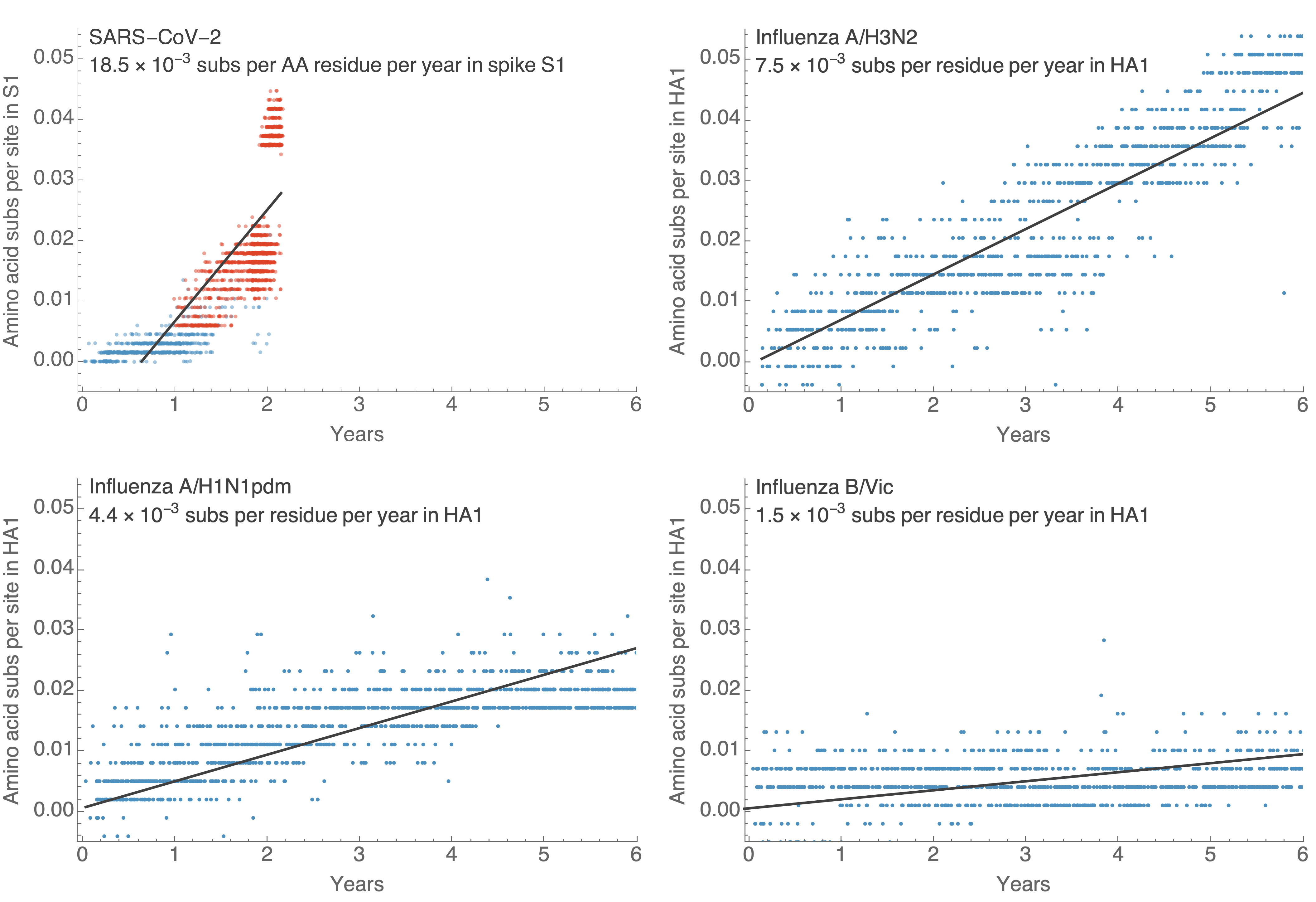
This is remarkably fast relative to seasonal influenza
Omicron spike mutations substantially drop VE against infection
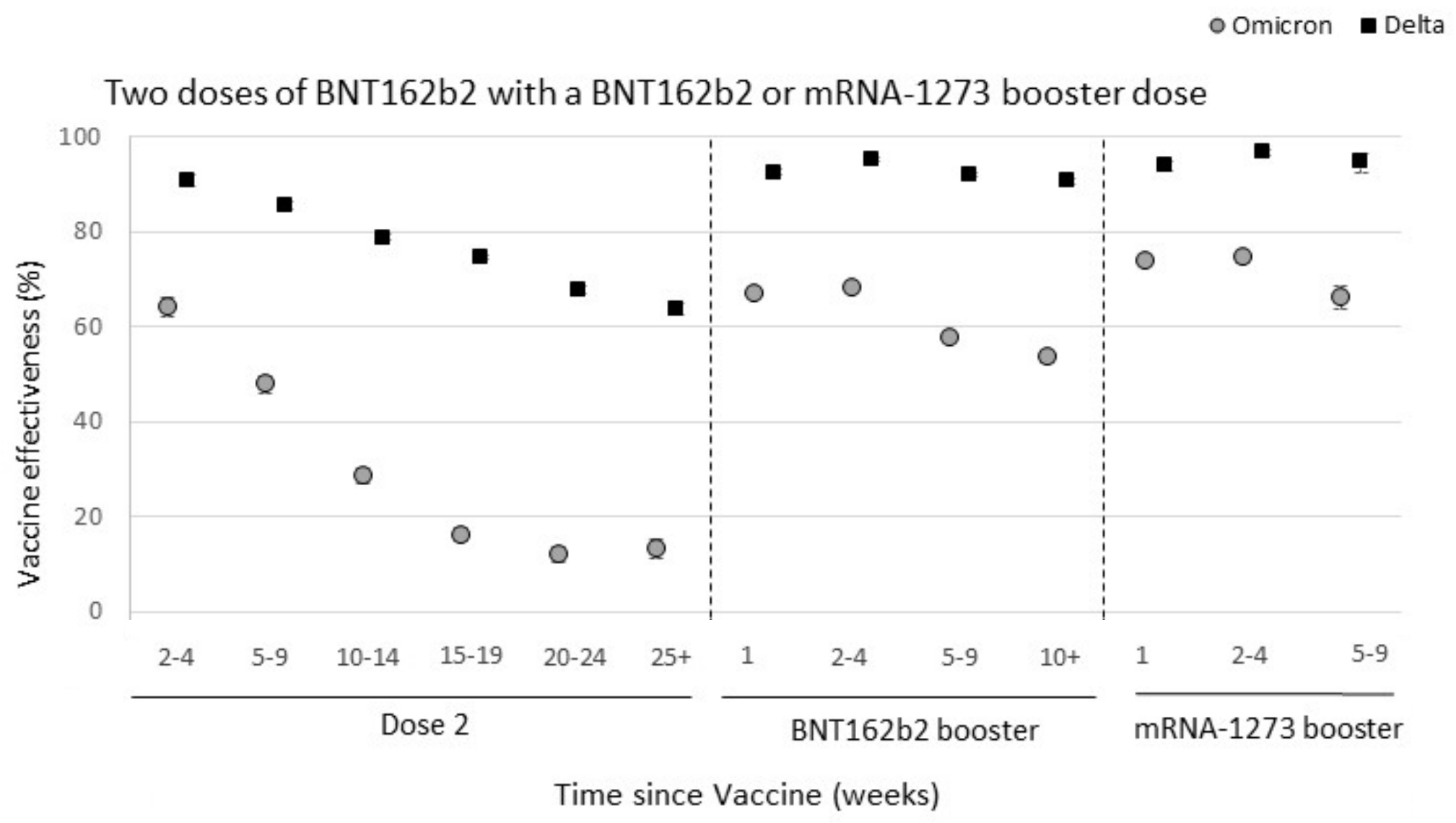
Significant immune escape drove large-scale epidemics
Starting Rt approached that of the initial wave in spring 2020
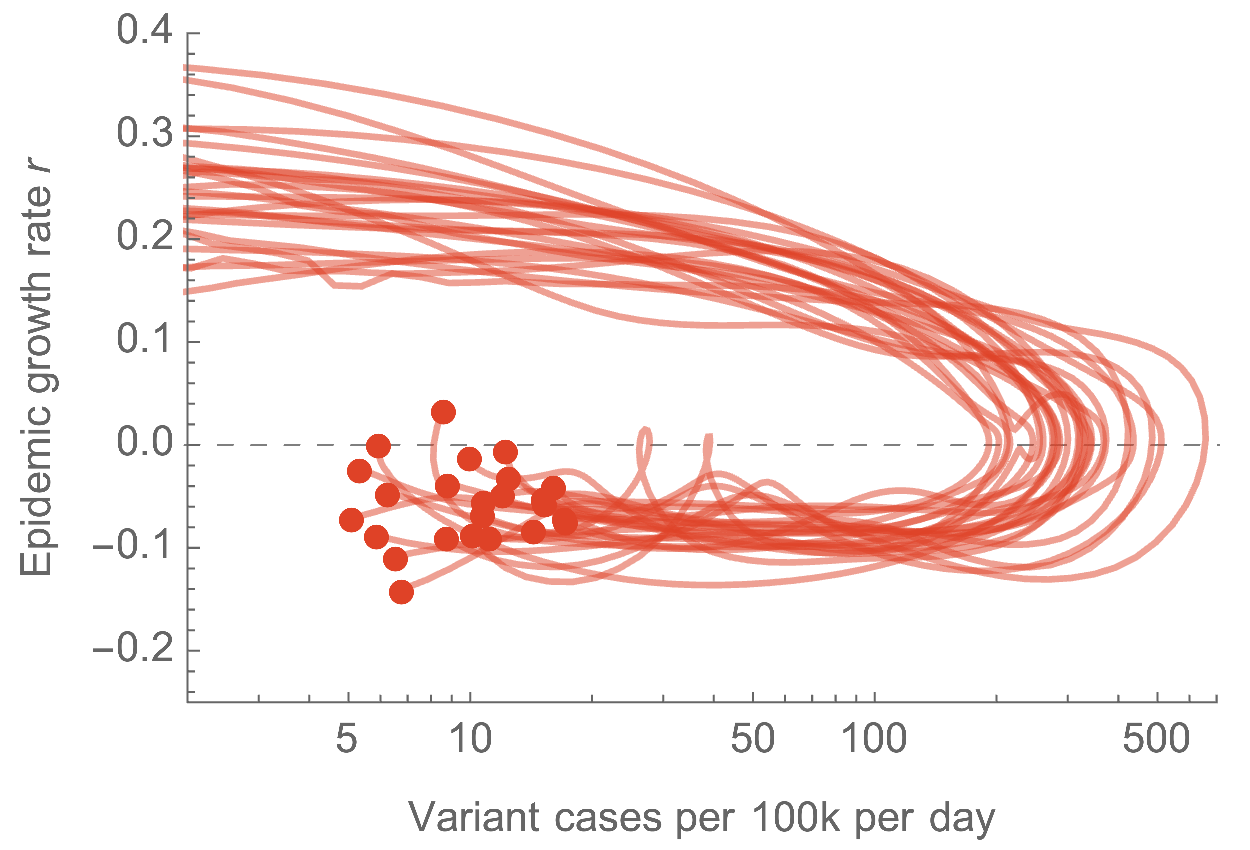
Omicron attack rate
- We estimate US has seen 9.8% of the population as confirmed cases of Omicron through Mar 3, with the large majority accumulating after Dec 15
- Assuming a case detection rate of 1 in 5 infections, we estimate almost 50% of the US infected with Omicron, most in the span of ~10 weeks
Similar epidemics across US states with downturn driven by immunity
Omicron BA.2 has been steadily displacing BA.1 across the world
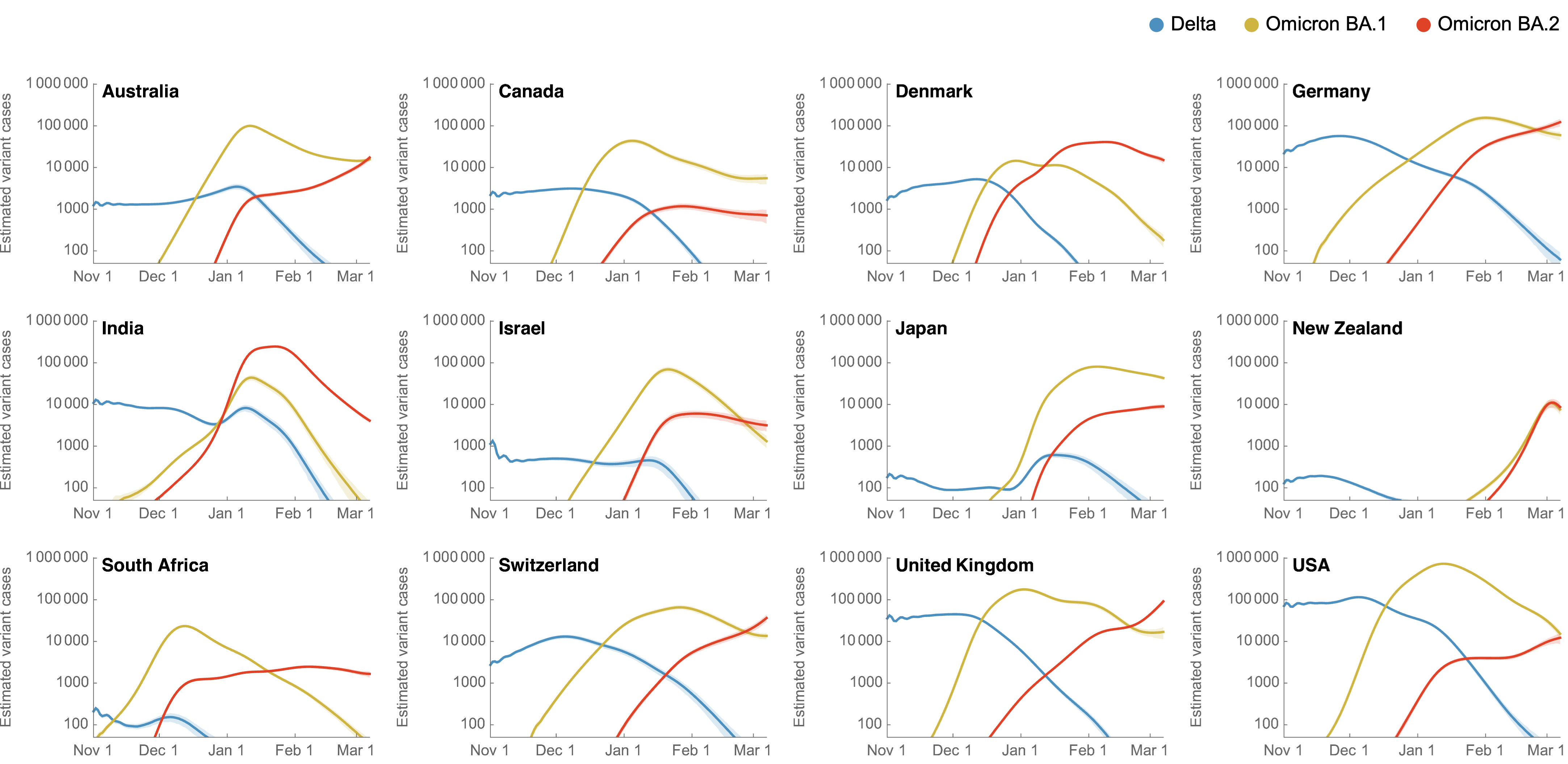
BA.2 shows a transmission advantage of ~30% over BA.1
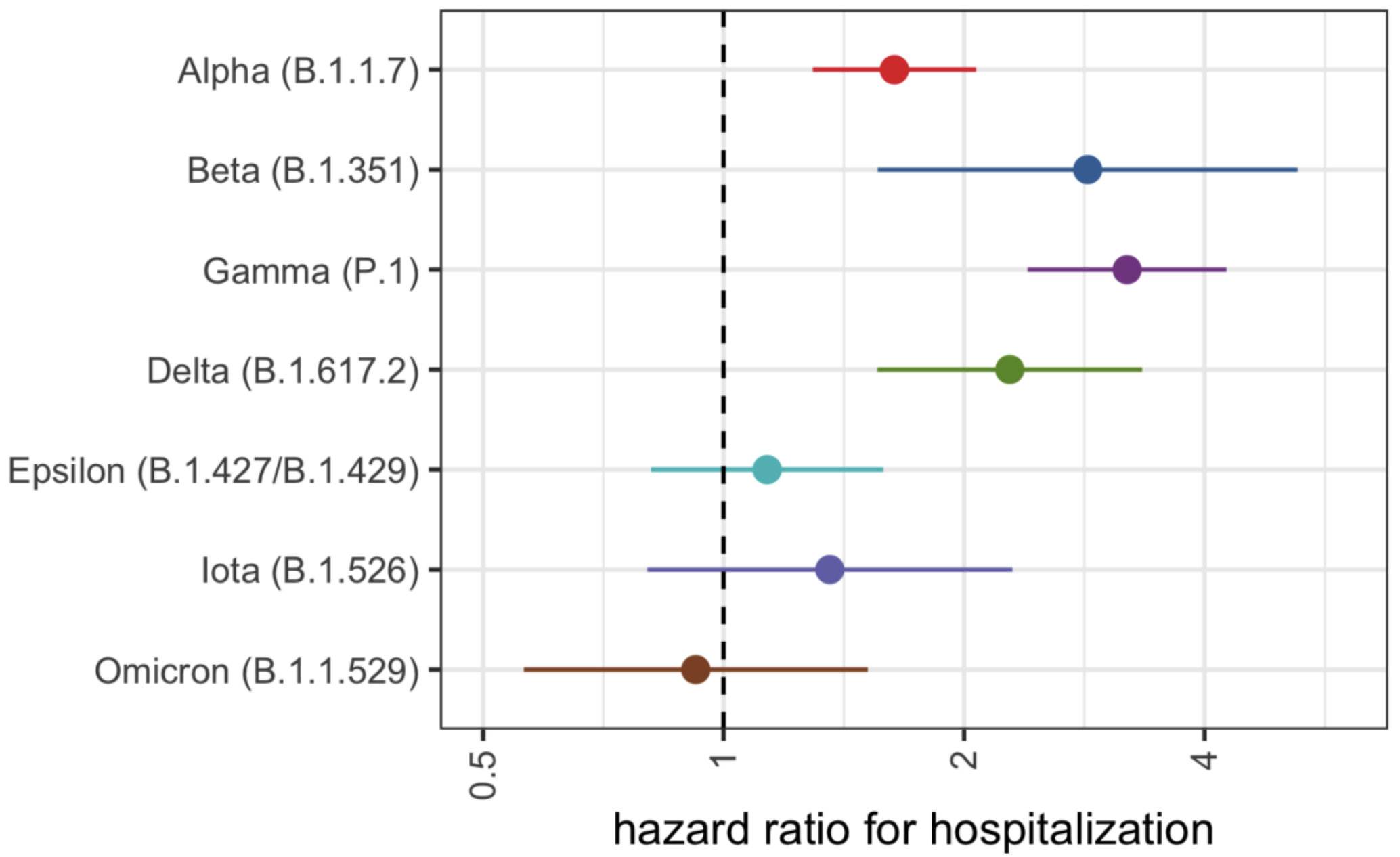
Reduced intrinsic severity relative to other VOC viruses
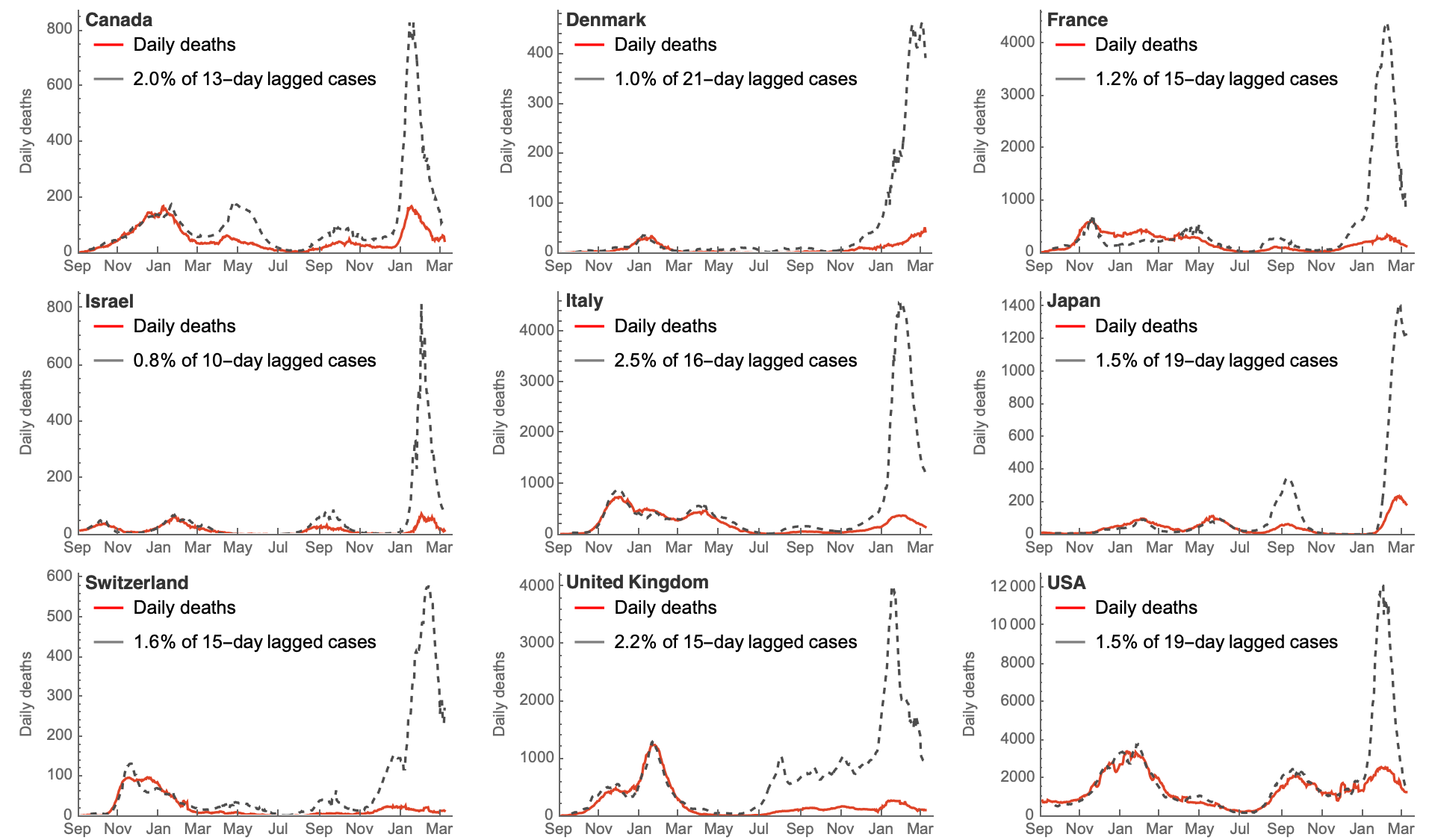
Immunity and reduced intrinsic severity has dropped CFR
Future perspectives
SARS-CoV-2 will continue to evolve to escape population immunity, though with multiple potential avenues
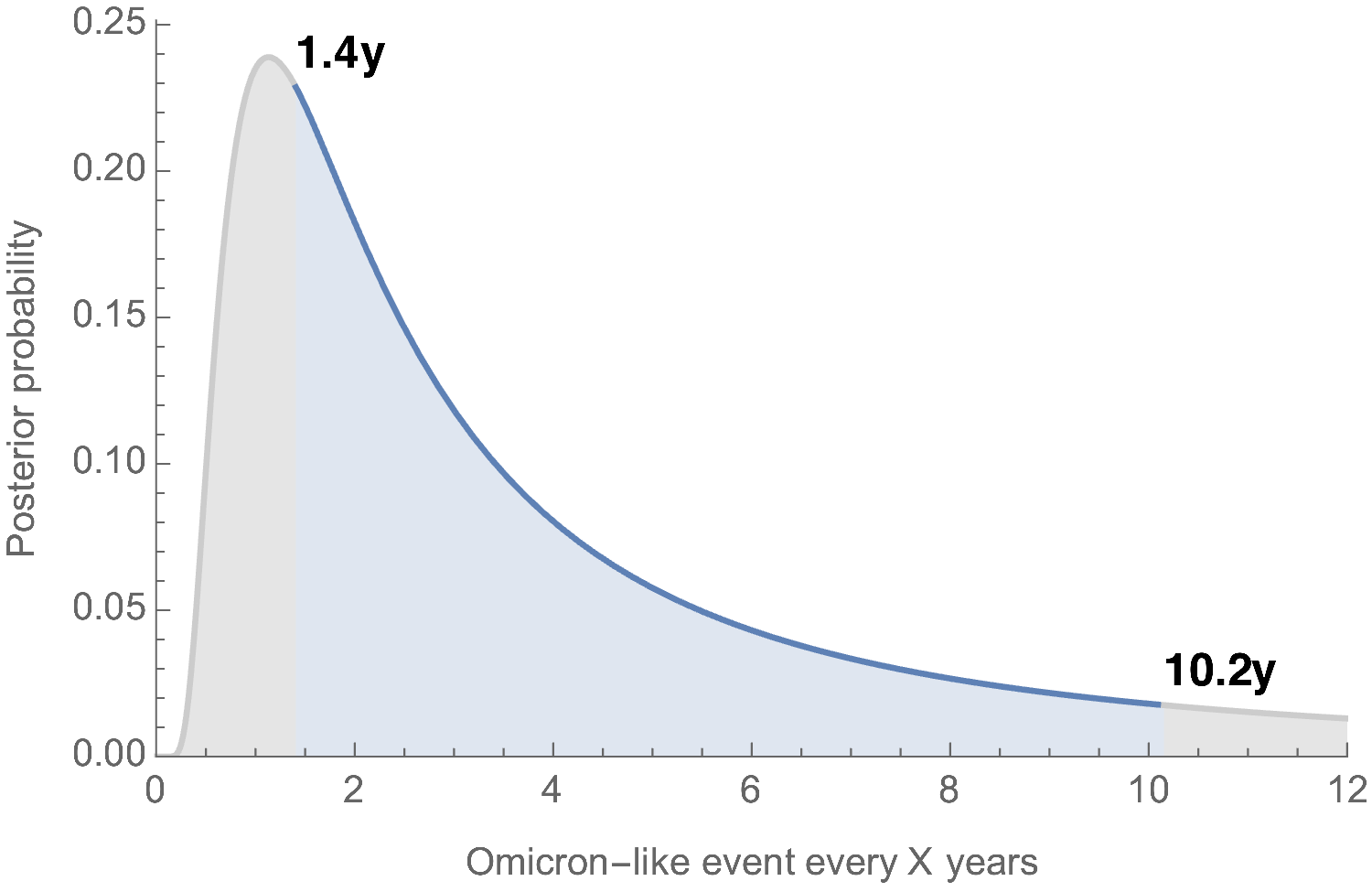
With 1 observation in ~2.3 years of virus evolution, it's currently unclear how common Omicron-like events will be
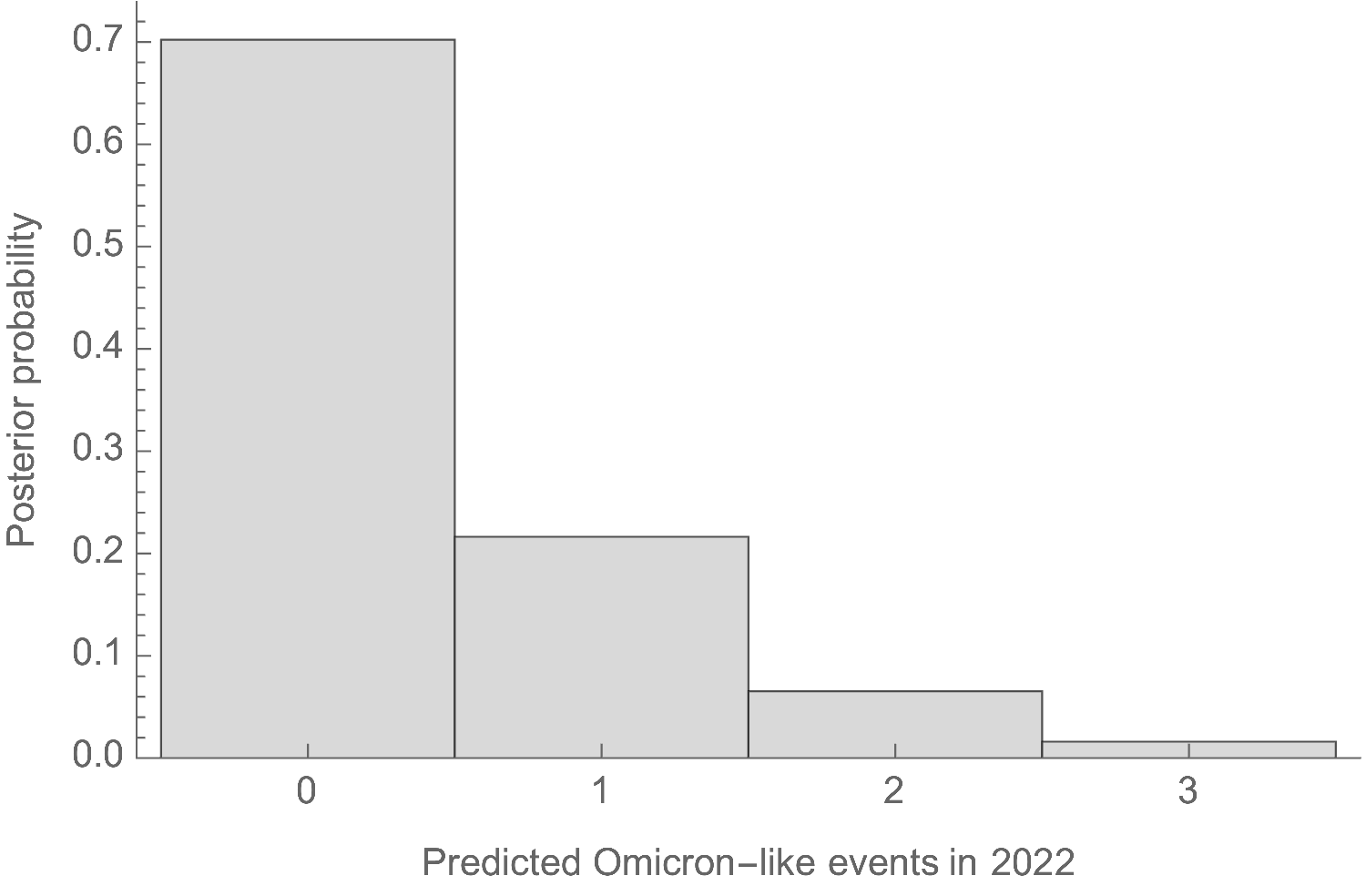
This rate distribution gives a naive prediction of Omicron-like emergence events occurring in the rest of 2022
Likely scenarios over the next 12 months
- Evolution within Omicron BA.2 to further increase intrinsic transmission and to escape from Omicron-derived immunity. This scenario sees lower attack rates with 2022-2023 epidemic driven by drift + waning + seasonality.
- Another Omicron-like emergence event in which a chronic infection initiated in ~2021 incubates a new wildly divergent virus. This scenario sees high attack rates with epidemic driven by variant emergence.
Broad expectations of endemicity based on comparison with flu
- R0 of Delta/Omicron is perhaps ~6 compared to R0 of flu of ~2. At same rates of evolution and waning expect more COVID circulation.
- Before Omicron rates of evolution in SARS-CoV-2 S1 had been roughly equal to H3N2 flu. Omicron equivalent to 5 years of H3N2 evolution.
- Infection fatality rate (IFR) of COVID is roughly comparable to flu once you have prior immunity.
1, 2 and 3 suggest a virus that circulates at higher levels than flu, but individual infections aren't much more severe in terms of mortality than H3N2 influenza.
Seasonality suggests winter "COVID season", but Omicron-like events could overcome seasonality.
Key parameters are seasonal attack rate and IFR
- Assume seasonal attack rate of 40%, with Omicron-like emergence driving 80% some years.
- Assume IFR of 0.05% (compared to early pandemic IFR of 0.5%), but new variant could push this higher.
- This would give somewhere around 60K yearly deaths at endemicity, but potential for variant emergence to push this over 100K.
Acknowledgements
SARS-CoV-2 genomic epi: Data producers from all over the world, GISAID and the Nextstrain team
Bedford Lab:
![]() John Huddleston,
John Huddleston,
![]() James Hadfield,
James Hadfield,
![]() Katie Kistler,
Katie Kistler,
![]() Louise Moncla,
Louise Moncla,
![]() Maya Lewinsohn,
Maya Lewinsohn,
![]() Thomas Sibley,
Thomas Sibley,
![]() Jover Lee,
Jover Lee,
![]() Cassia Wagner,
Cassia Wagner,
![]() Miguel Paredes,
Miguel Paredes,
![]() Nicola Müller,
Nicola Müller,
![]() Marlin Figgins,
Marlin Figgins,
![]() Denisse Sequeira,
Denisse Sequeira,
![]() Victor Lin,
Victor Lin,
![]() Jennifer Chang
Jennifer Chang






