Evolutionary dynamics of SARS-CoV-2
Trevor Bedford (@trvrb)
Associate Professor, Fred Hutchinson Cancer Research Center
12 May 2021
COVID-19 Scenario Planning Workshop
Bill & Melinda Gates Foundation
Slides at: bedford.io/talks
1. Real-time tracking of SARS-CoV-2 evolution
2. Emergence of variants of concern
3. Current circulation patterns
4. Expectations for antigenic evolution
Real-time tracking of SARS-CoV-2 evolution
Spike protein is critical for cell invasion by the virus and is the primary target for human immune response
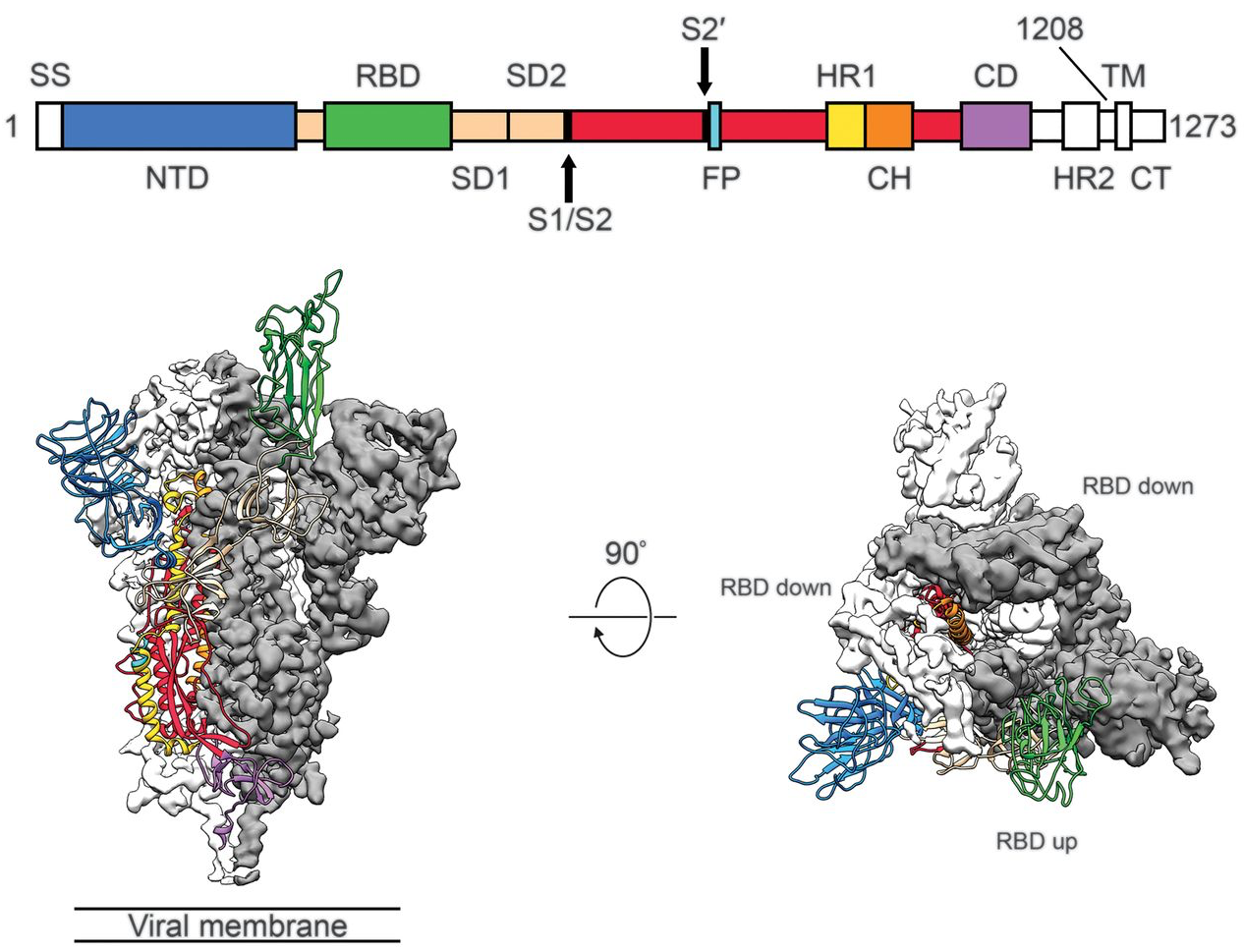
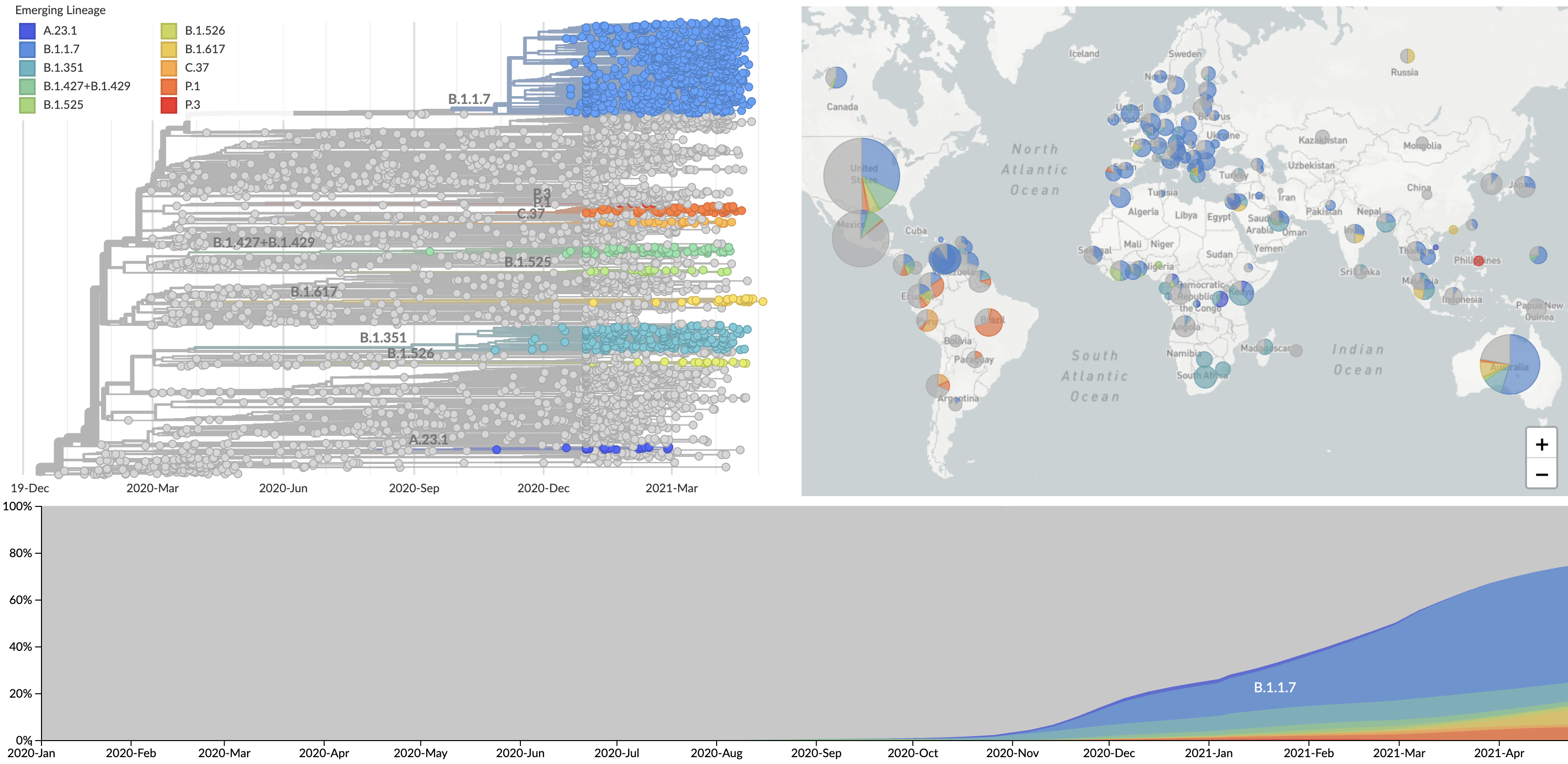
Over 1.4M SARS-CoV-2 genomes shared to GISAID and evolution tracked in real-time at nextstrain.org
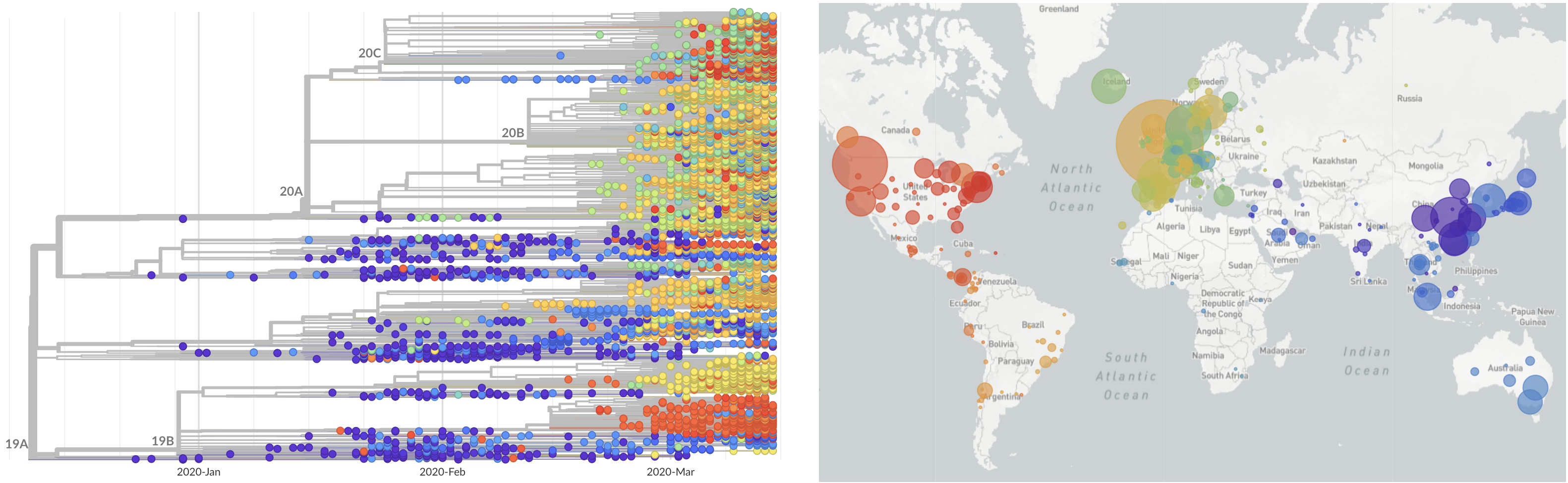
SARS-CoV-2 lineages establish globally in February and March
Limited early mutations like D614G spread globally during initial wave
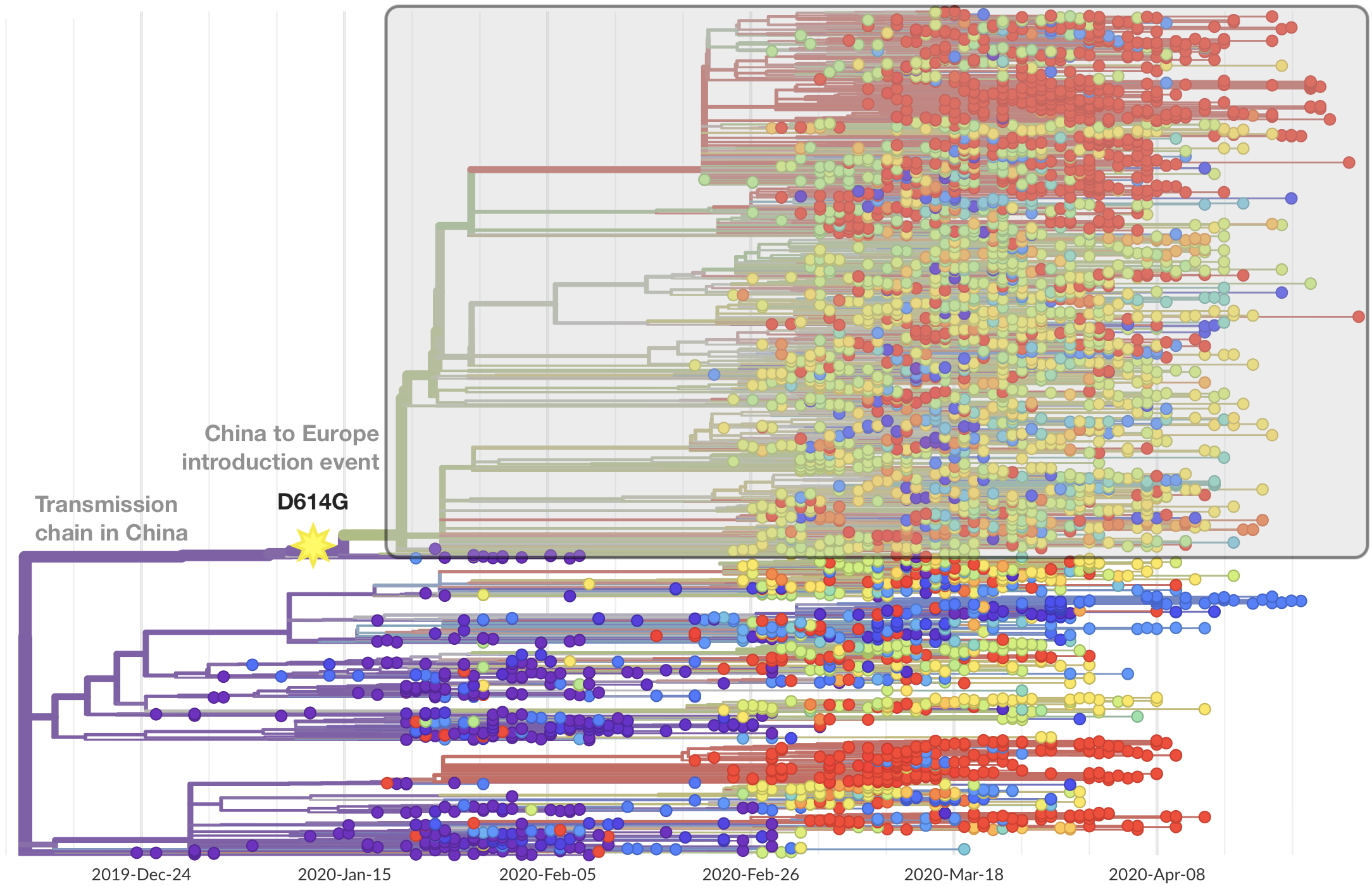
After initial wave, with mitigation
efforts and decreased travel,
regional clades emerge
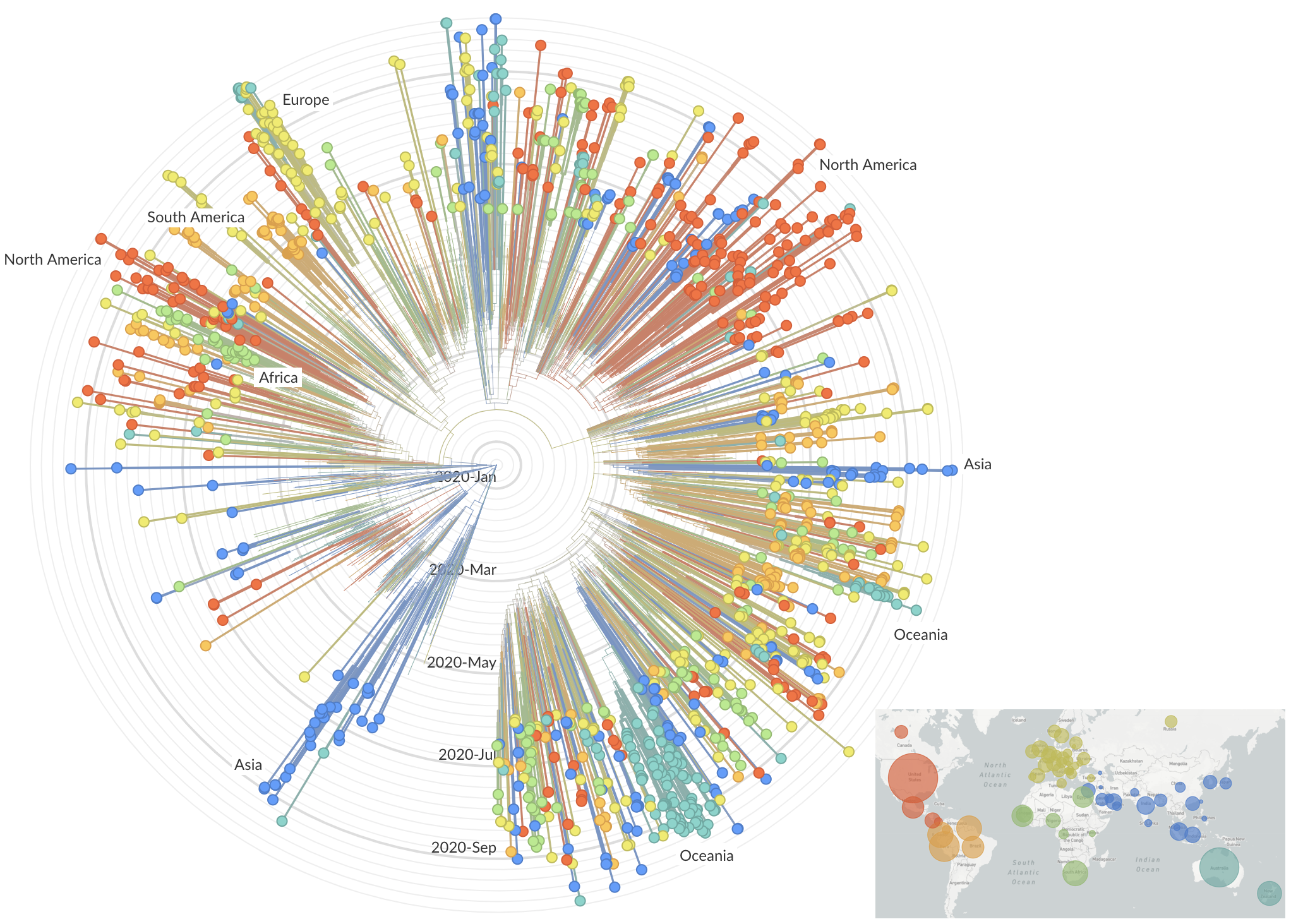
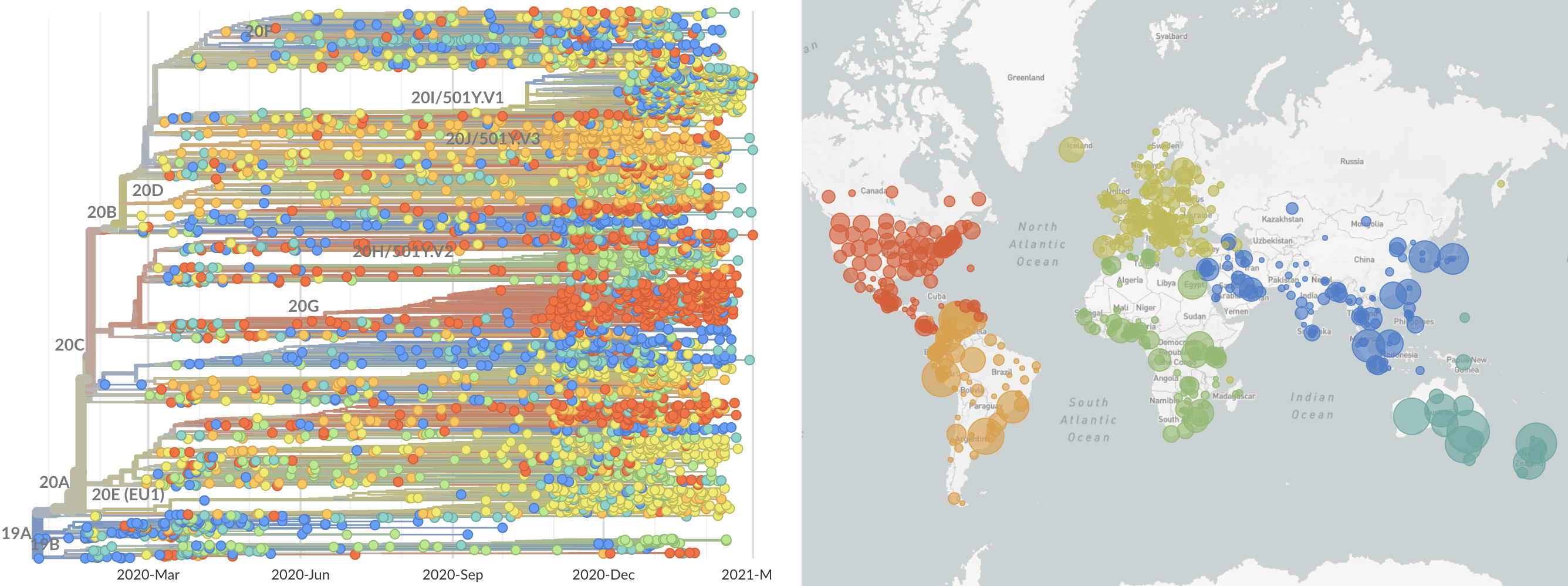
Mutations in summer and fall 2020 were confined to regional dominance
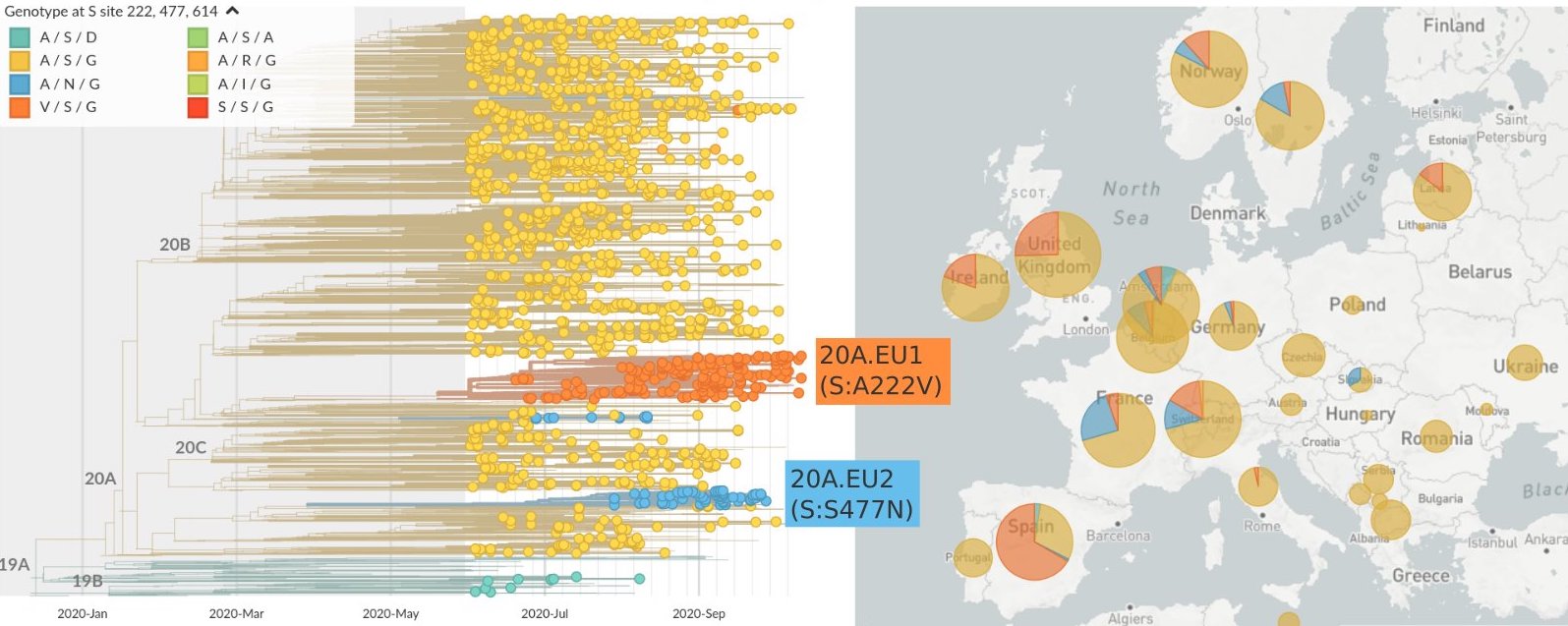
Emergence of variants of concern
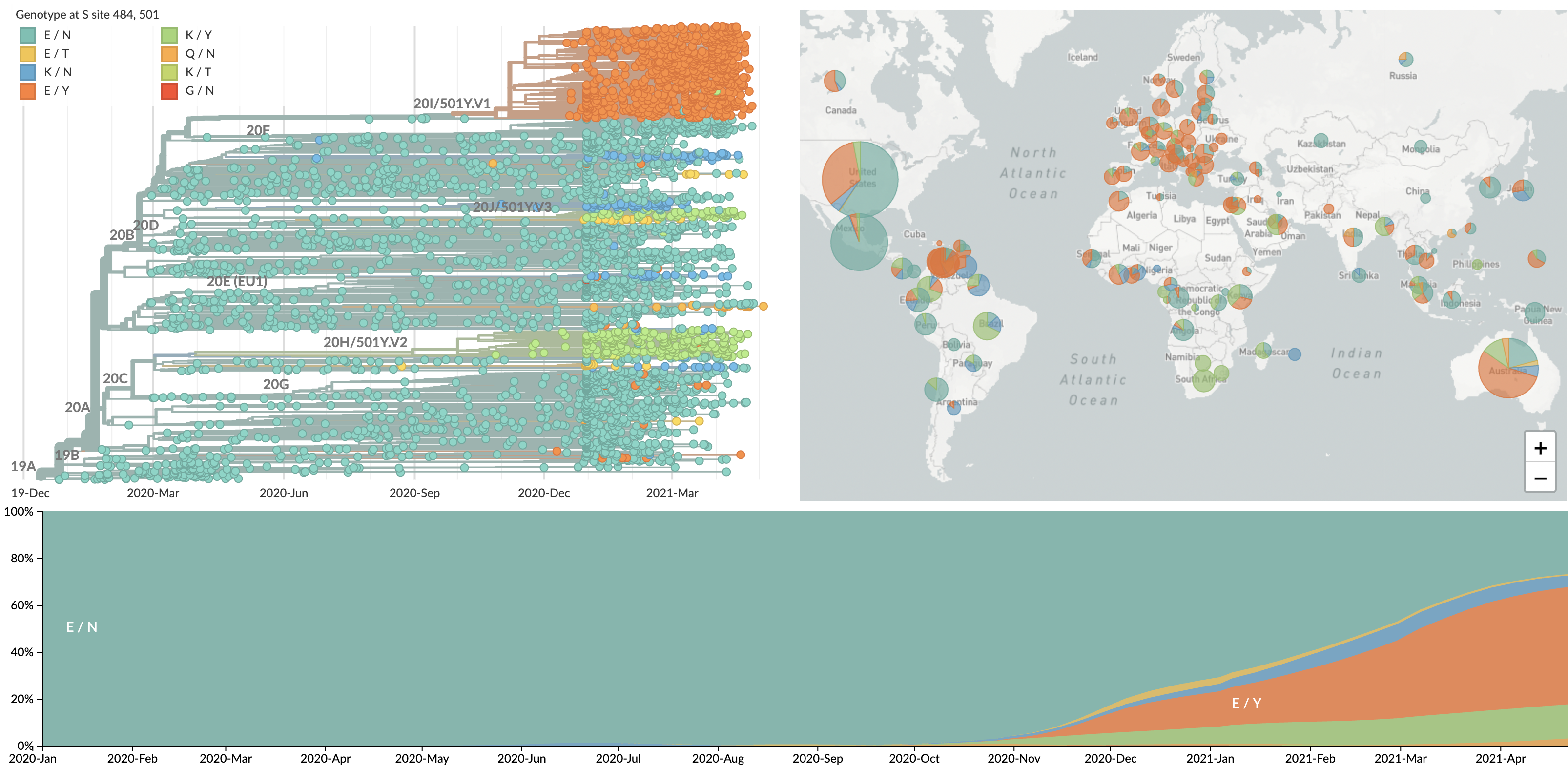
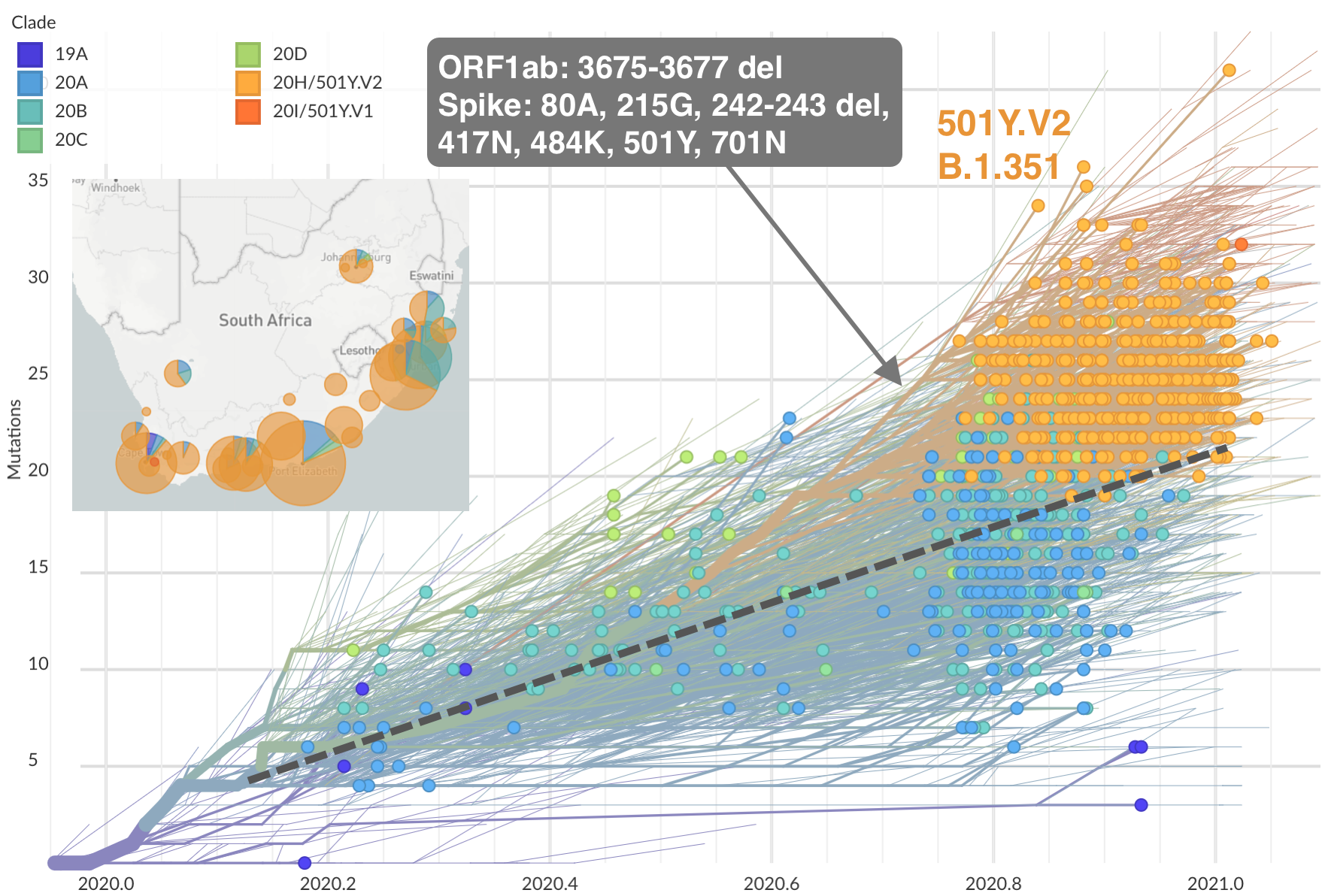
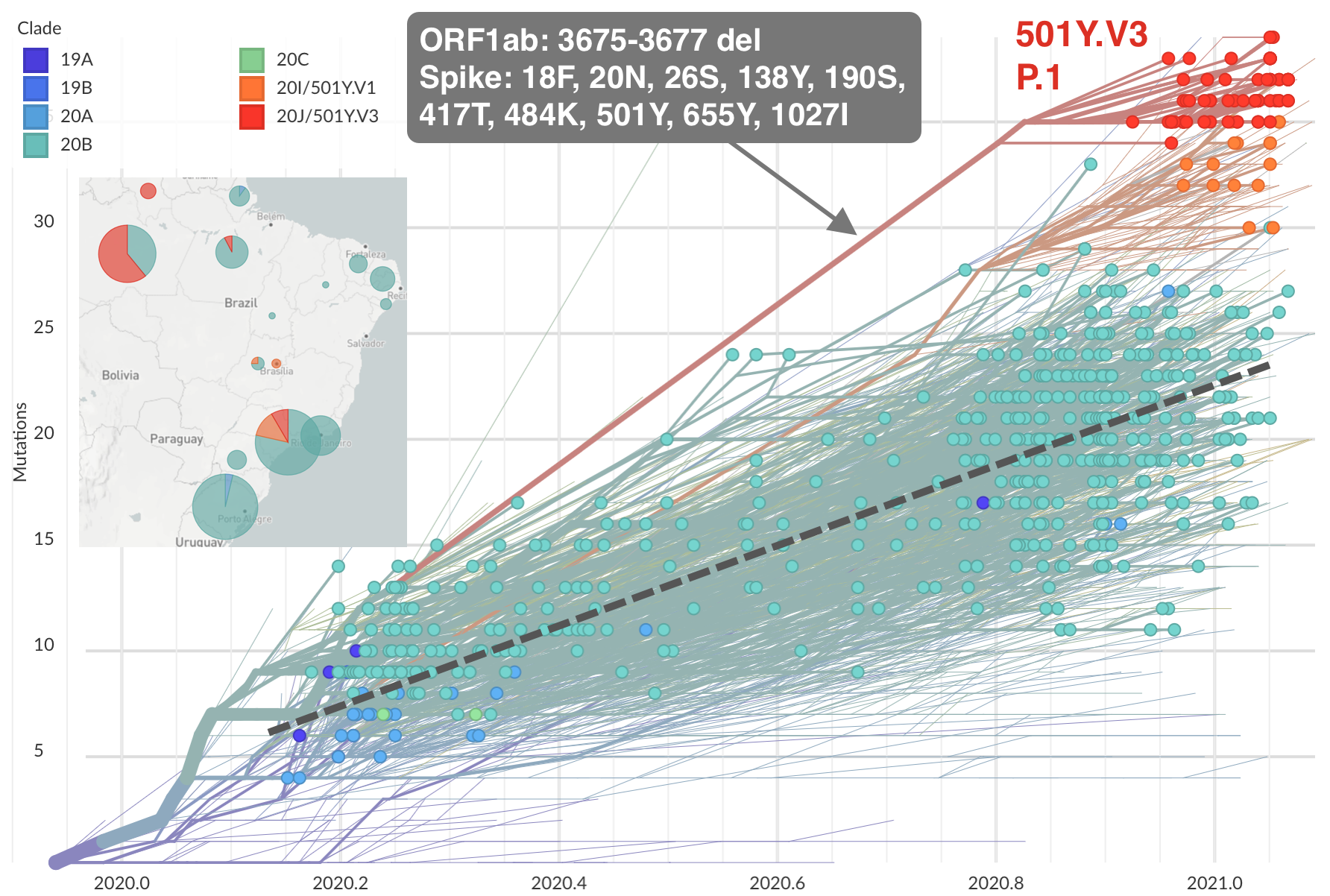
484K and 501Y repeatedly emerging across the world
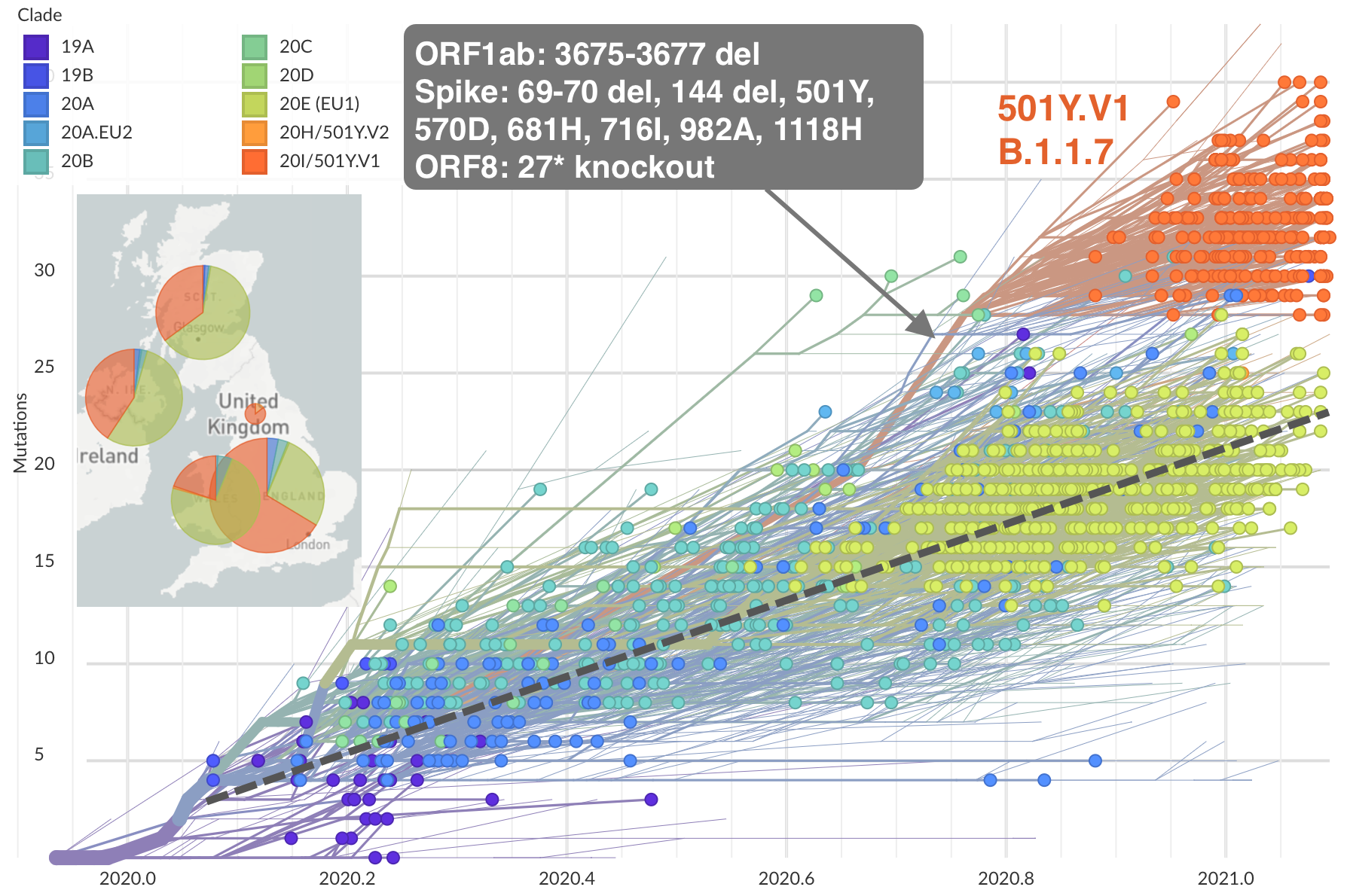
Emergence of 501Y.V1 (B.1.1.7) in the UK
Emergence of 501Y.V2 (B.1.351) in the South Africa
Emergence of 501Y.V3 (P.1) in the Brazil
Working hypothesis of within-host evolution occurring during prolonged infection, driven by natural selection for immune escape.
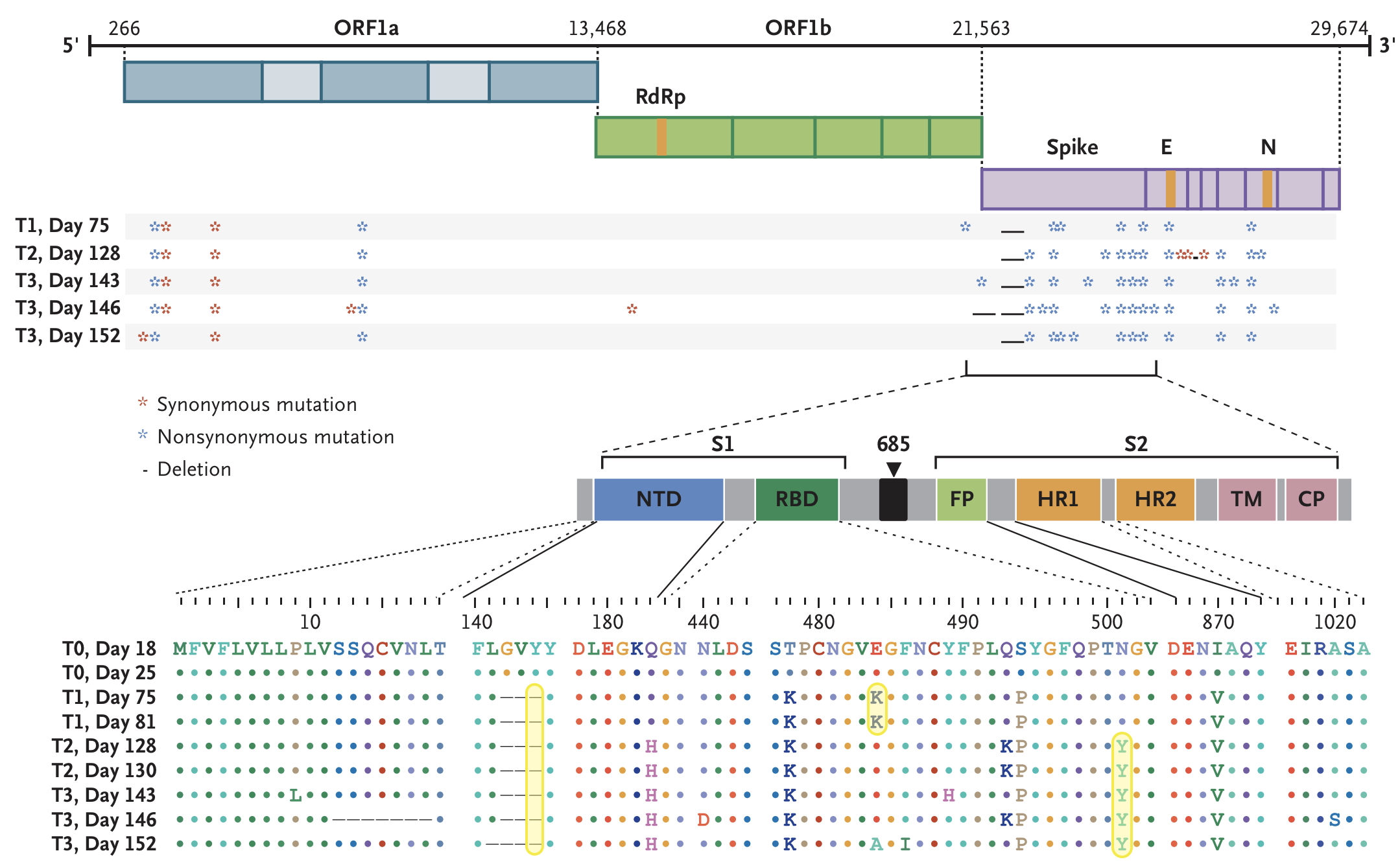
Rapid within-host evolution during persistent infection
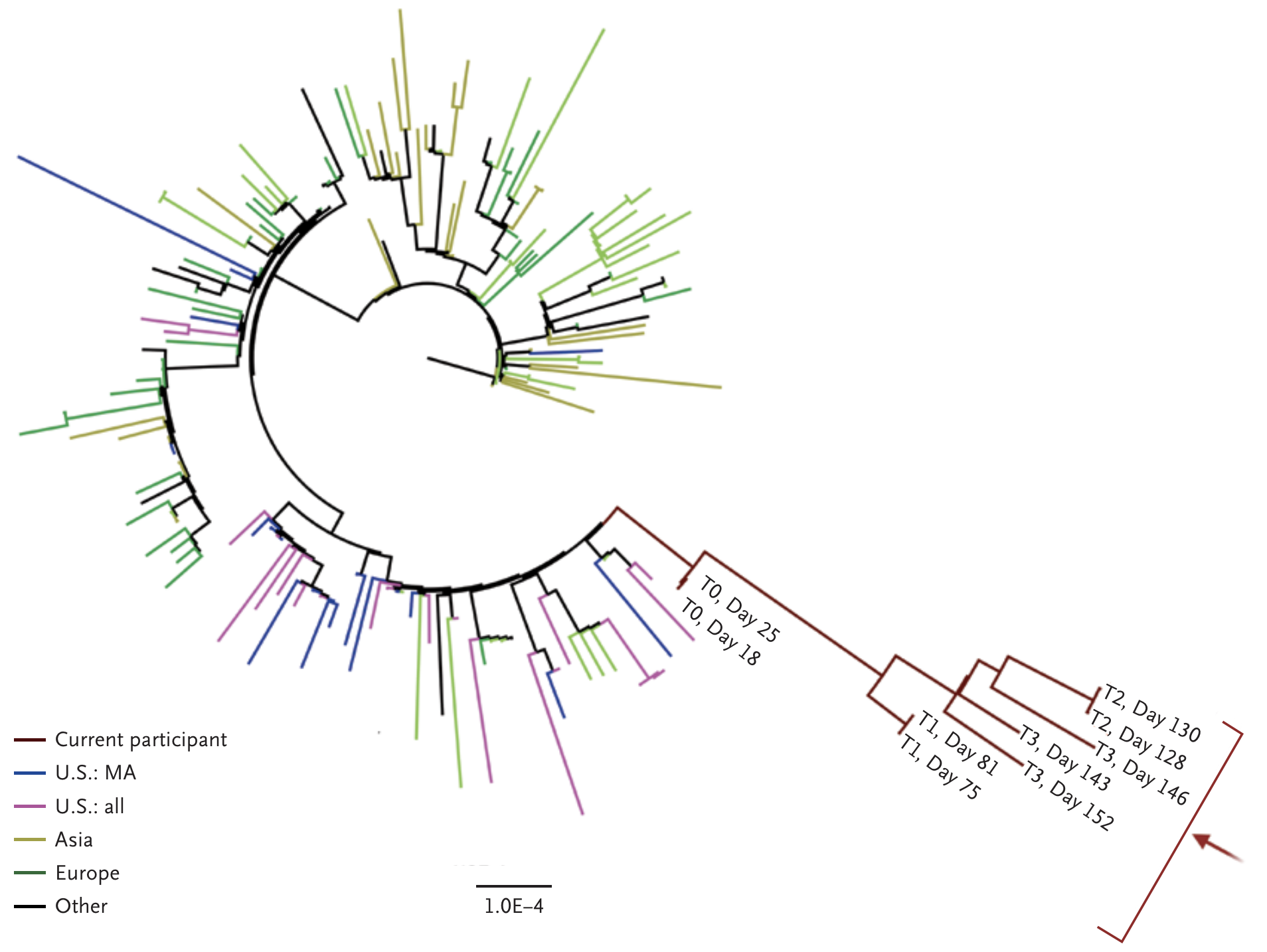
484K and 501Y observed during this evolution
Current circulation patterns
VUI / VOC lineages are rapidly expanding across the world
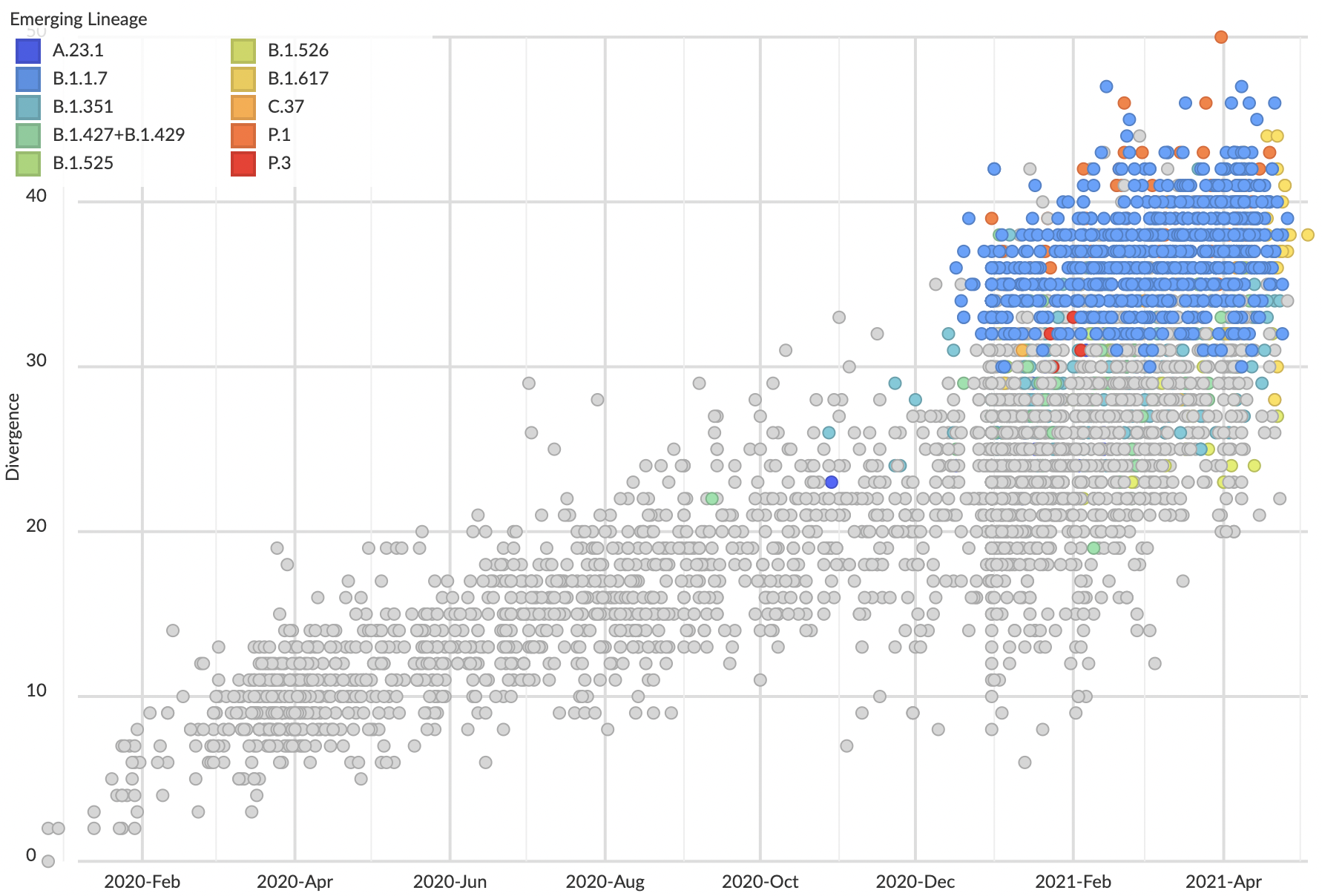
These lineages show excess mutations across the genome
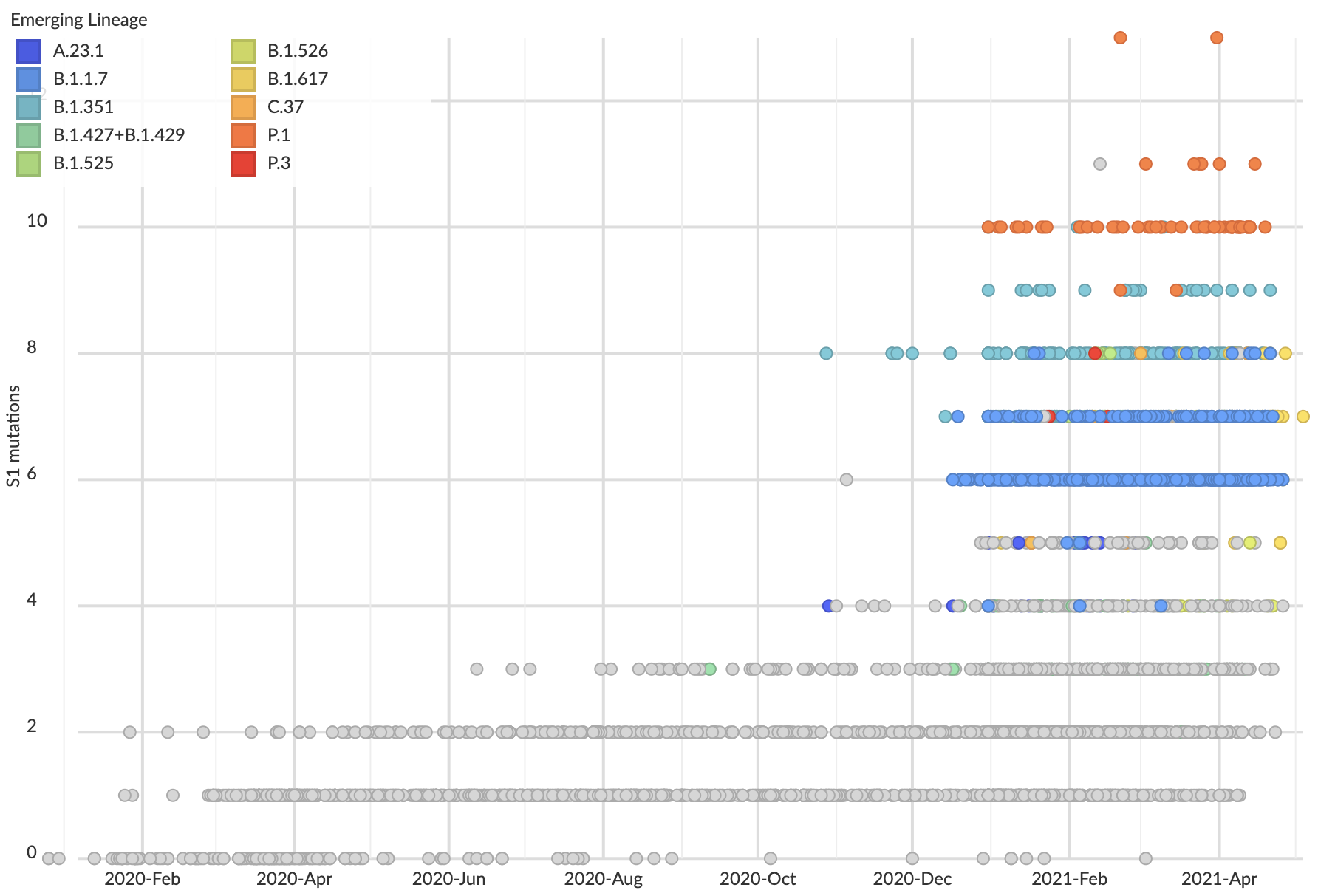
But show most substantial excess in the S1 domain of spike
Evolutionary rate in context
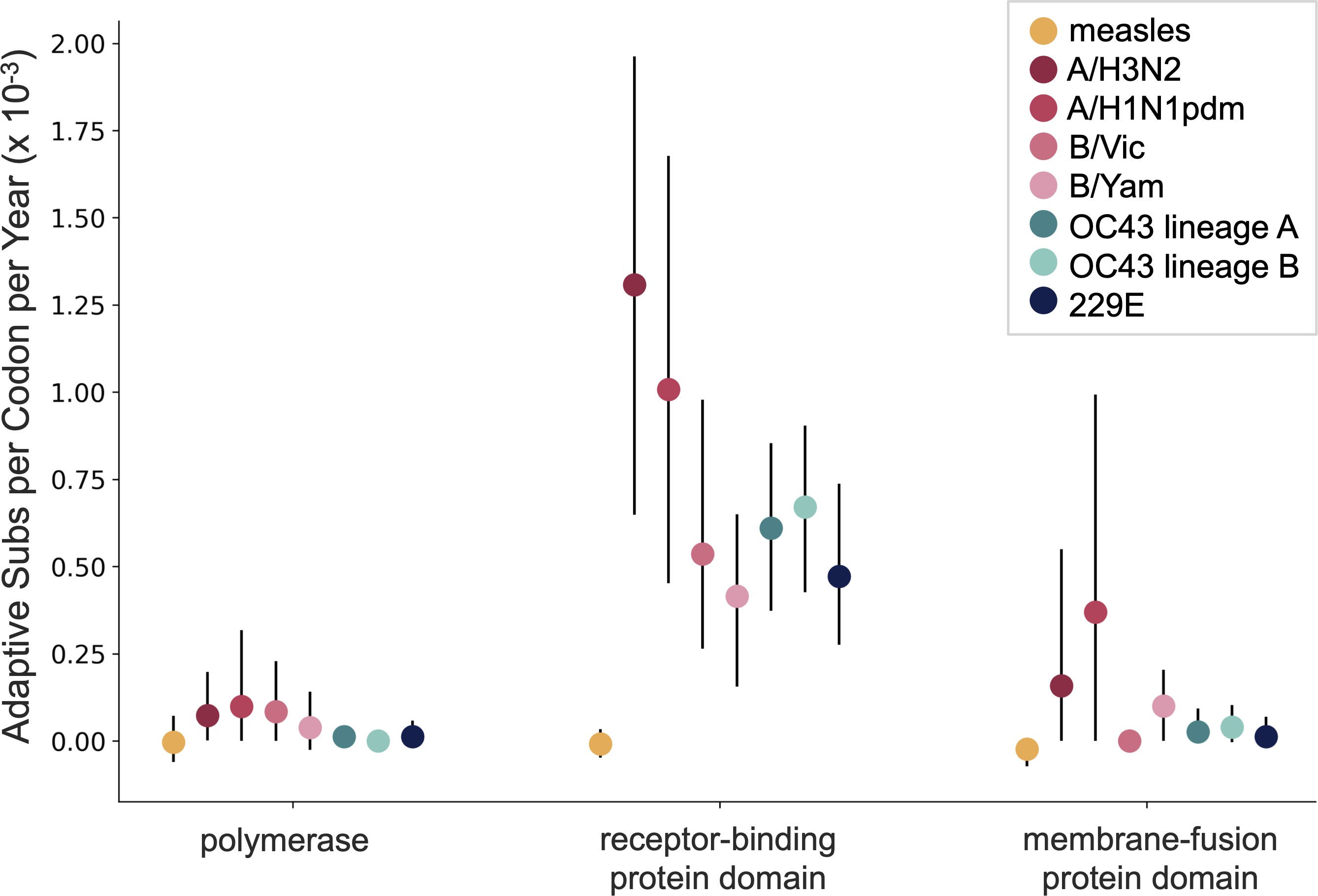
Up to December, my expectation was evolution as seen in seasonal coronaviruses
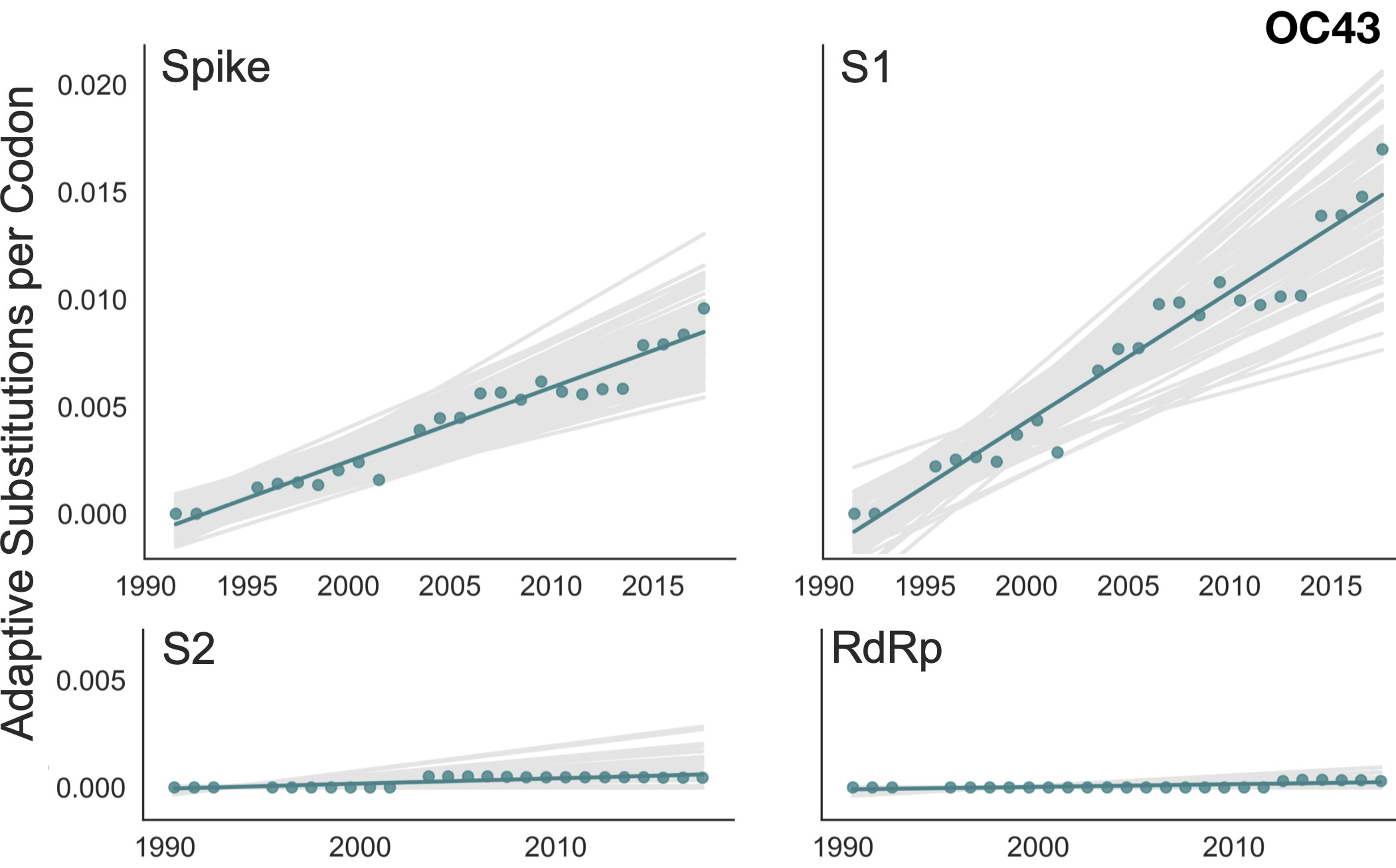
OC43 and 229E show flu B-like rates of adaptive evolution in S1
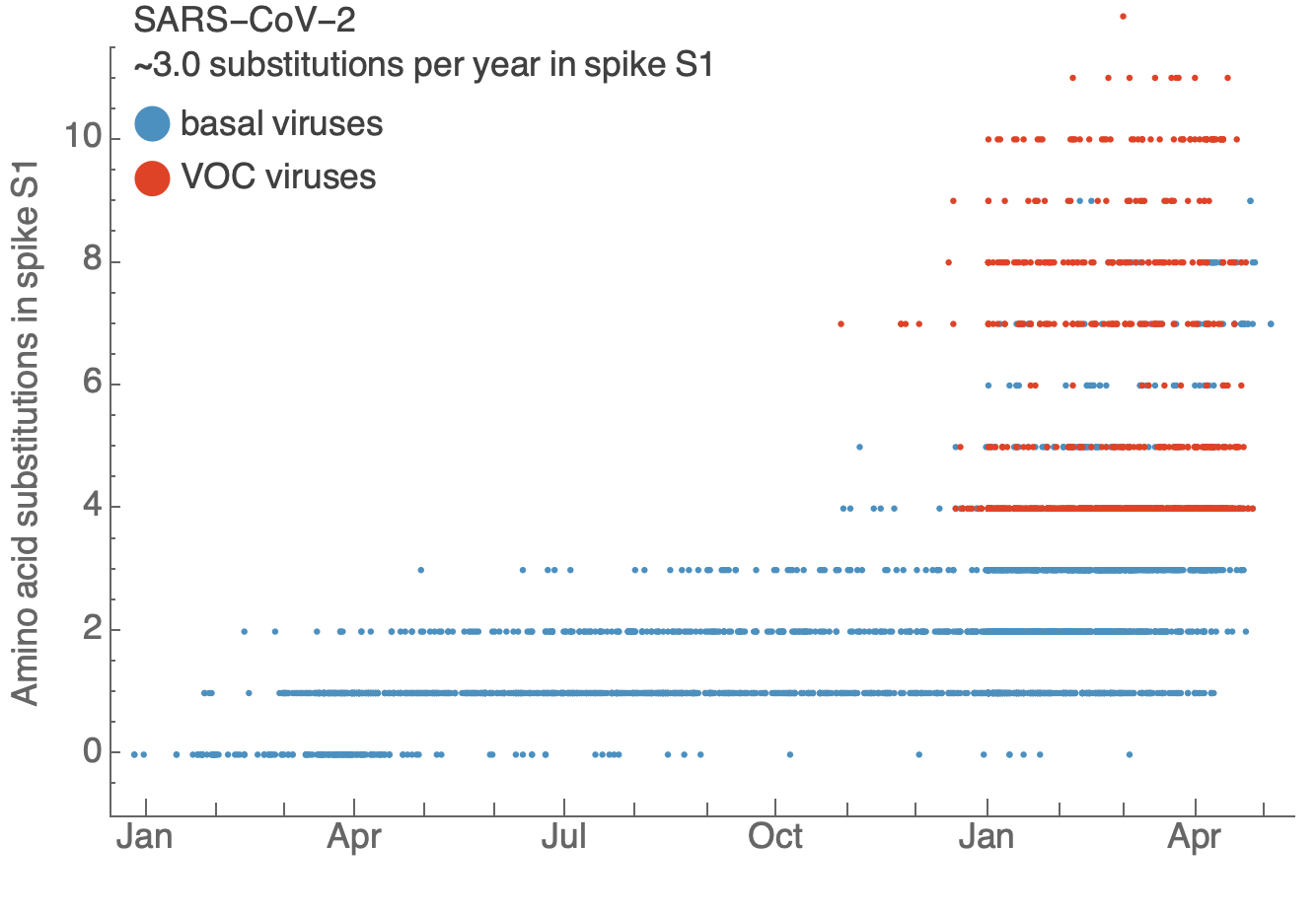
SARS-CoV-2 evolves at a rate of ~3.0 subs per year in spike S1
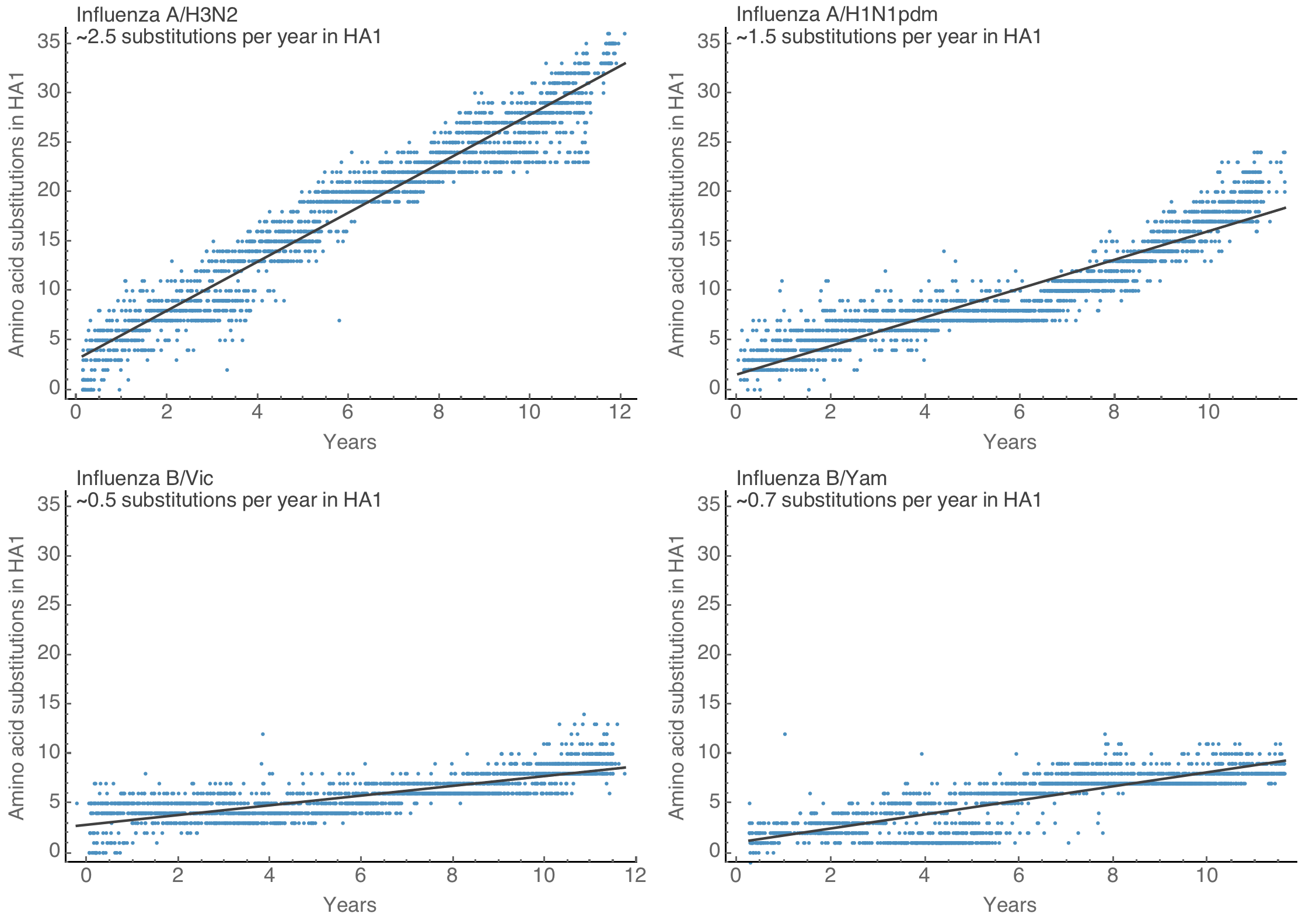
Spike S1 rate of ~3.0 subs per year similar to rate in HA1 of flu A
Influenza H3N2 drifts at ~1 two-fold HI dilution per year
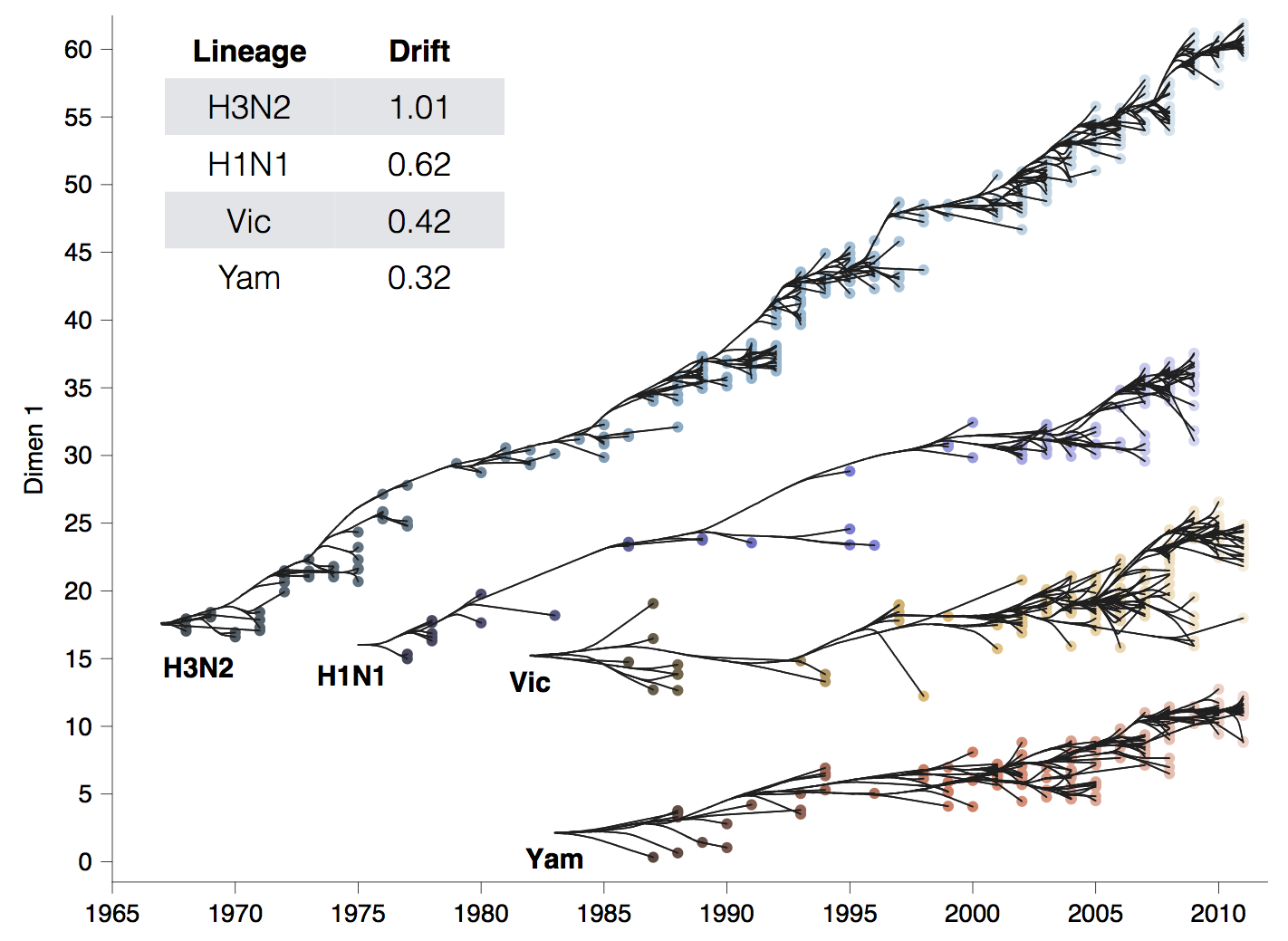
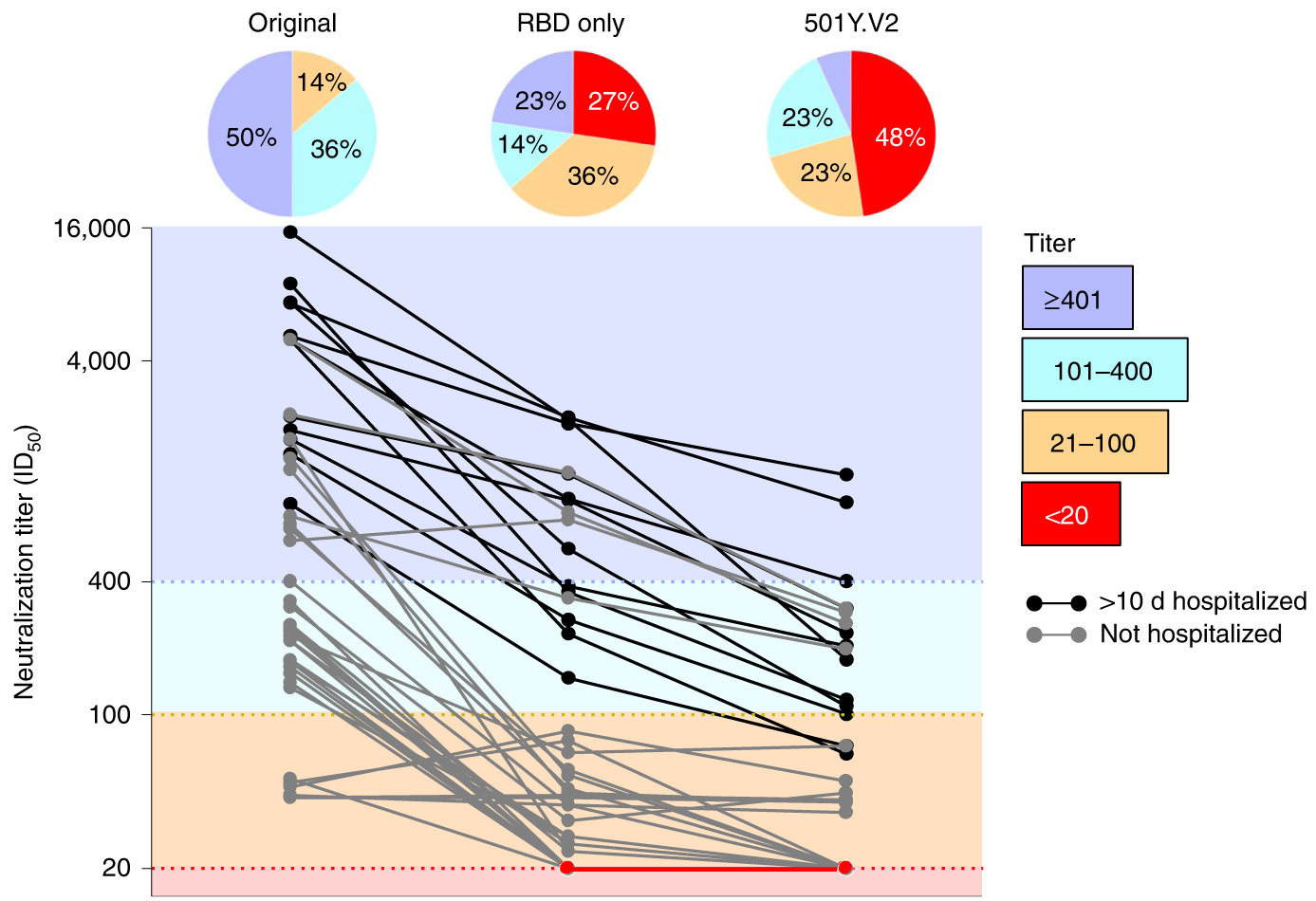
501Y.V2 (B.1.351) shows an ~8-fold drop in neutralization titer, equivalent to ~3 years of H3N2 evolution
Three scenarios for ongoing viral evolution
- Seasonal coronavirus-like scenario: SARS-CoV-2 evolution slows after selection for replication completes. S1 is relatively inflexible in terms of antigenic evolution. New strains emerge rarely and replace existing diversity. Vaccine updates required every ~5 years.
- Influenza H3N2-like scenario: SARS-CoV-2 evolution slows after selection for replication completes, but S1 remains highly antigenically flexible. New strains emerge often and replace existing diversity. Vaccine updates required every 1-2 years.
- HIV-like scenario: SARS-CoV-2 evolution slows after selection for replication completes, but S1 remains highly antigenically flexible. New strains emerge and co-circulate leading to continual antigenic diversification. Vaccine updates cannot cover diversity of circulating viruses.
Acknowledgements
SARS-CoV-2 genomic epi: Data producers from all over the world, GISAID and the Nextstrain team
Bedford Lab:
![]() Alli Black,
Alli Black,
![]() John Huddleston,
John Huddleston,
![]() James Hadfield,
James Hadfield,
![]() Katie Kistler,
Katie Kistler,
![]() Louise Moncla,
Louise Moncla,
![]() Maya Lewinsohn,
Maya Lewinsohn,
![]() Thomas Sibley,
Thomas Sibley,
![]() Jover Lee,
Jover Lee,
![]() Kairsten Fay,
Kairsten Fay,
![]() Misja Ilcisin,
Misja Ilcisin,
![]() Cassia Wagner,
Cassia Wagner,
![]() Miguel Paredes,
Miguel Paredes,
![]() Nicola Müller,
Nicola Müller,
![]() Marlin Figgins,
Marlin Figgins,
![]() Eli Harkins
Eli Harkins






