SARS-CoV-2 evolutionary dynamics
Trevor Bedford (@trvrb)
Associate Professor, Fred Hutchinson Cancer Research Center
12 Nov 2021
Emerging Variants Meeting
Massachusetts Consortium on Pathogen Readiness
Slides at: bedford.io/talks
1. Current circulation patterns
2. Assessing adaptive evolution
3. Variant transmission dynamics
4. Potential for epistasis
5. Future perspectives
Current circulation patterns
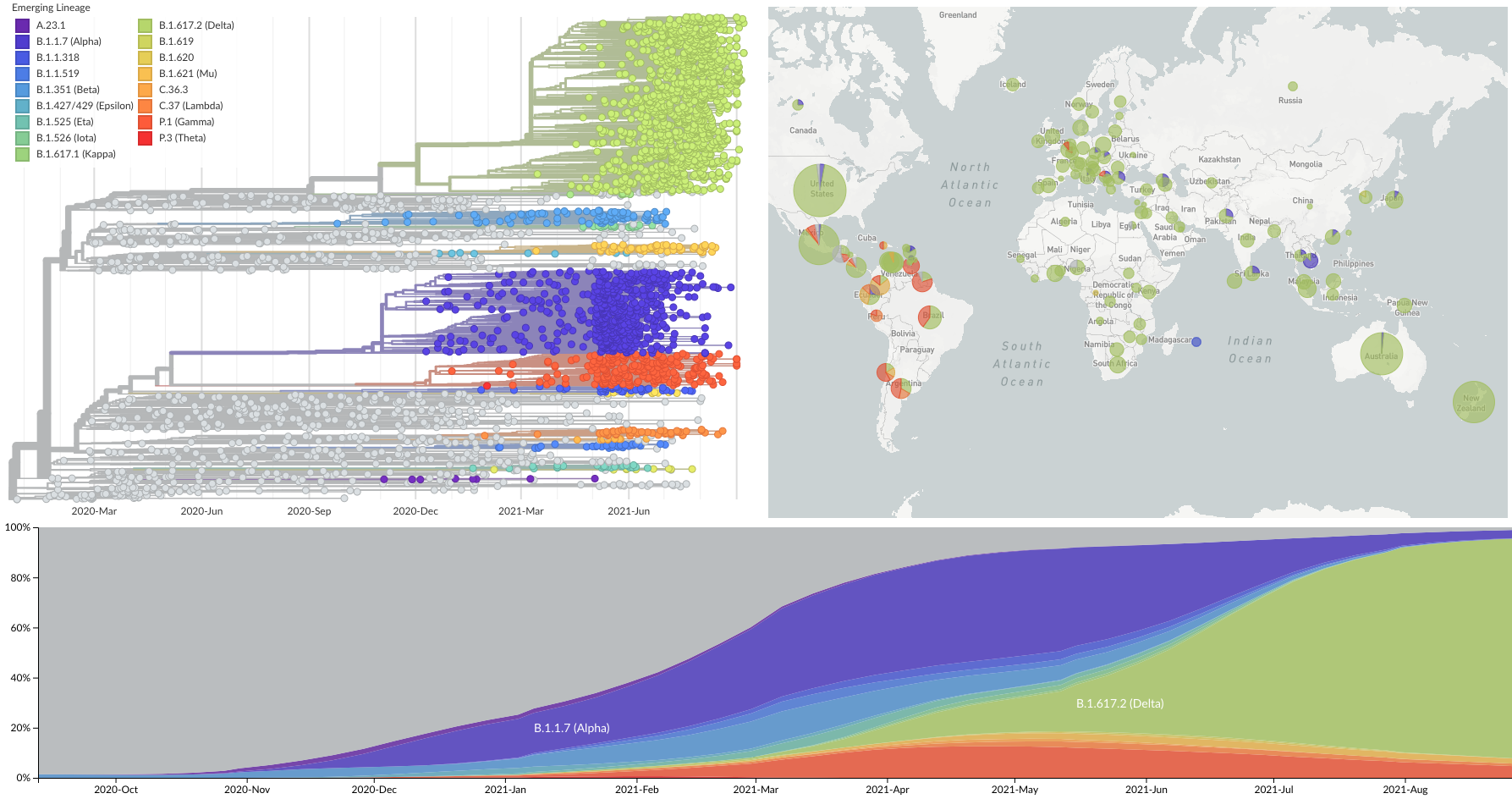
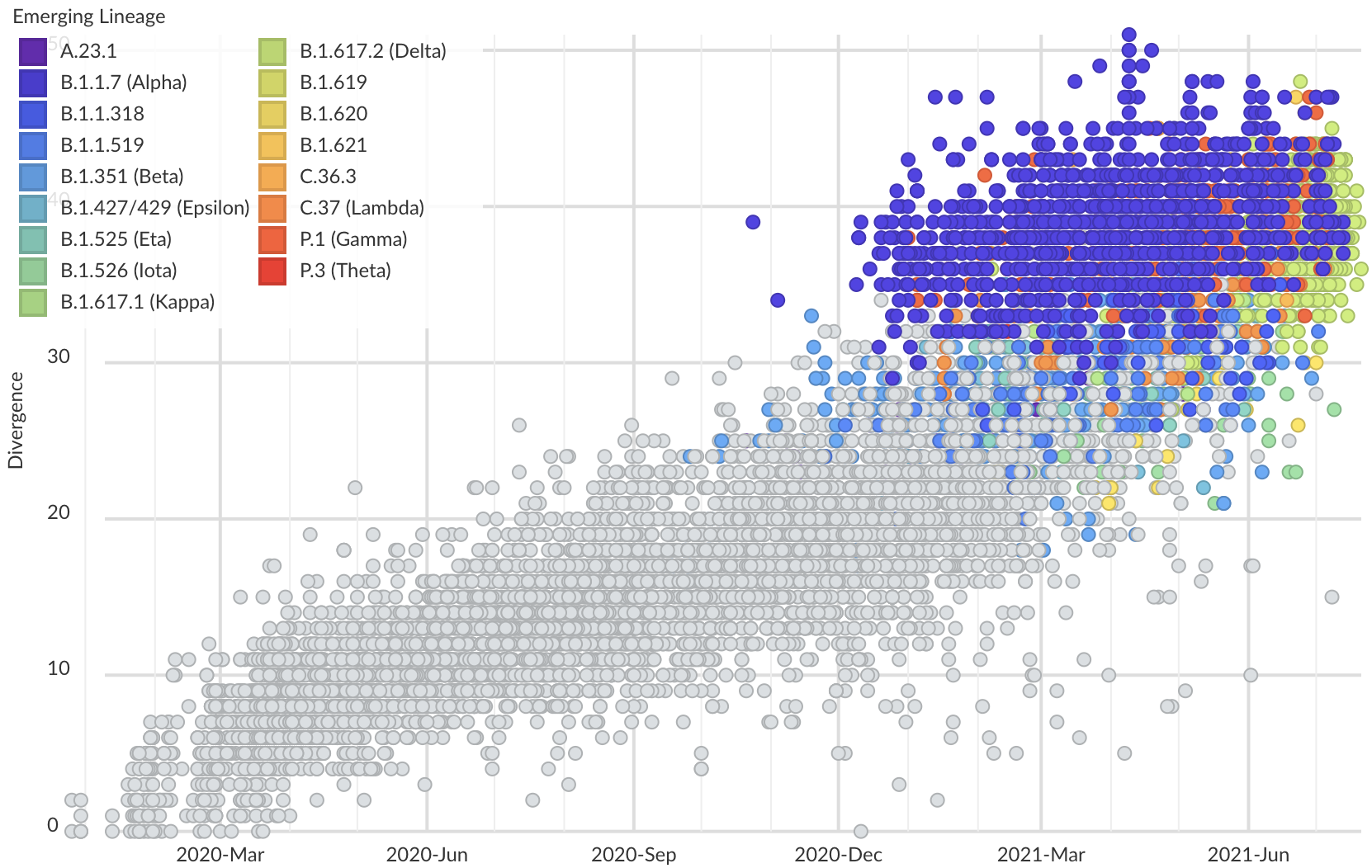
Spread of VOC / VOI lineages across the world
Delta is outcompeting other variants and is on track to sweep
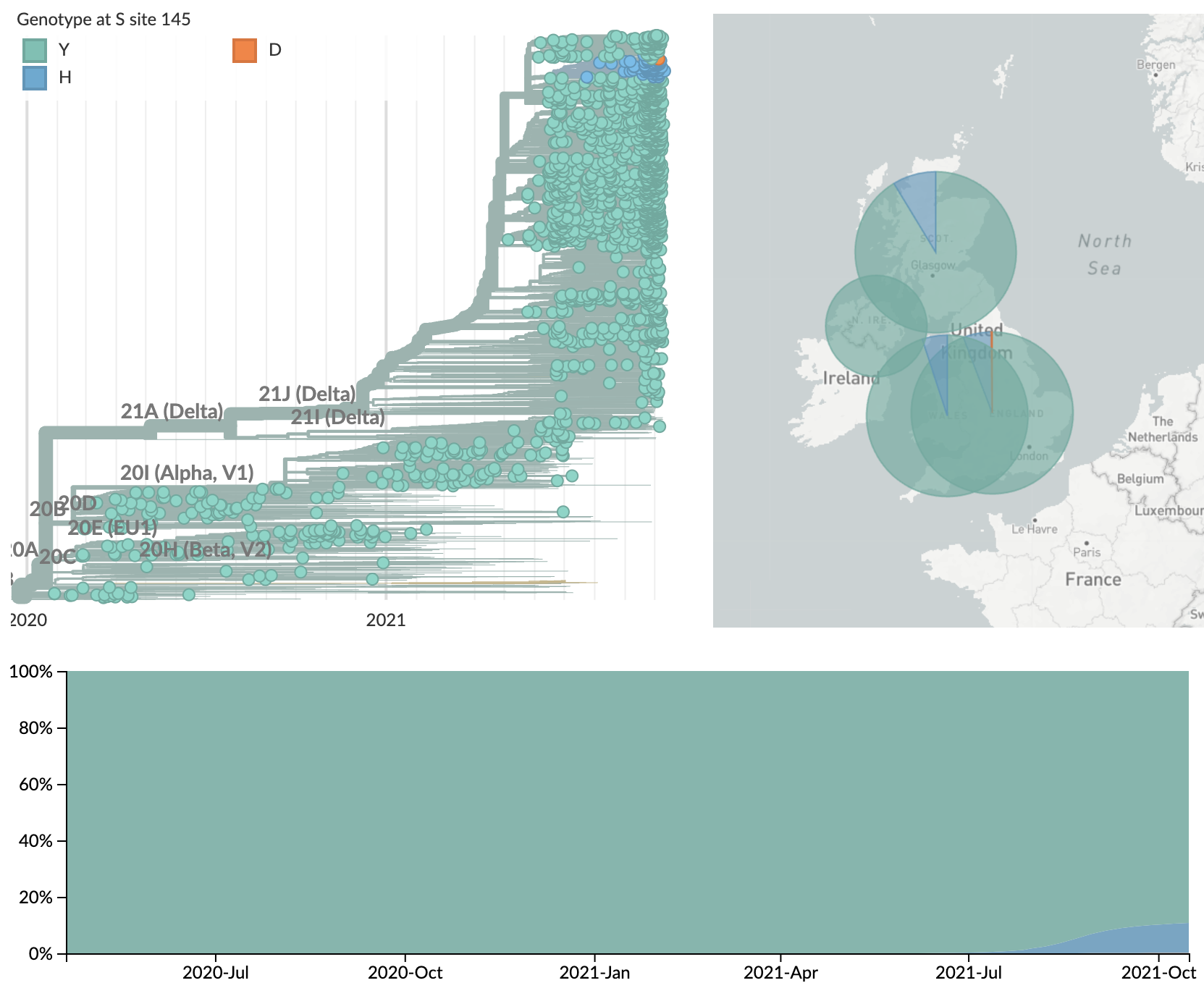
Locally emerging sublineages such as AY.4.2 with spike mutations 145H and 222V
Variants show excess mutations across the genome
But show most substantial excess in the S1 domain of spike
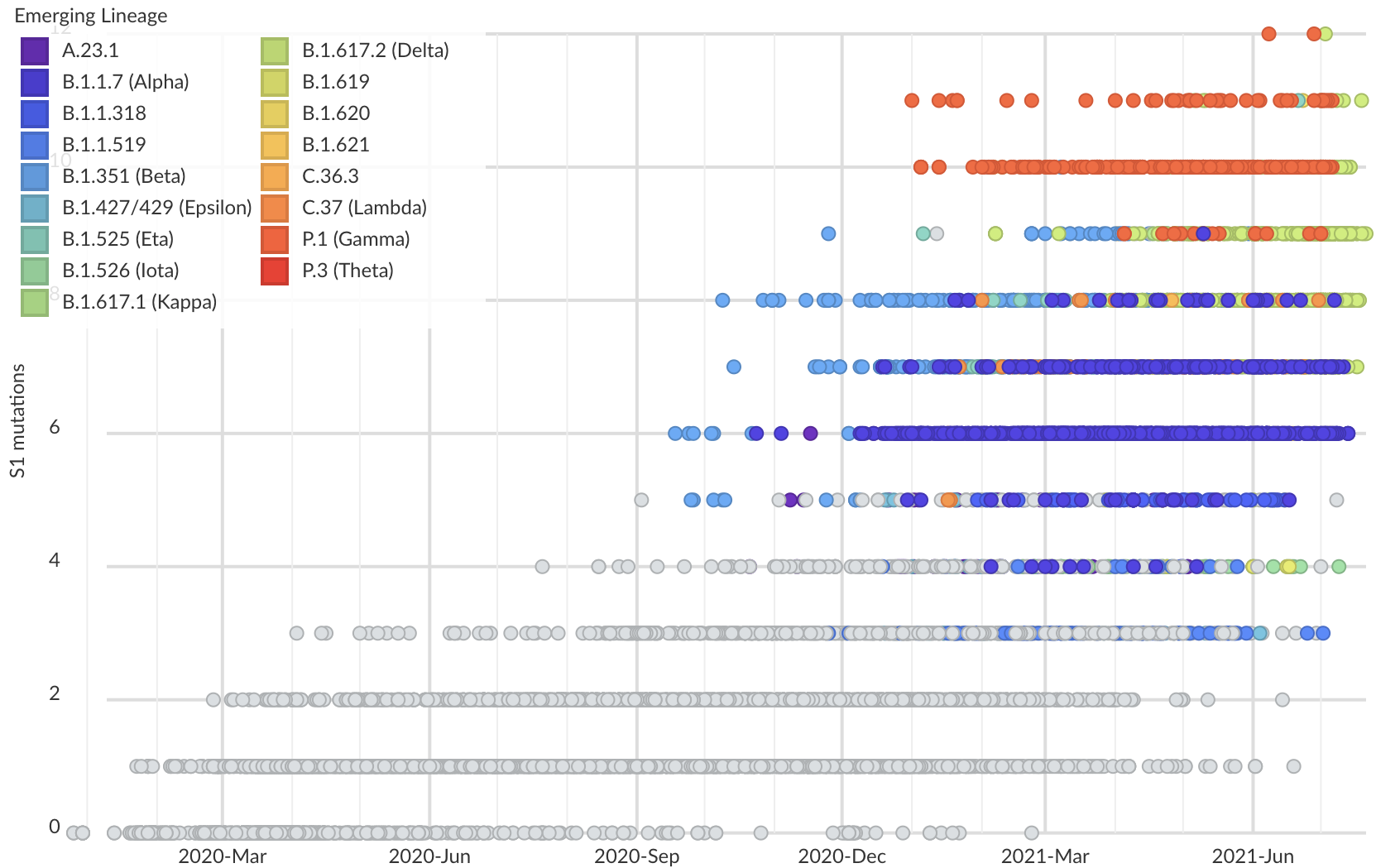
Assessing adaptive evolution
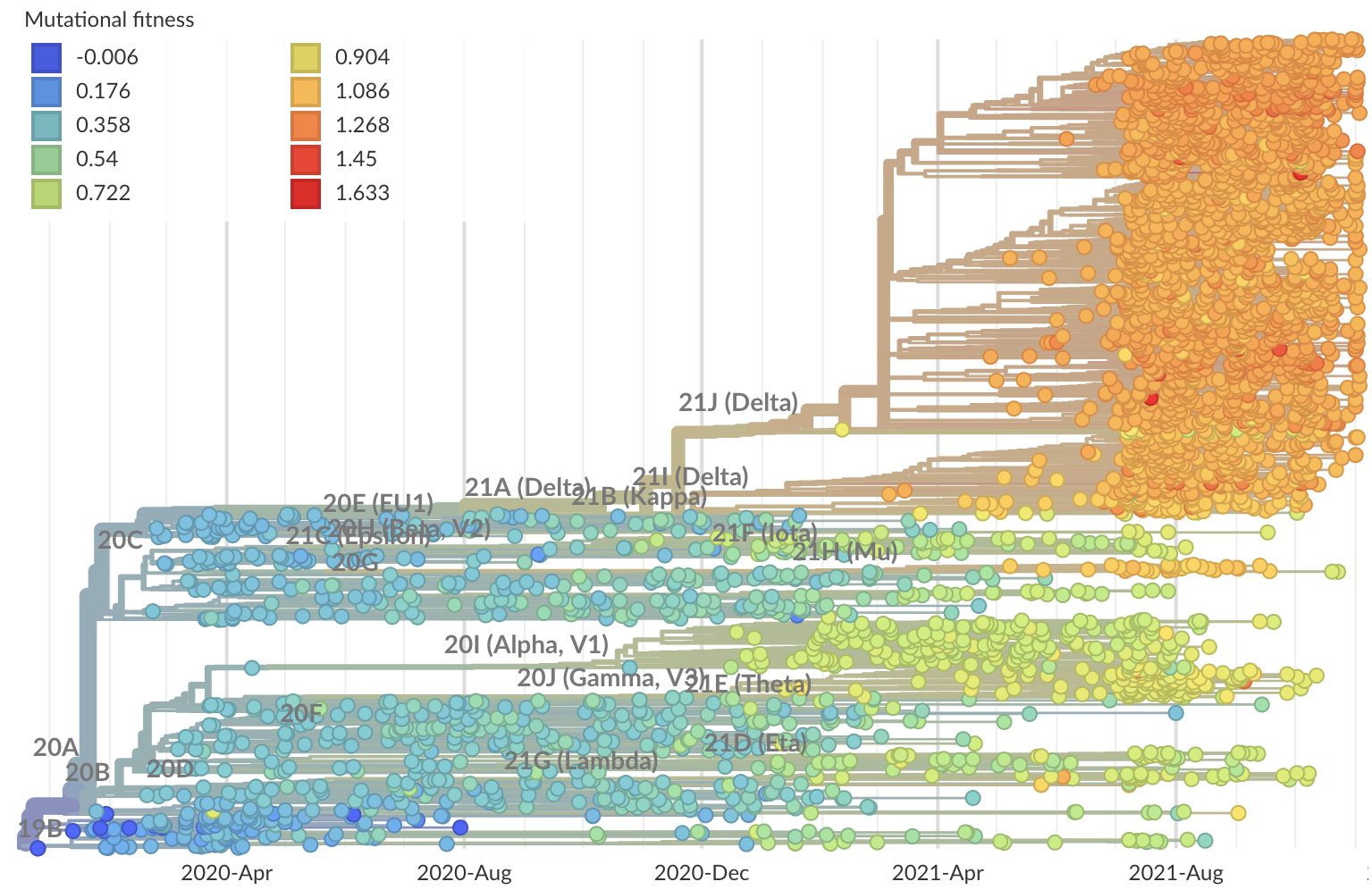
Rapid and parallel adaptive mutations in spike S1 drive clade success in SARS-CoV-2
 Katie Kistler,
Katie Kistler,
 John Huddleston
John Huddleston
Phylogeny of 10k genomes equitably sampled in space and time
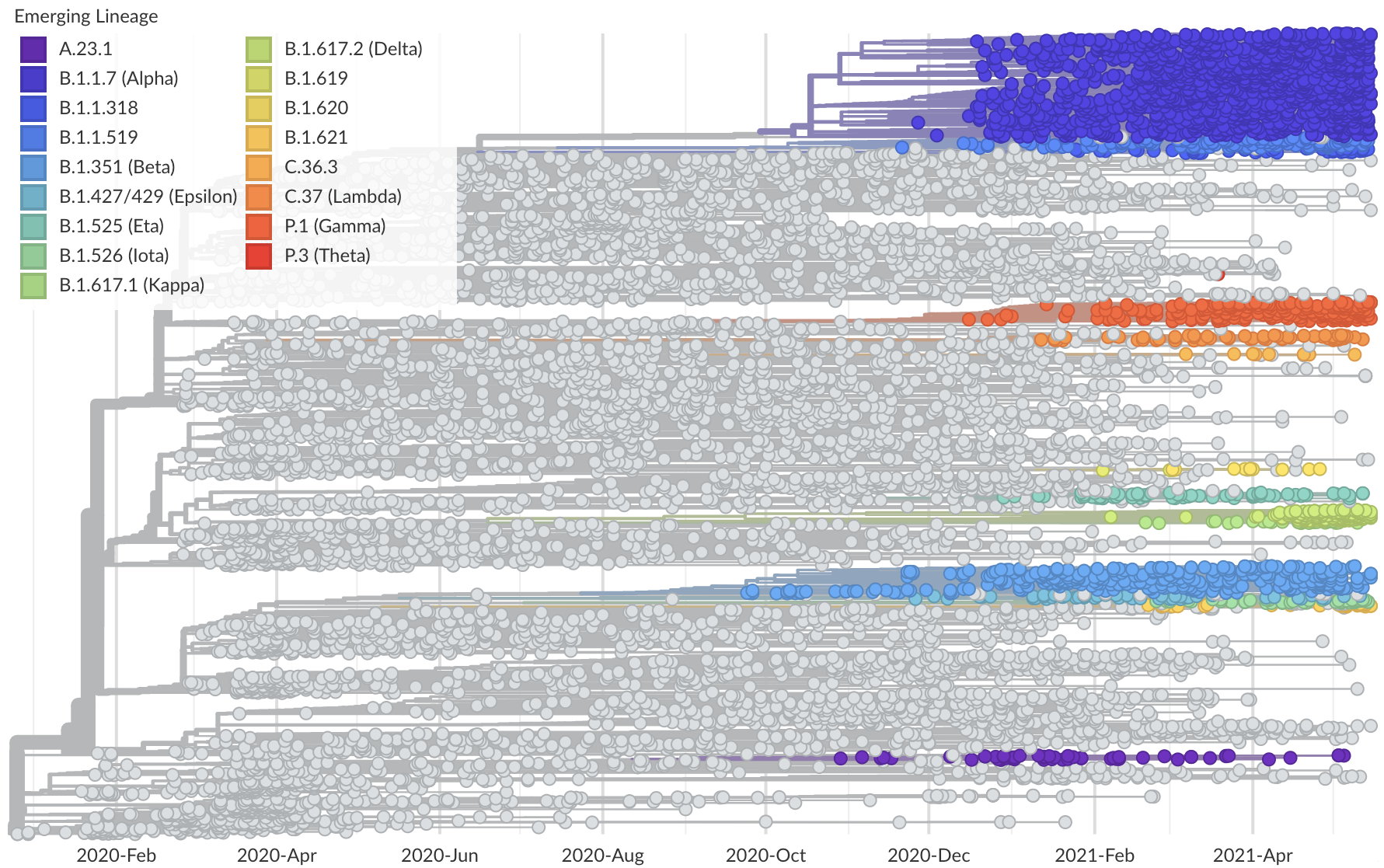
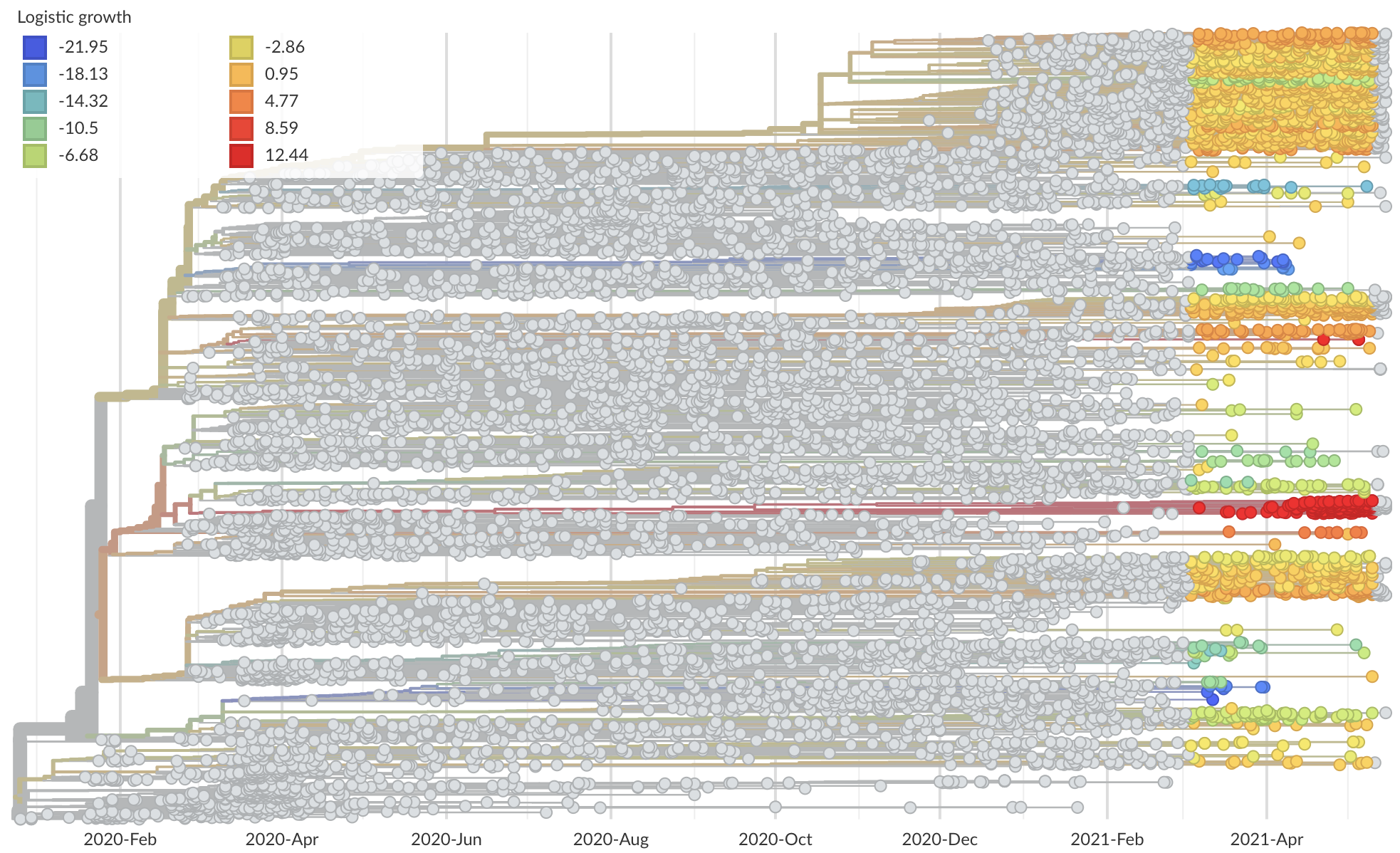
Measure clade growth as a proxy for viral fitness
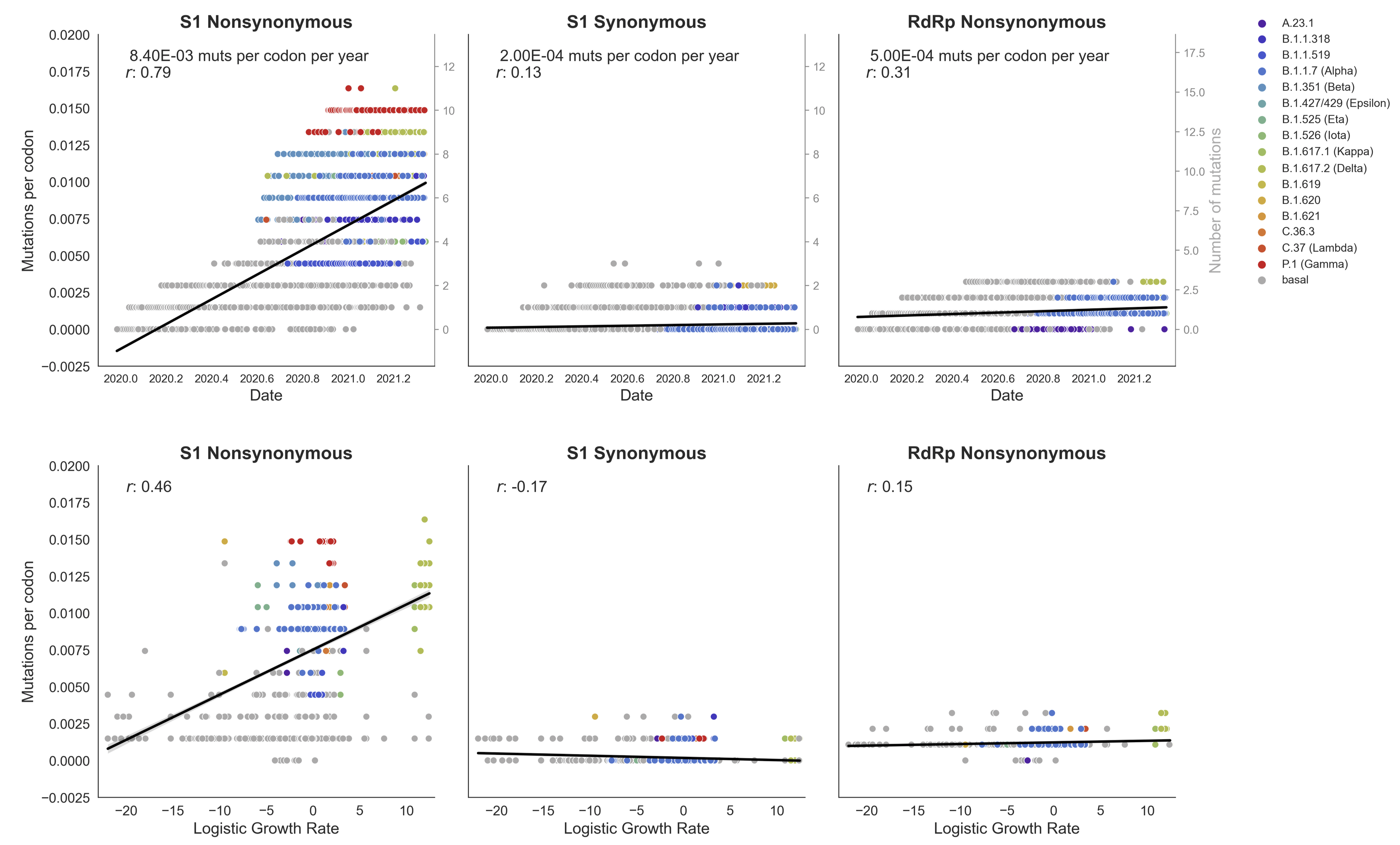
Clades with more S1 nonsynonymous mutations grow faster
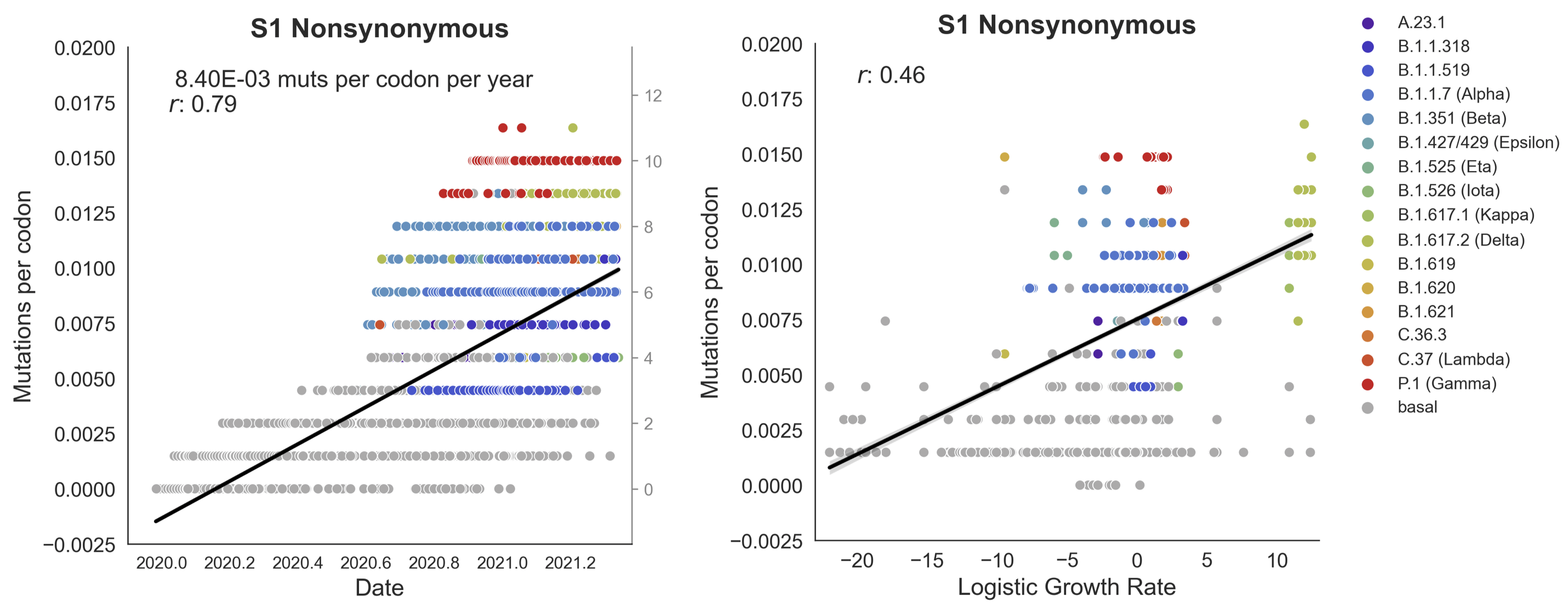
This correlation is absent in control regions
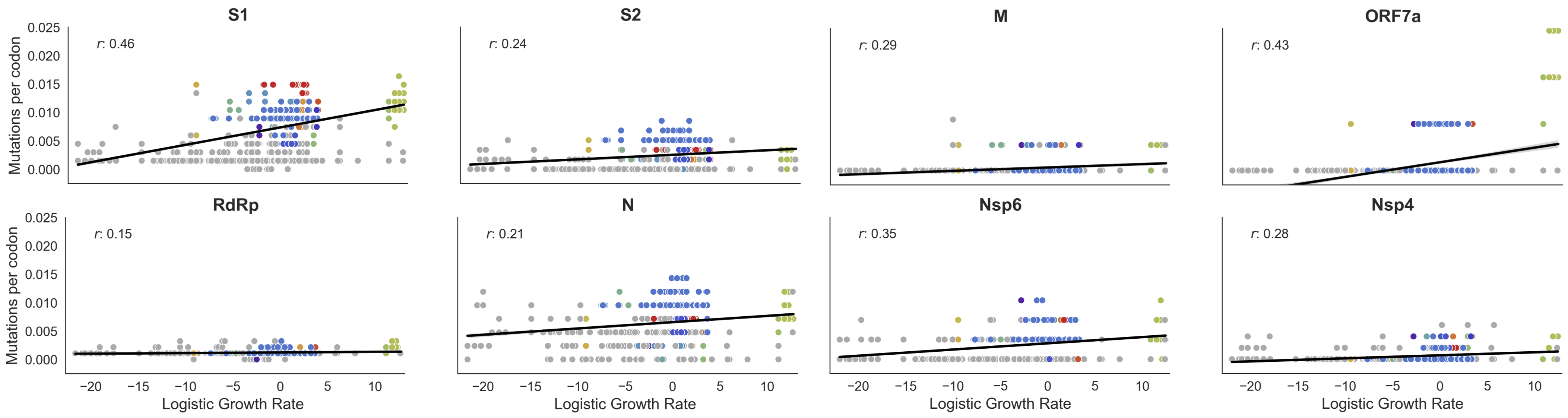
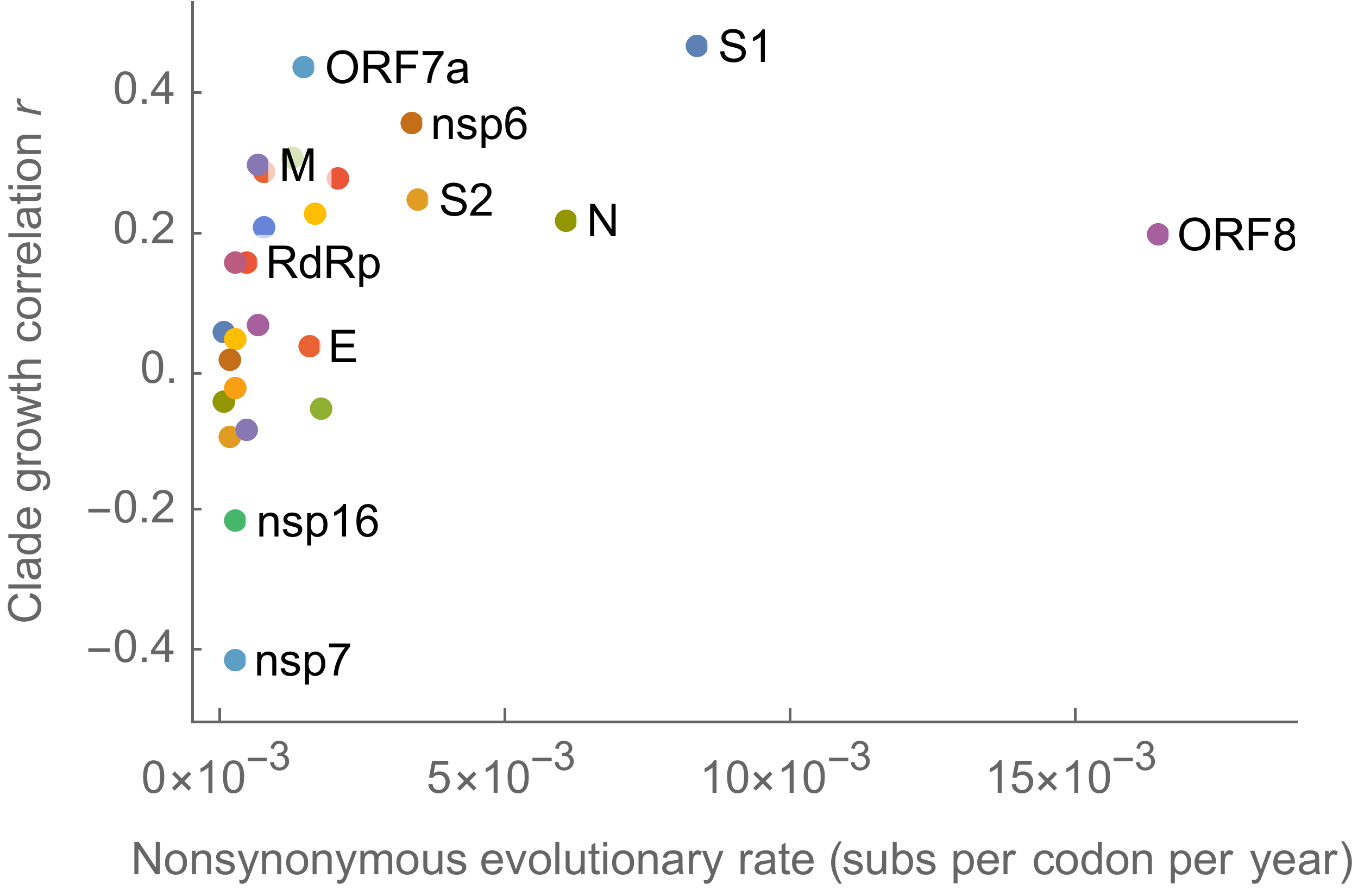
This correlation is stronger in S1 than other genes
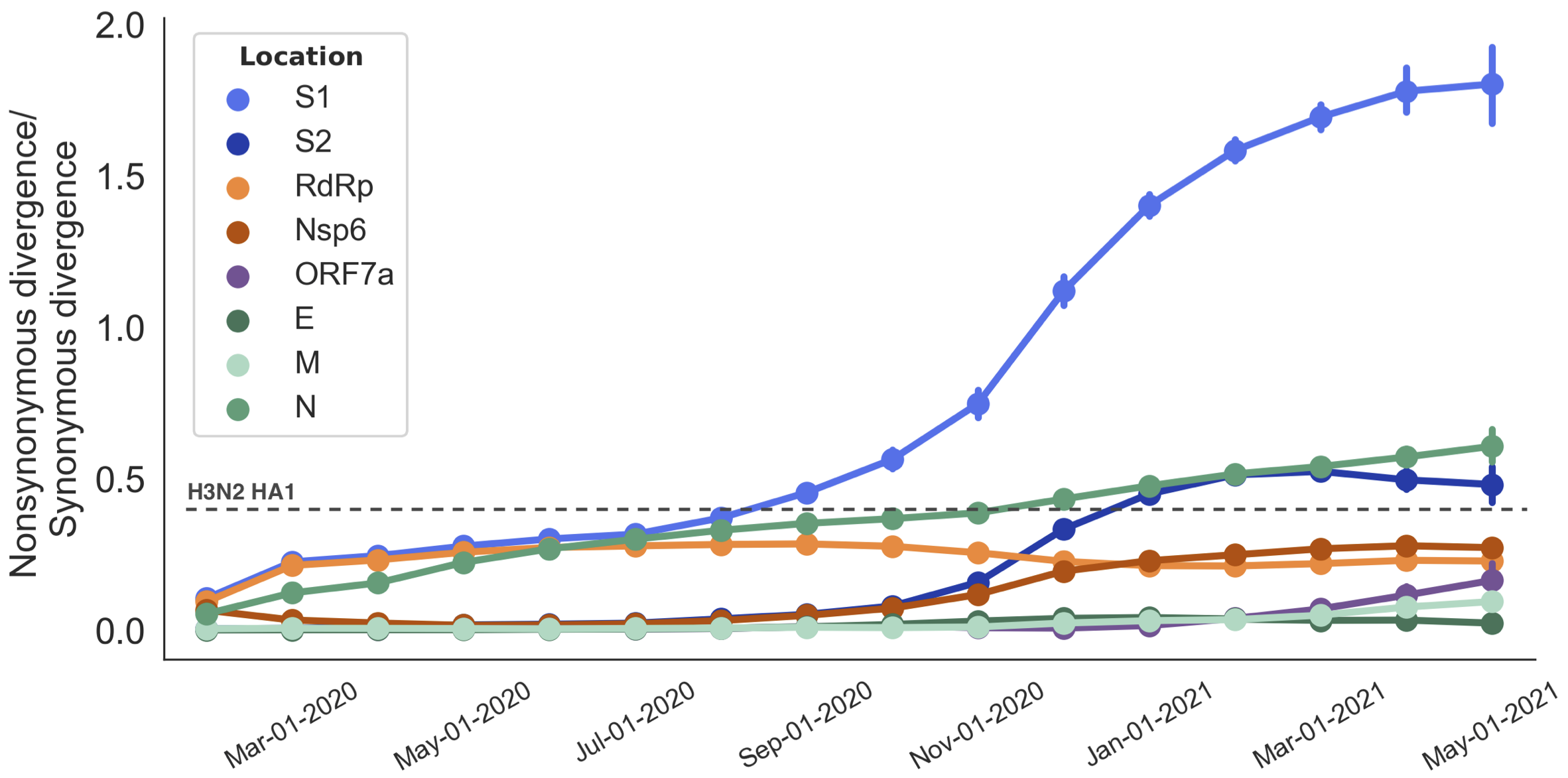
S1 is quickly evolving and highly correlated with clade growth
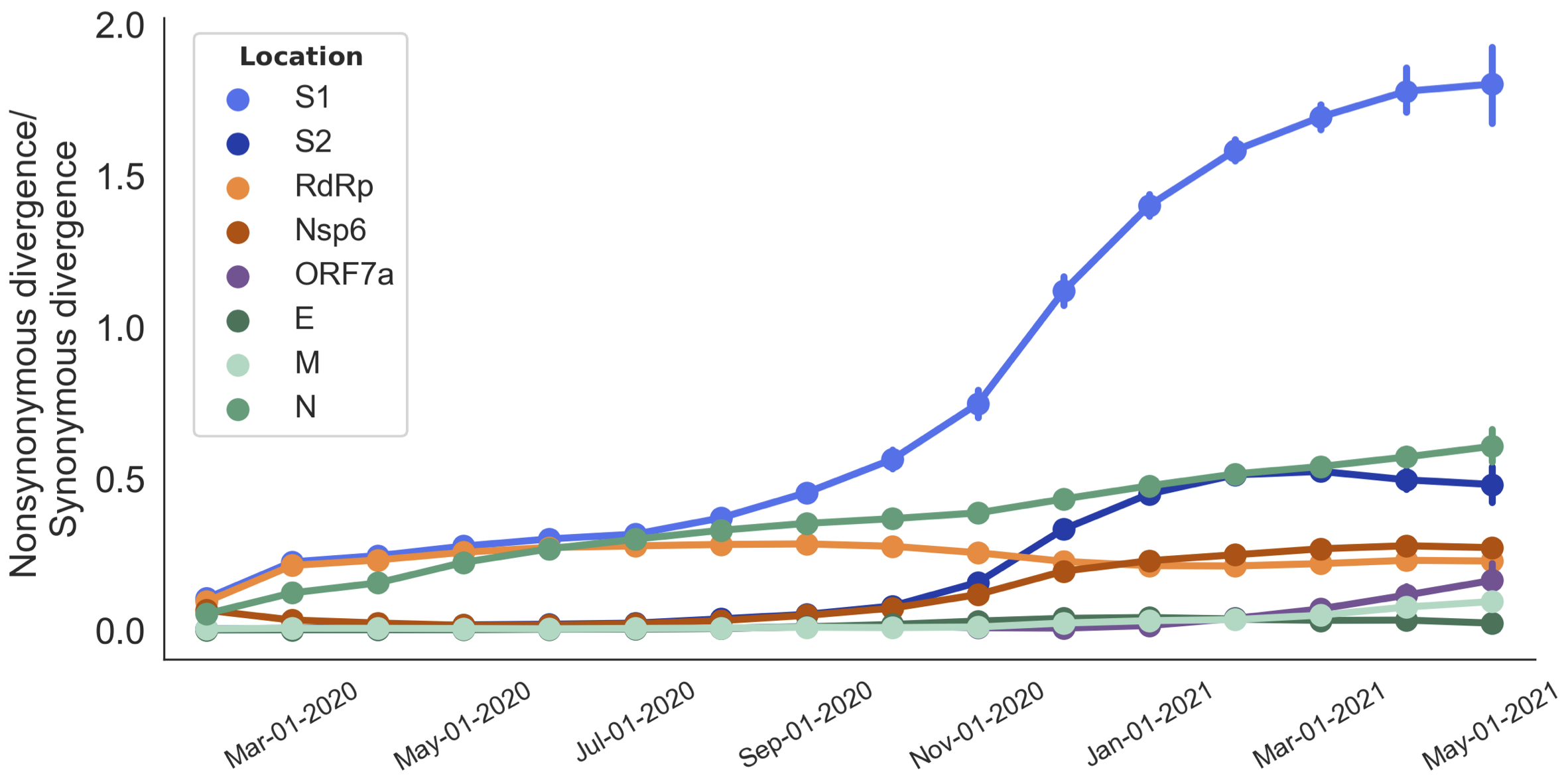
dN/dS through time further highlights adaptive evolution
Rapid pace of adaptive evolution relative to H3N2 influenza
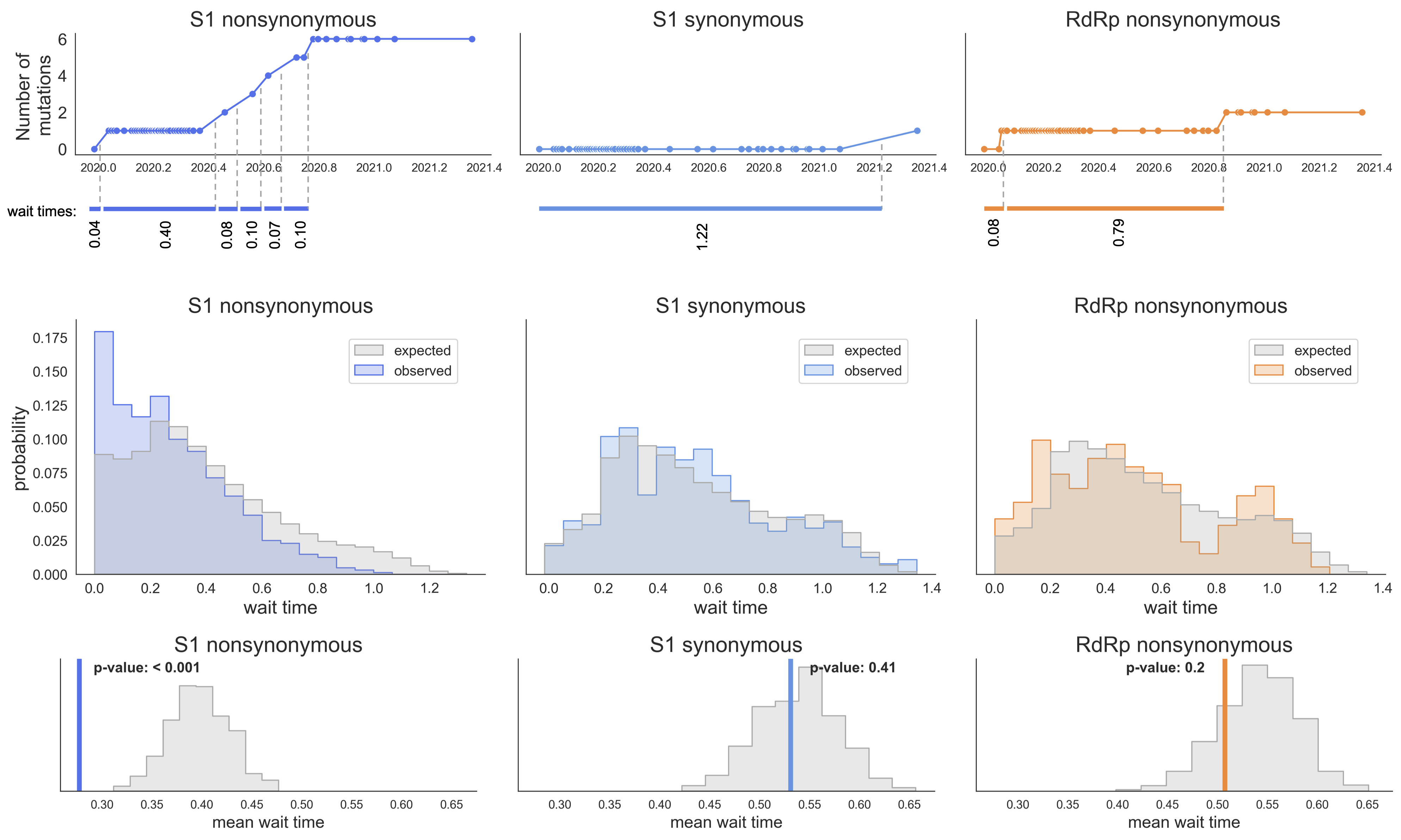
S1 mutations cluster phylogenetically, suggesting bursts of evolution
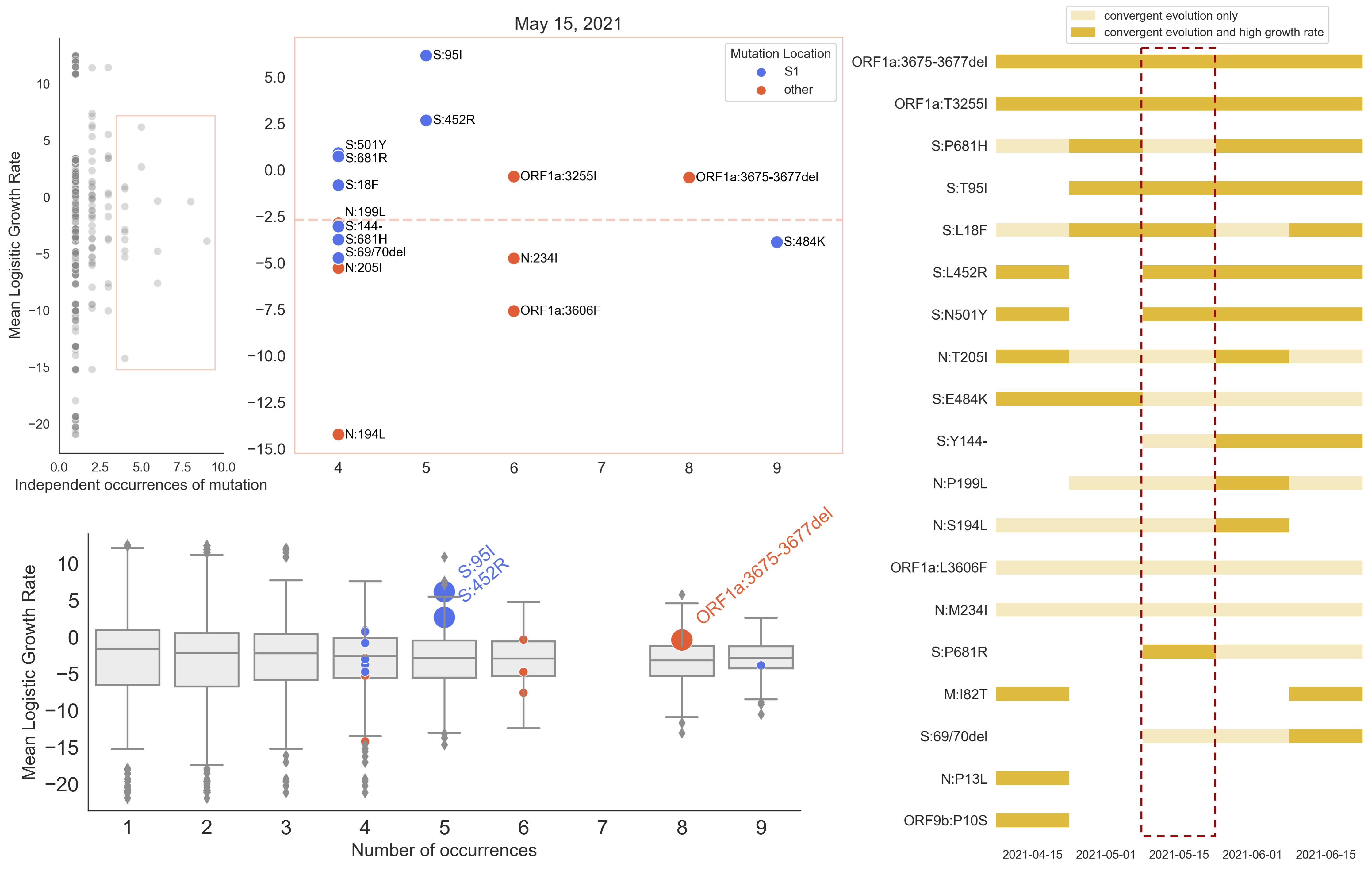
Many S1 mutations show strong convergent evolution
Mutations in S1 arising via within-host pressures result increase viral fitness and are enriched in the viral population by natural selection
Although selection has not been primarily for antigenic drift, observed level of adaptability suggests its potential
Variant transmission dynamics
Multiple approaches to modeling fitness differences between circulating variants
Multinomial logistic regression models work well on frequencies
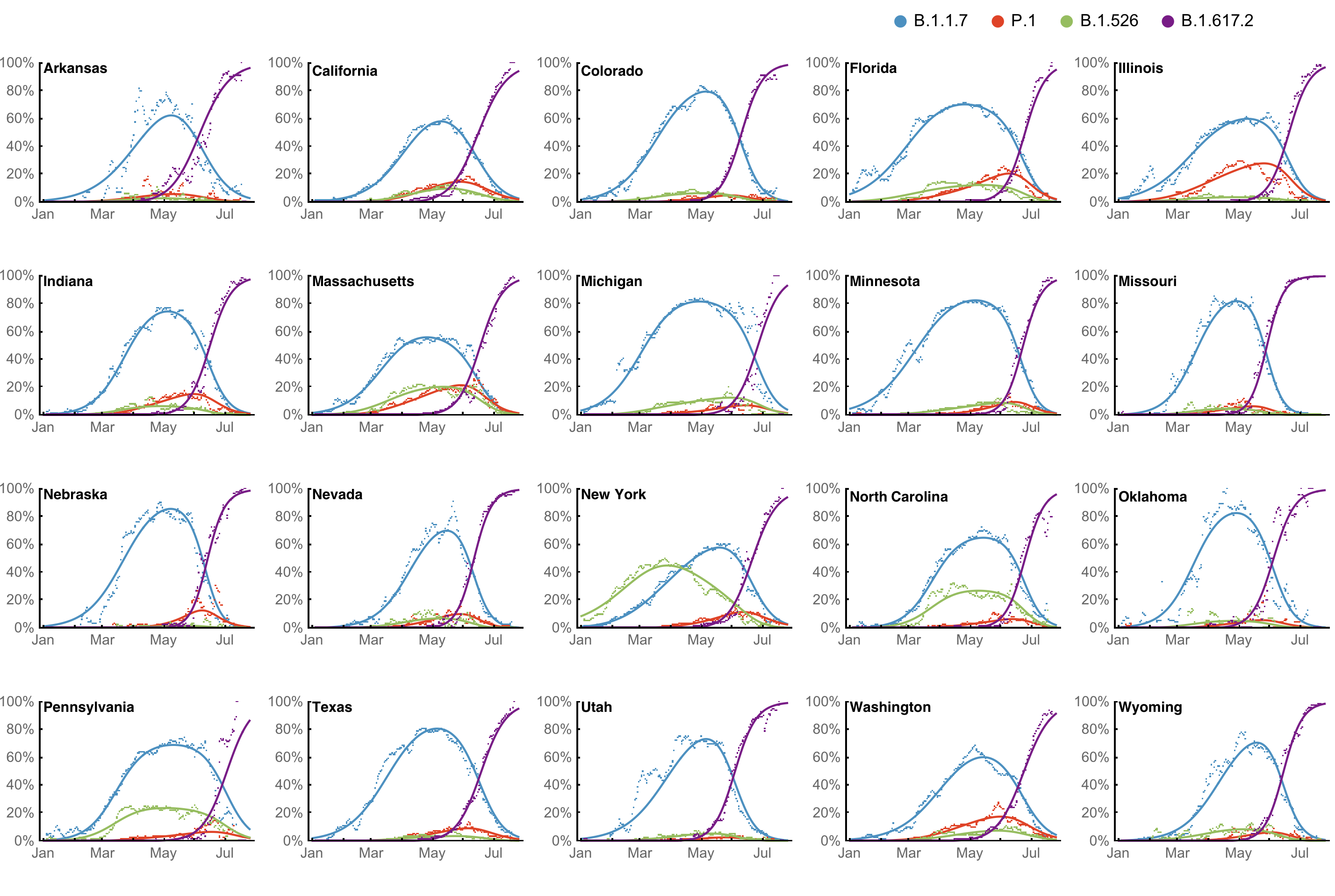
However, frequency of a variant may rise while cases fall
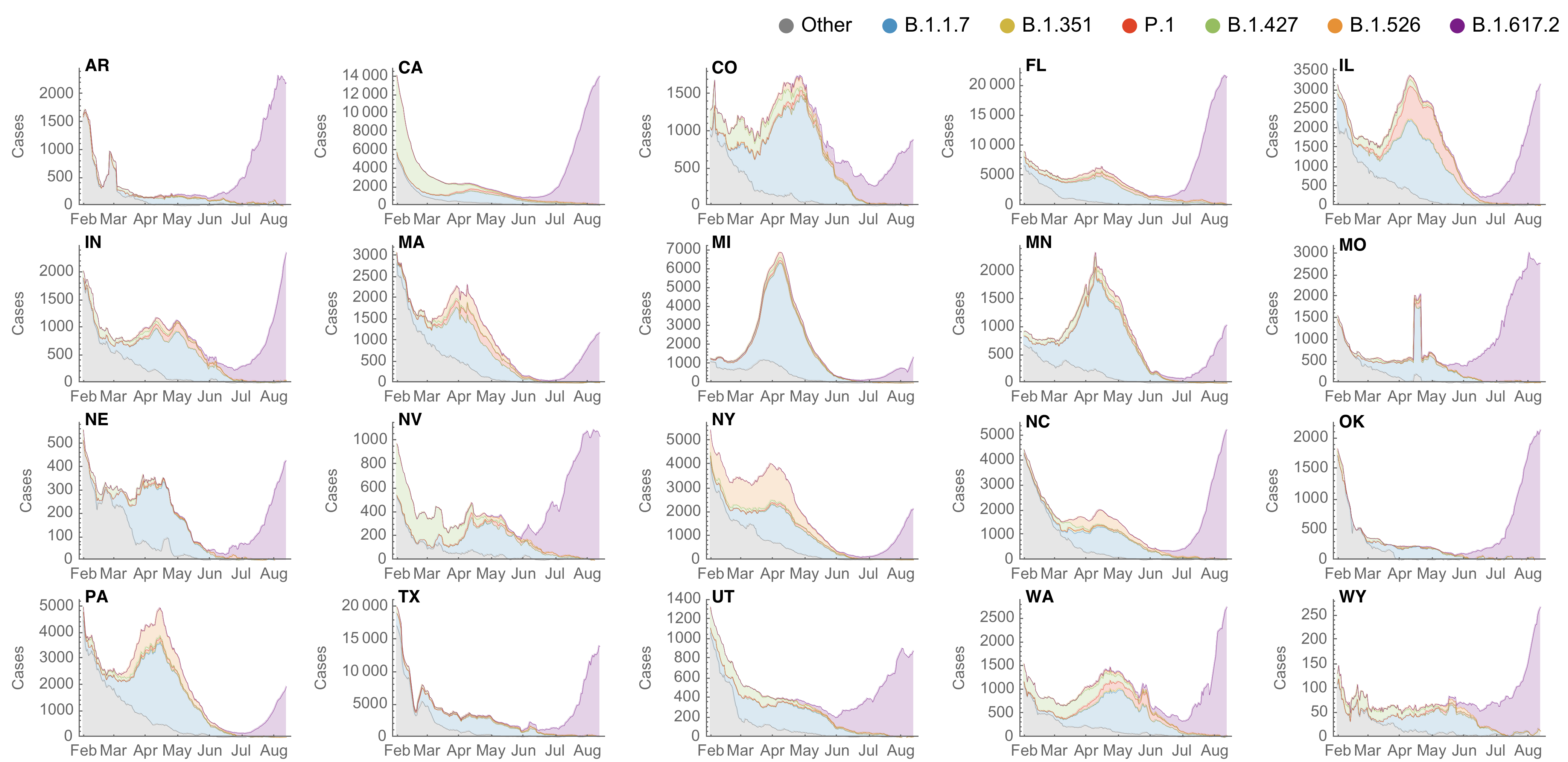
SARS-CoV-2 variant dynamics across US states show consistent differences in transmission rates
 Marlin Figgins
Marlin Figgins
Estimation of variant-specific Rt through time using state-level data
State-level case counts are partitioned based on frequencies of sequenced cases
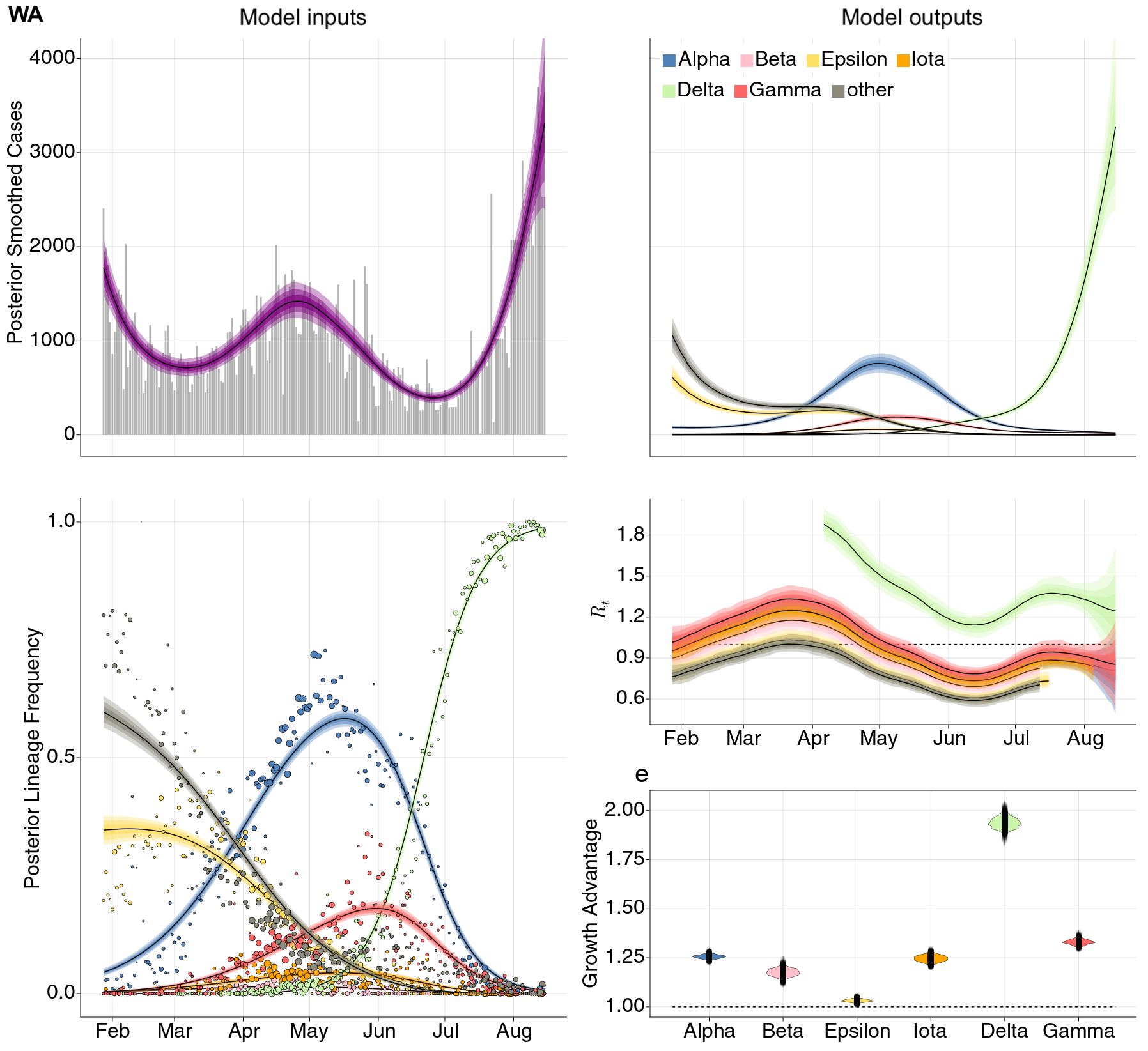
Differences in intrinsic Rt across variants, but all trending downwards
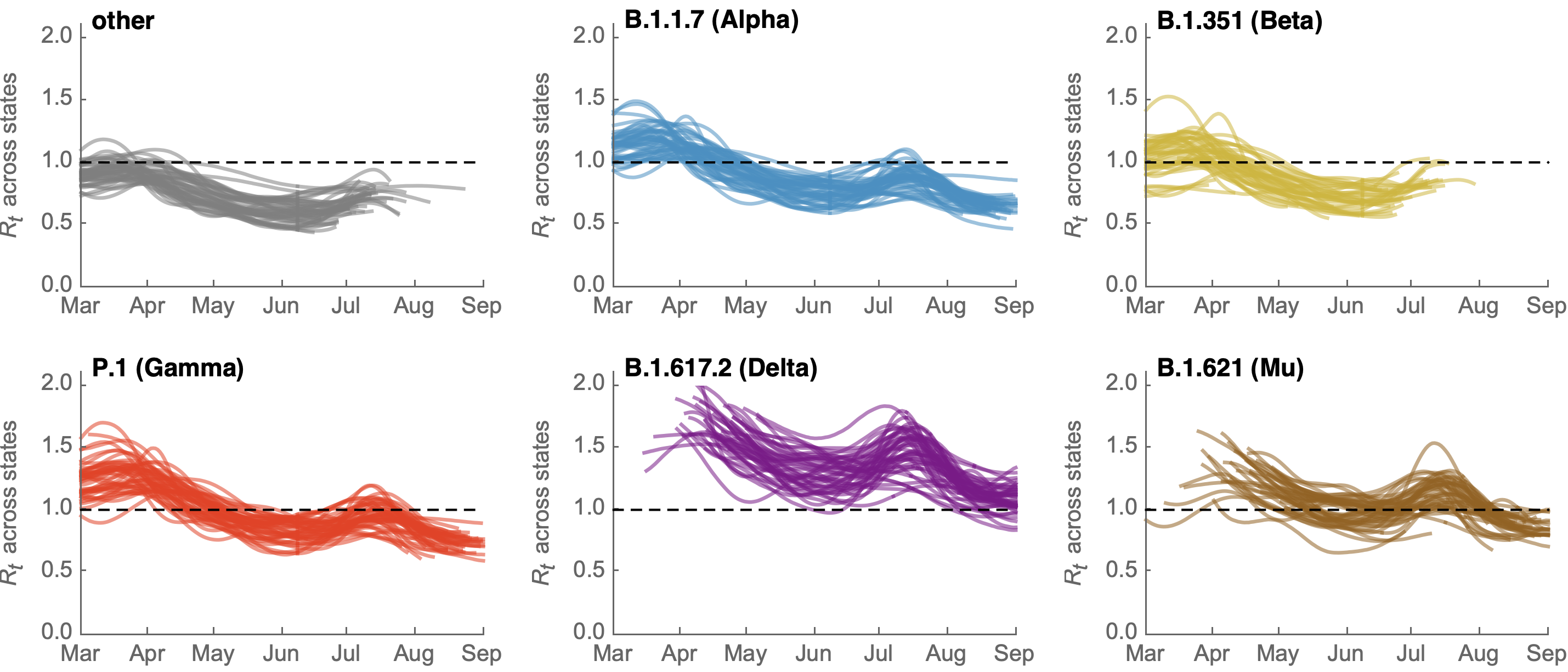
Consistent differences in variant-specific transmission rate across states
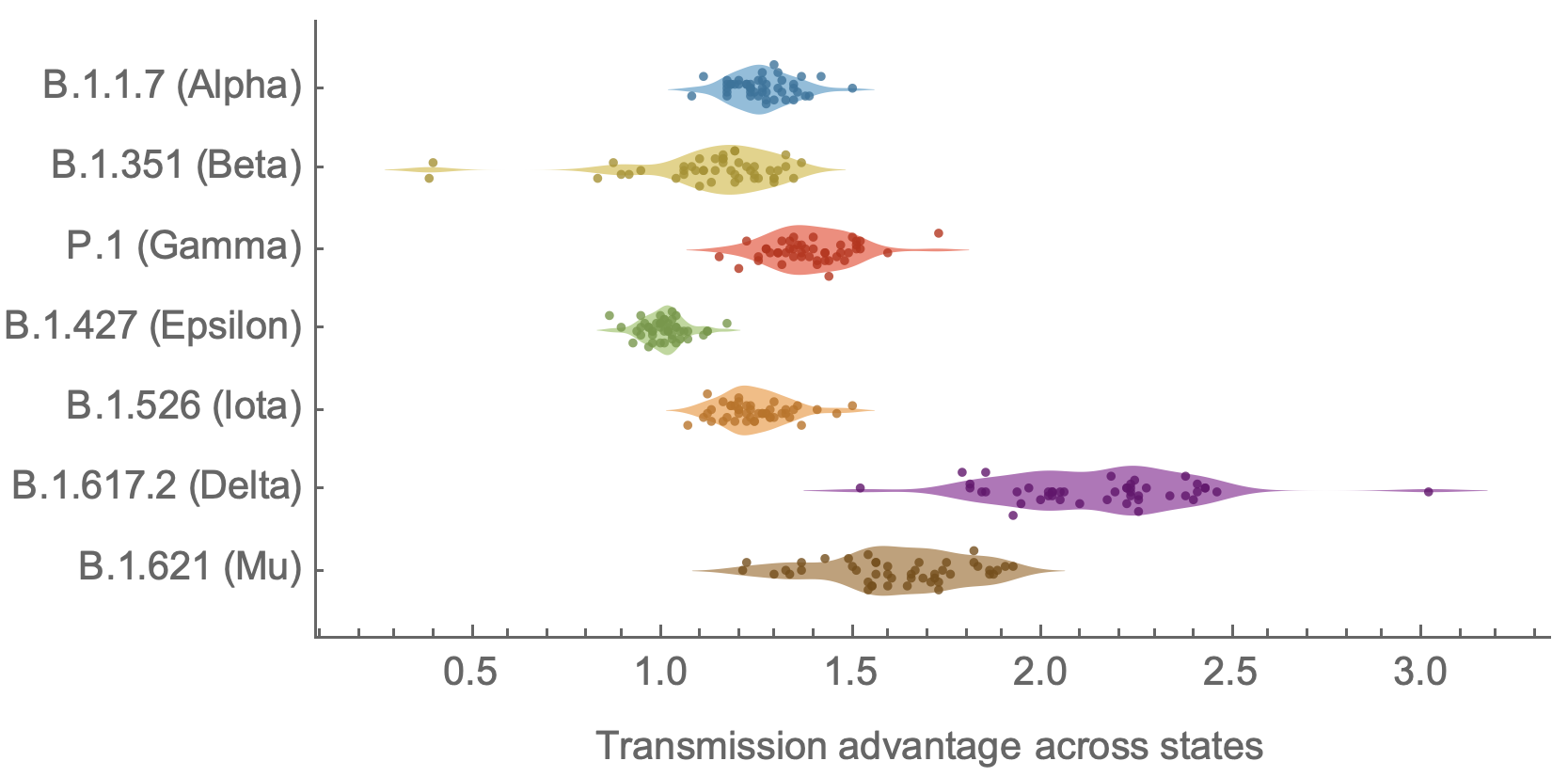
Consistent reductions in variant-specific Rt from vaccination
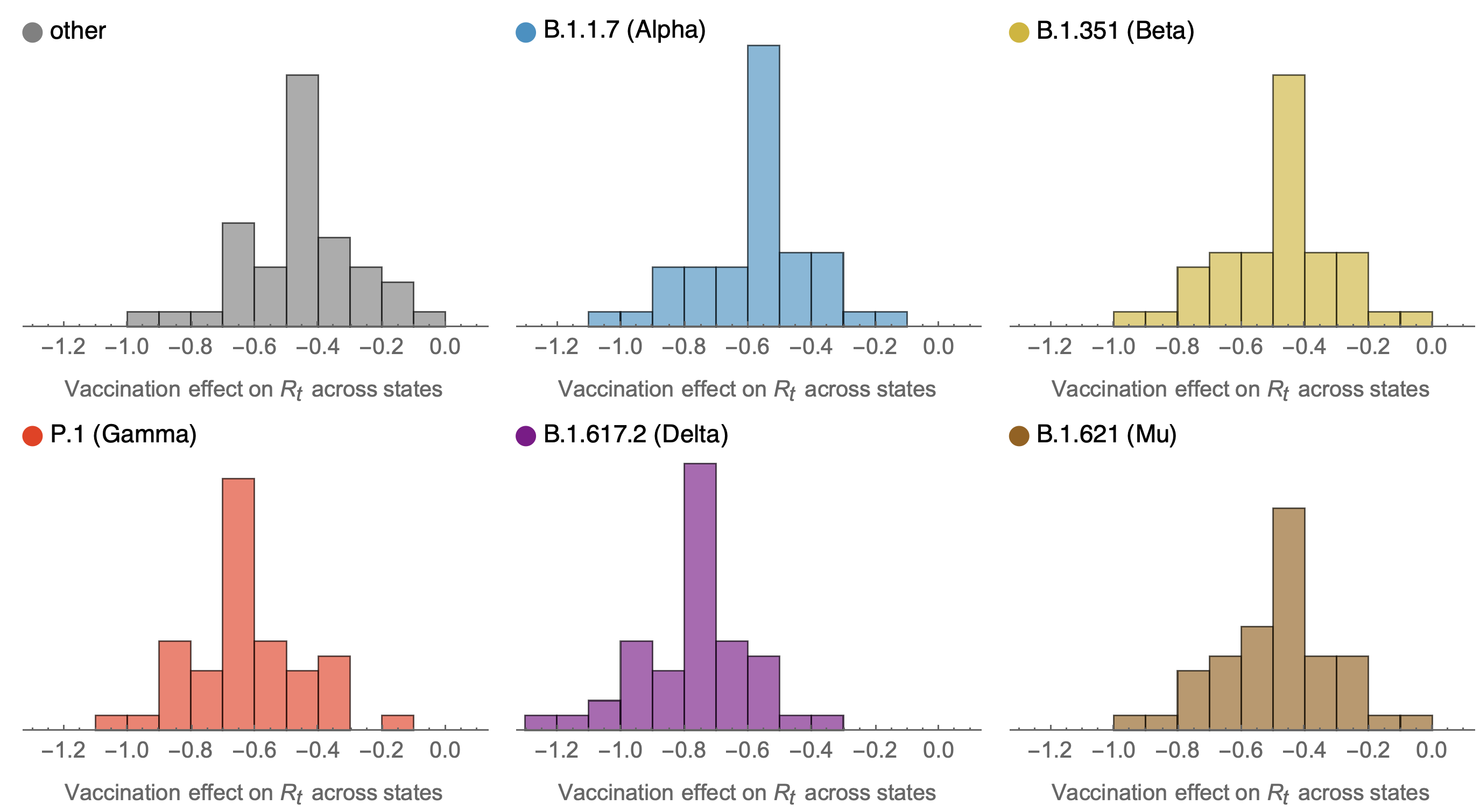
Future work would ideally tie together granular empirical estimates of viral fitness from frequency data together with mutational and phenotypic predictors to learn what is driving variant success
Model of additive effects of individual mutations shows little fitness variation within Delta
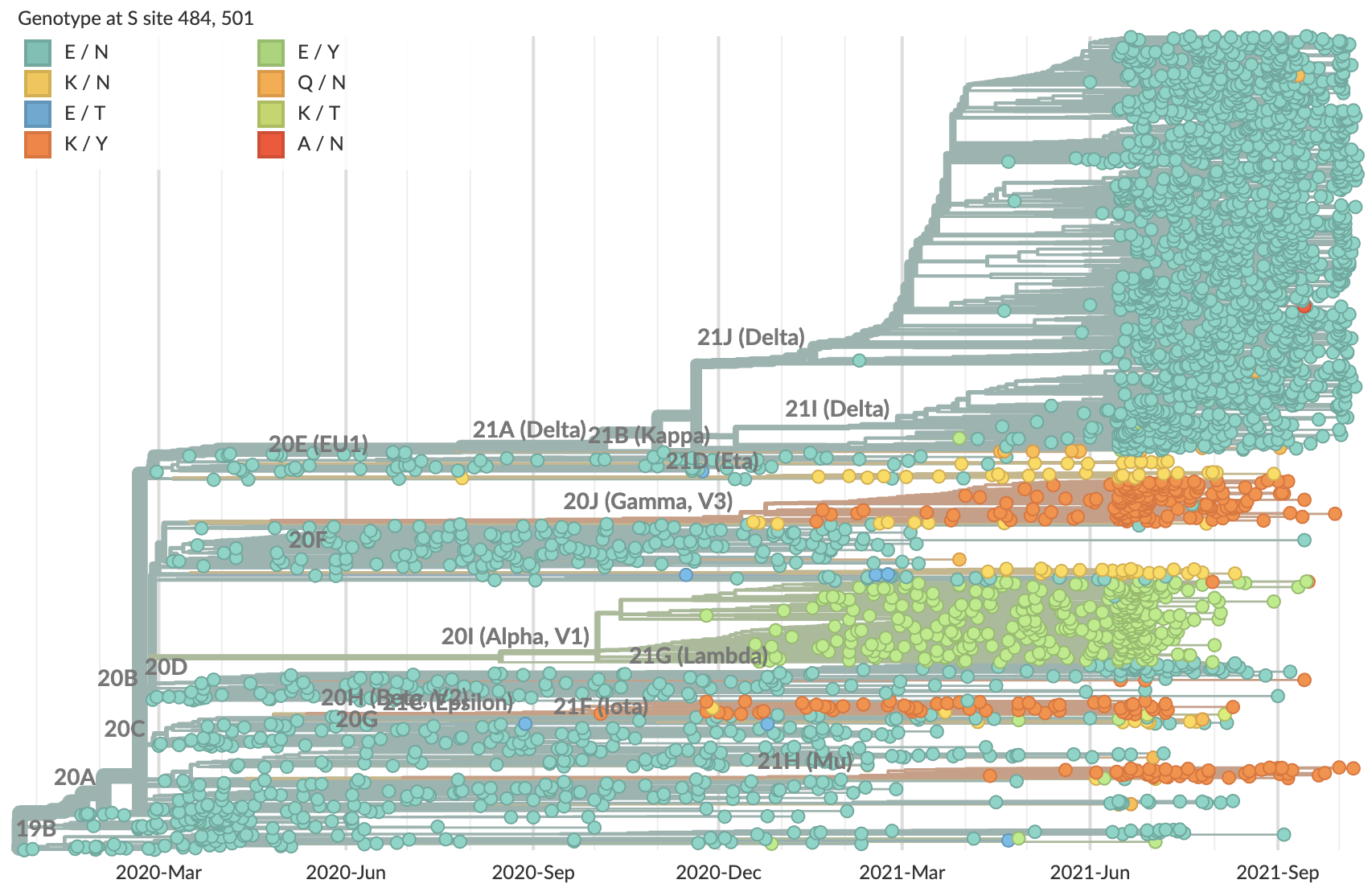
Potential for epistasis
Mutations at spike 484 and 501 perhaps less fit on Delta background
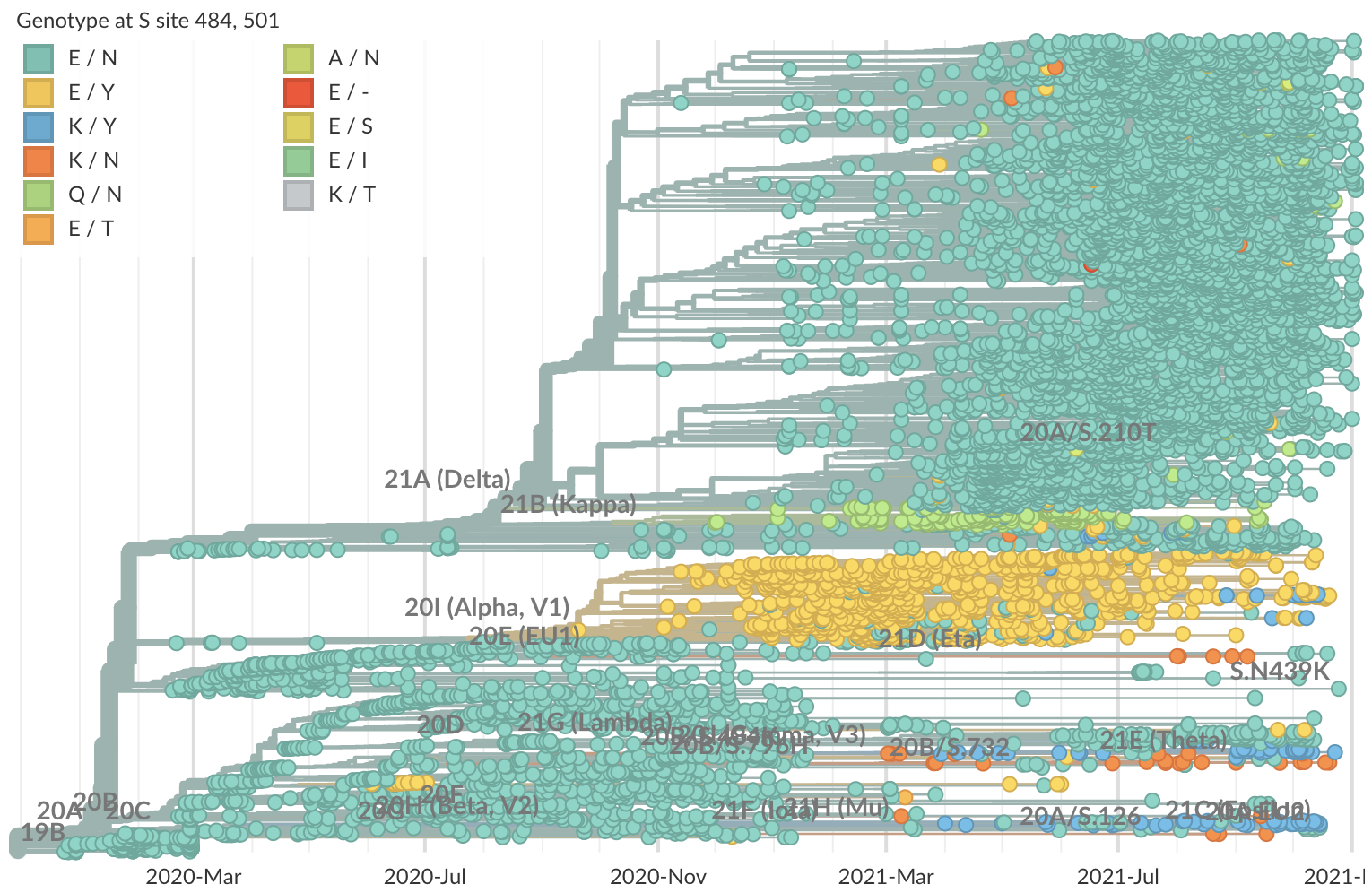
Mutations at spike 484 and 501 perhaps less fit on Delta background (zoomed to Delta)
Future perspectives
Broad expectations based on comparison with flu
- R0 of Delta is perhaps ~5 compared to R0 of flu of ~2. At same rates of evolution and waning expect more COVID circulation.
- Rates of adaptive sequence evolution in SARS-CoV-2 have been about ~5X that of H3N2 flu. Expect this to slow down, but suggests that virus will be generally quite adaptable.
- IFR of COVID is roughly comparable to flu once you have prior immunity granting durable memory responses.
1, 2 and 3 suggest a virus that circulates like flu does at perhaps higher levels (given R0), but individual infections aren't much more severe in terms of mortality than H3N2 influenza. Expect perhaps 50-100K deaths in the US each year.
SARS-CoV-2 scientific (and prosaic) priorities
- Forecasting evolution and ensuring vaccine updates match circulating viruses
- Universal vaccine to engender broader immune response and/or to target more conserved epitopes
- Plentiful testing, contact tracing and a public health apparatus
- Treatments, ventilation, etc...
Acknowledgements
SARS-CoV-2 genomic epi: Data producers from all over the world, GISAID and the Nextstrain team
Bedford Lab:
![]() John Huddleston,
John Huddleston,
![]() James Hadfield,
James Hadfield,
![]() Katie Kistler,
Katie Kistler,
![]() Louise Moncla,
Louise Moncla,
![]() Maya Lewinsohn,
Maya Lewinsohn,
![]() Thomas Sibley,
Thomas Sibley,
![]() Jover Lee,
Jover Lee,
![]() Cassia Wagner,
Cassia Wagner,
![]() Miguel Paredes,
Miguel Paredes,
![]() Nicola Müller,
Nicola Müller,
![]() Marlin Figgins,
Marlin Figgins,
![]() Eli Harkins,
Eli Harkins,
![]() Denisse Sequeira
Denisse Sequeira






