Real-time tracking of virus evolution
Trevor Bedford (@trvrb)
27 Jan 2016
Combi Seminar
Genome Sciences, University of Washington
Slides at bedford.io/talks/
Phylogenies
Phylogenies describe history
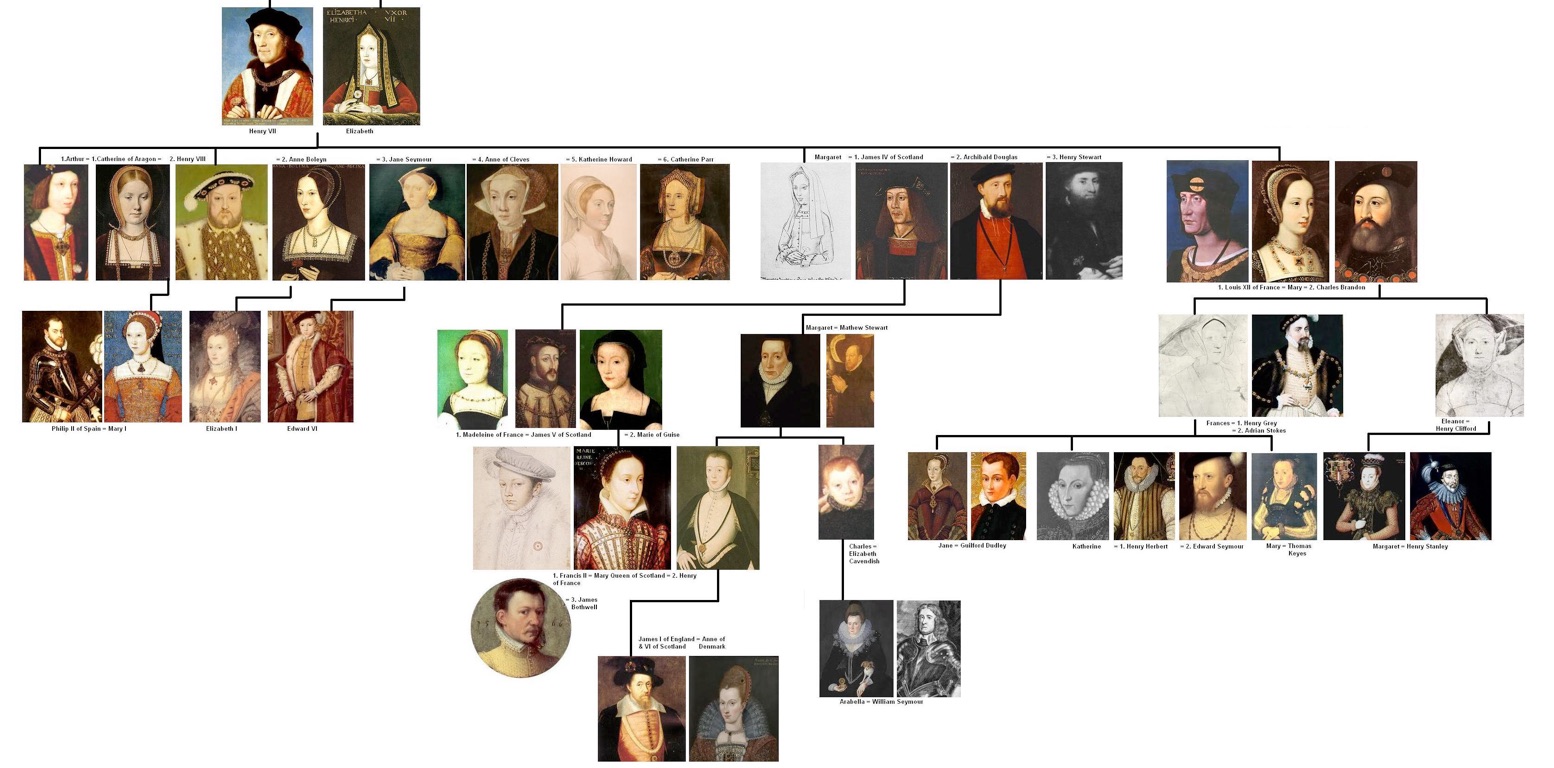
Phylogenies describe history
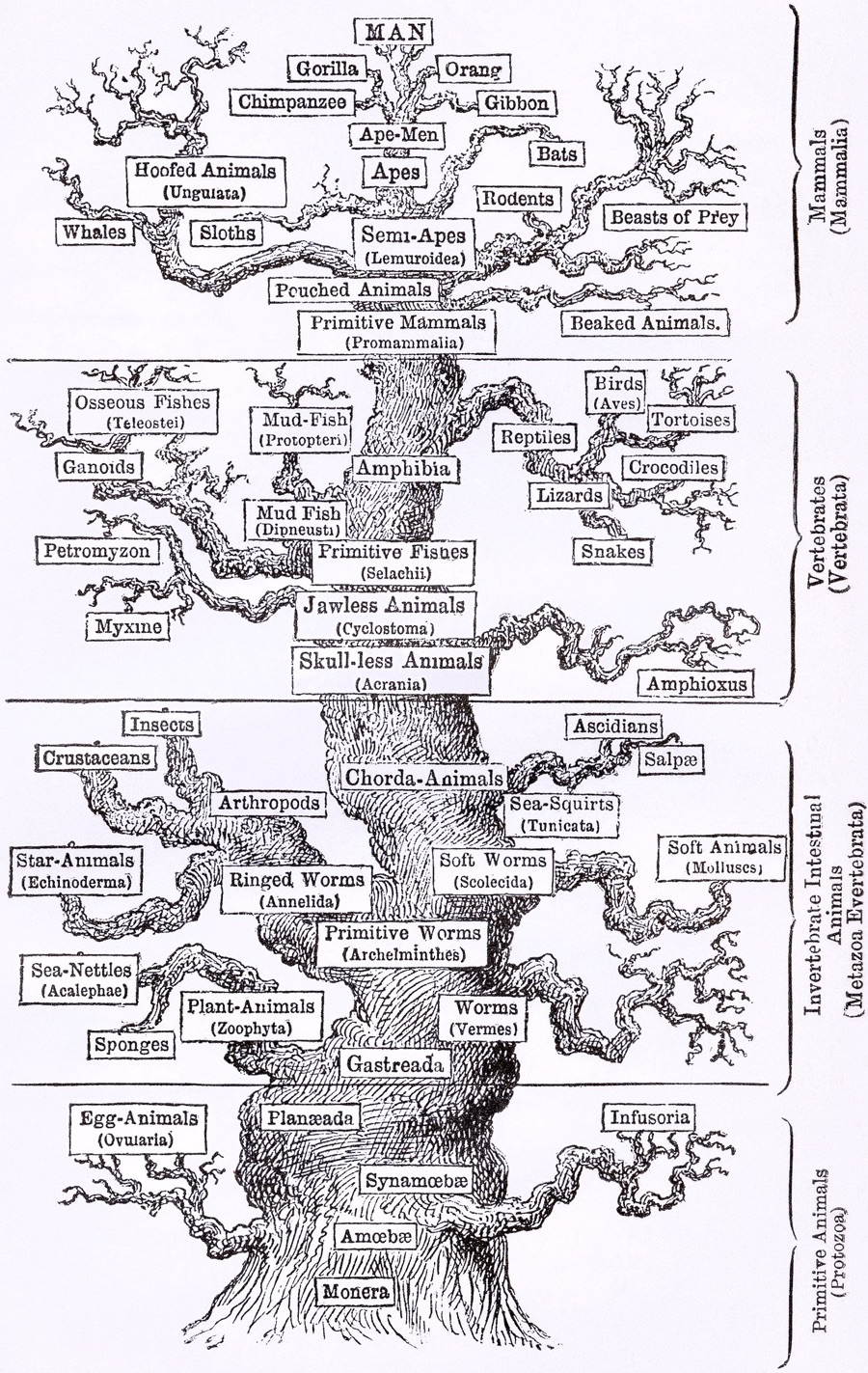
Phylogenies describe history
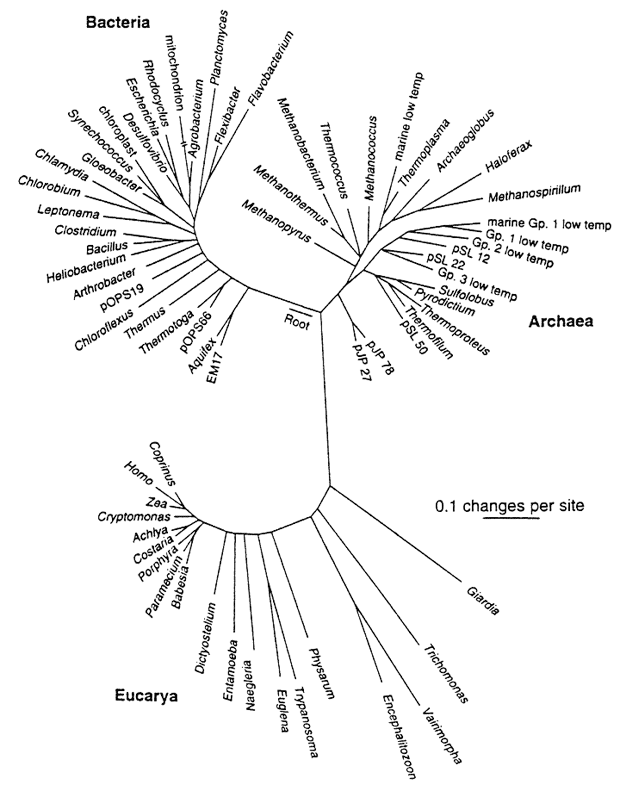
Phylogenies reveal process
Epidemics
Epidemic process
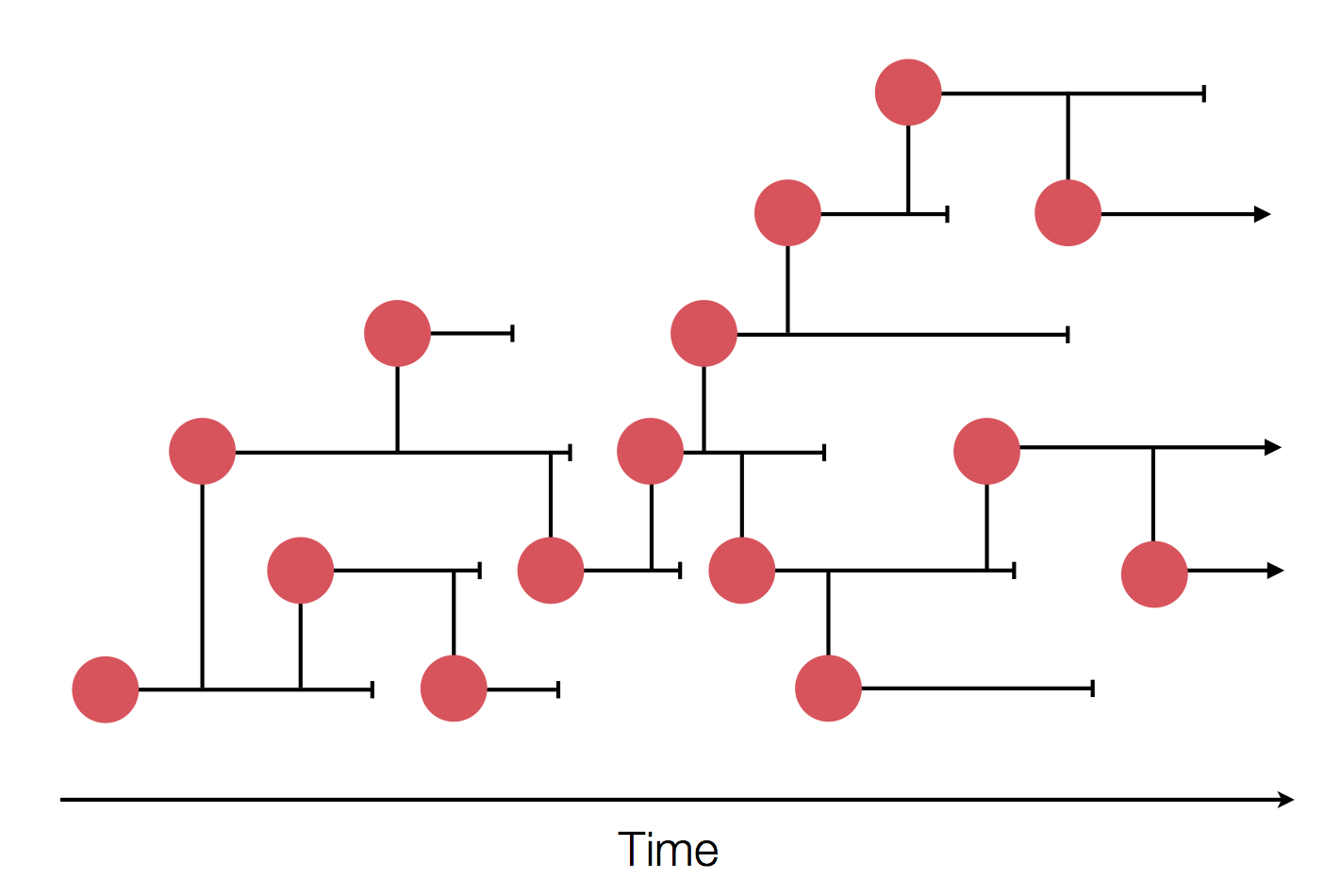
Sample some individuals
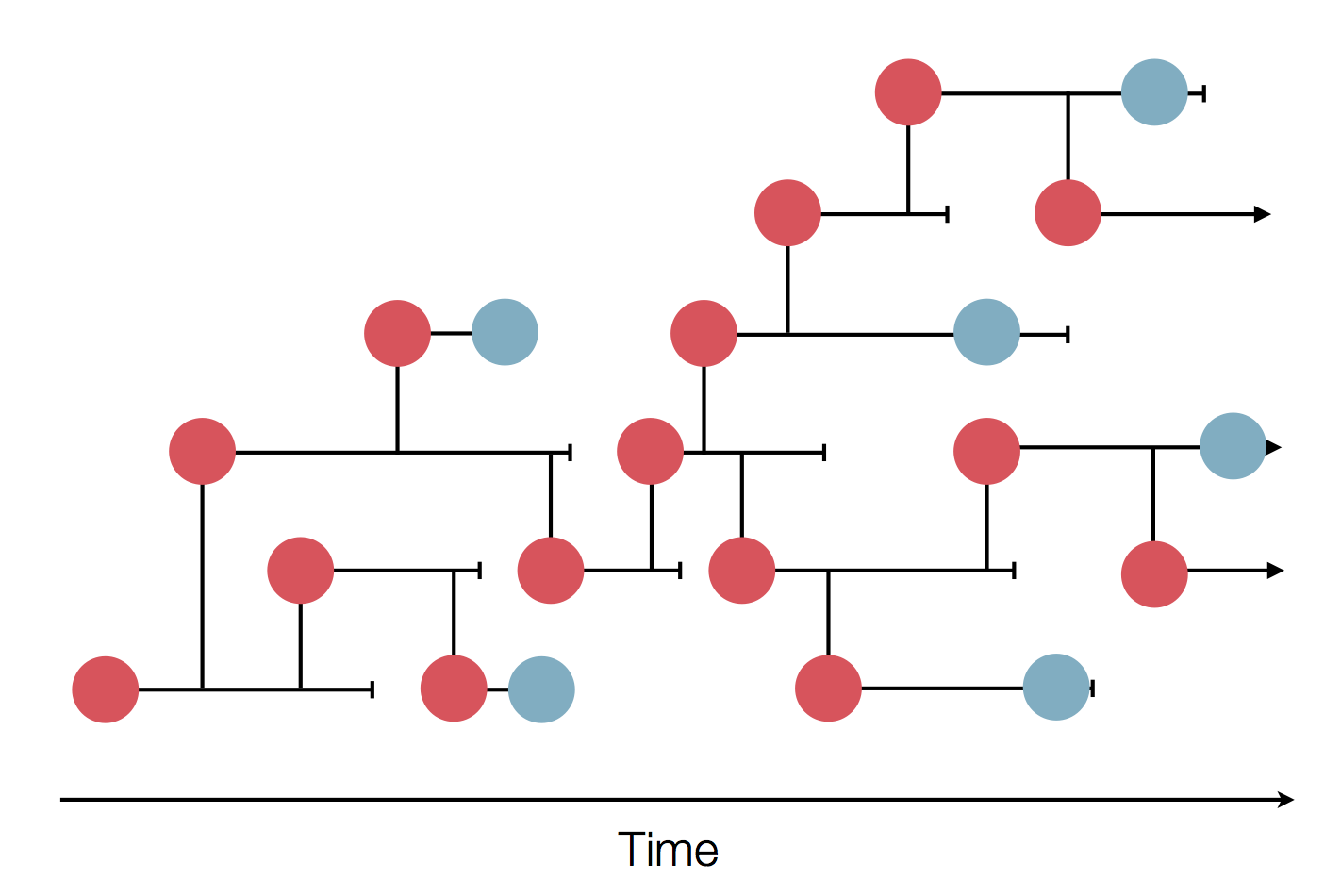
Sequence and determine phylogeny
Sequence and determine phylogeny
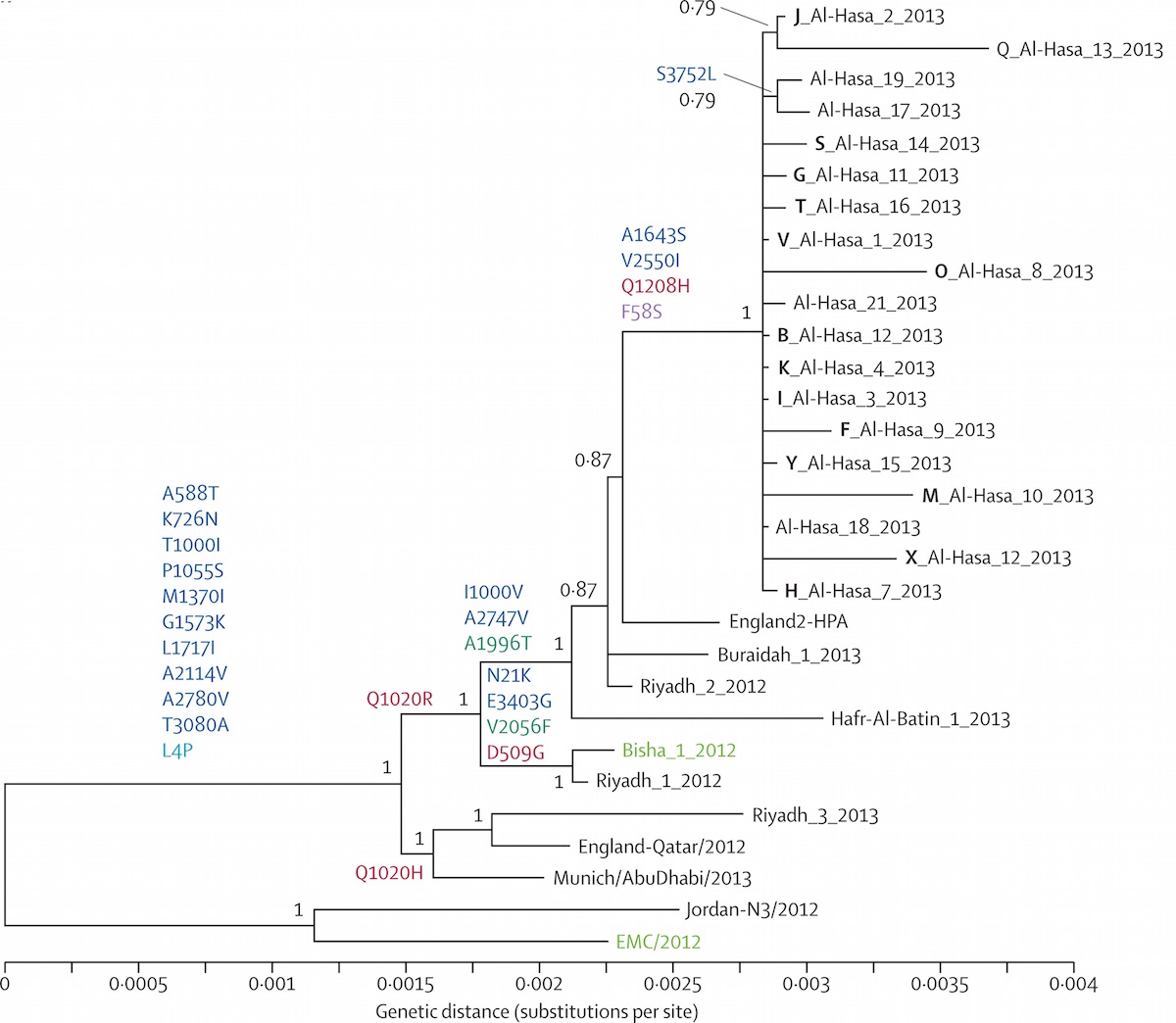
Middle Eastern MERS-CoV phylogeny
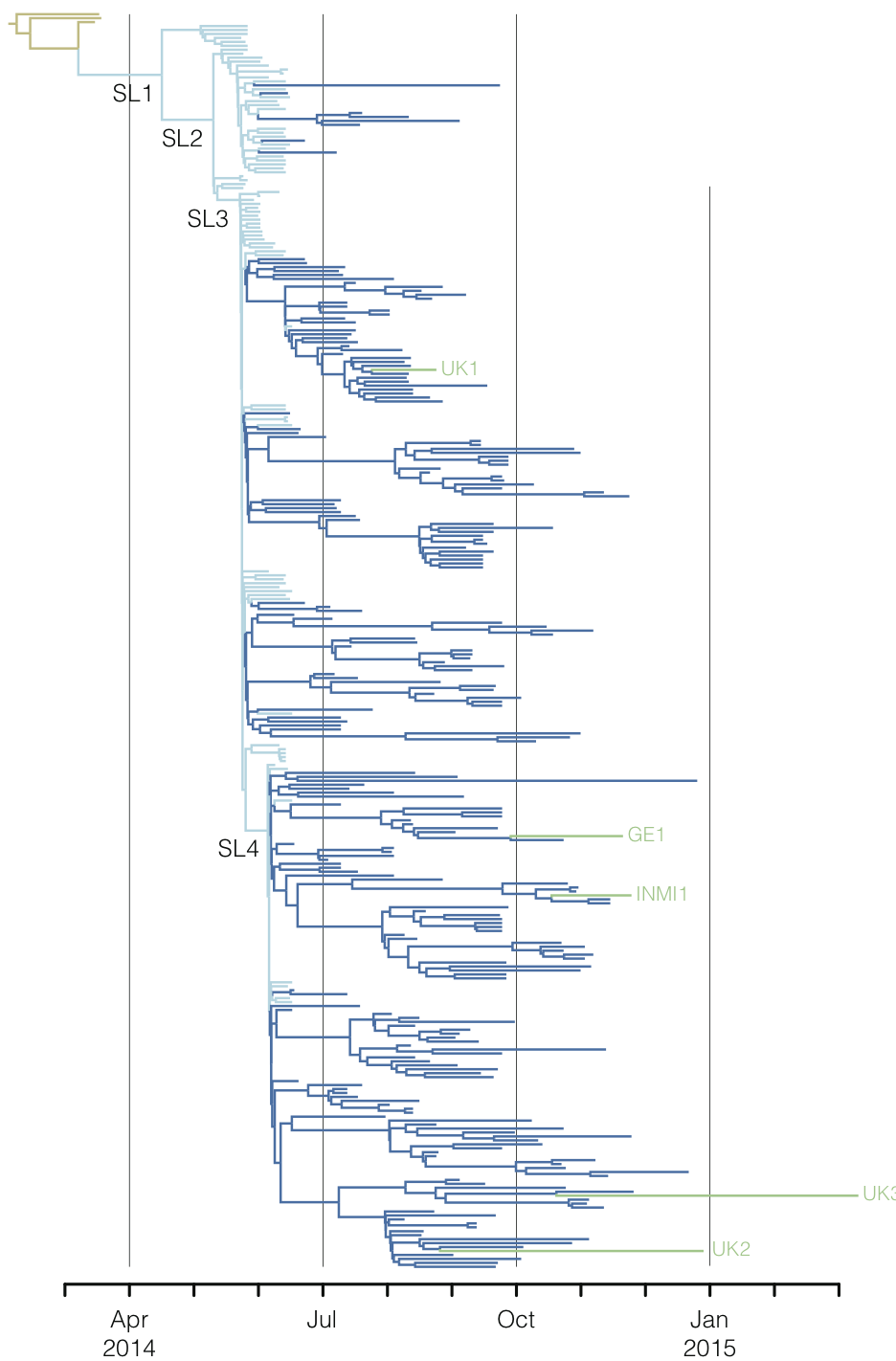
West African Ebola phylogeny
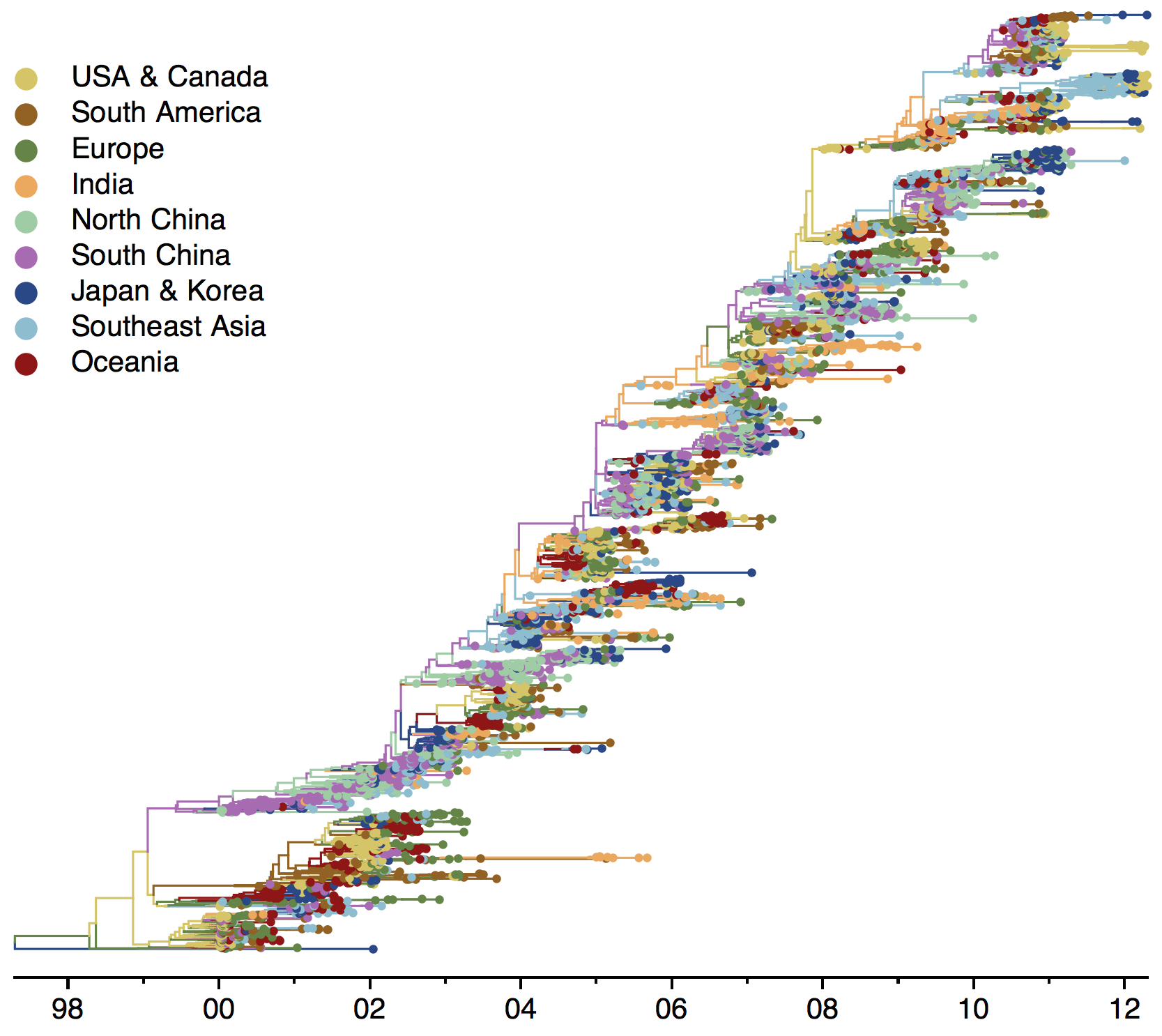
Global influenza phylogeny
Applications of evolutionary analysis for influenza vaccine strain selection and charting outbreak spread
Influenza
Influenza virion
Influenza H3N2 vaccine updates
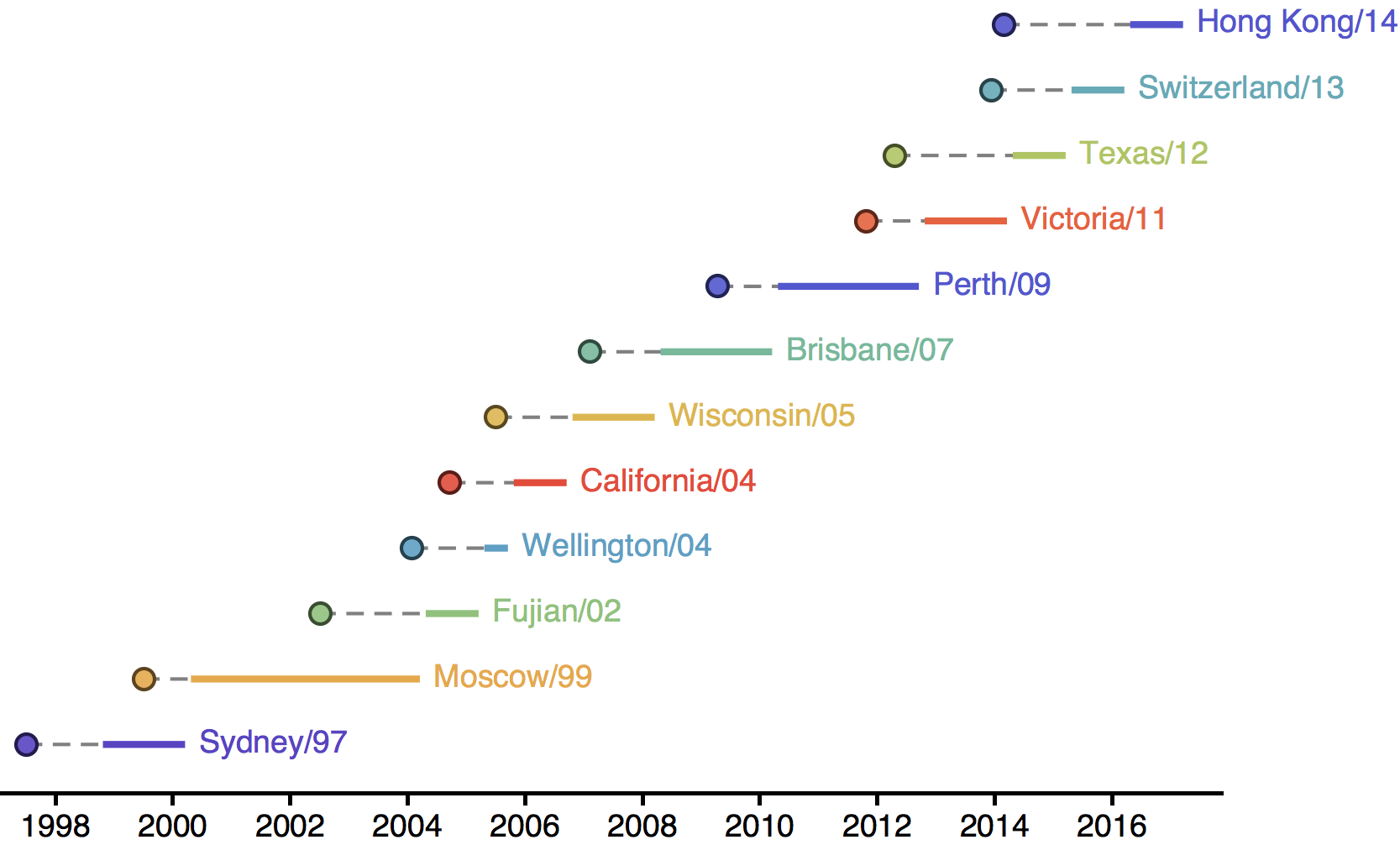
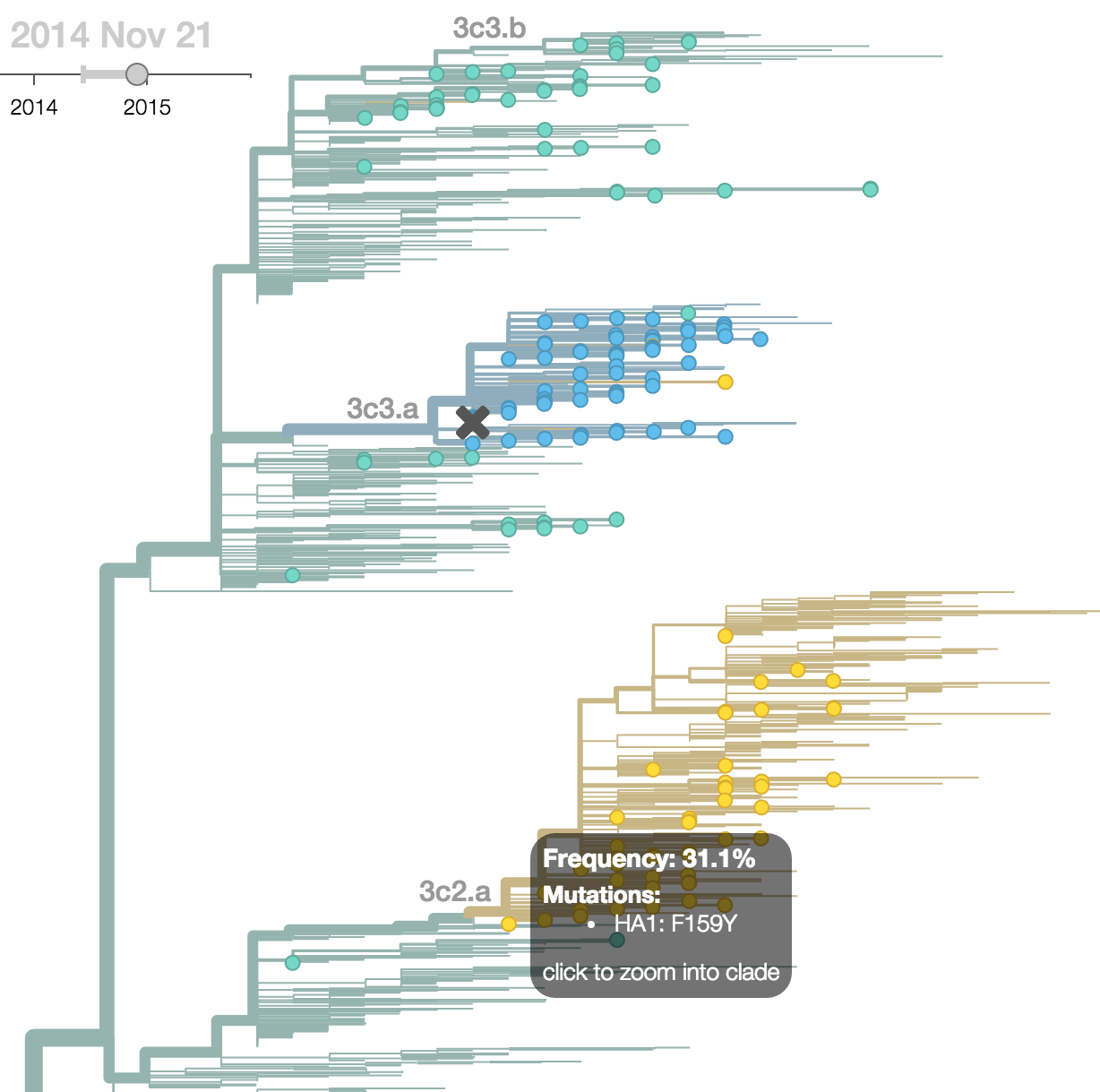
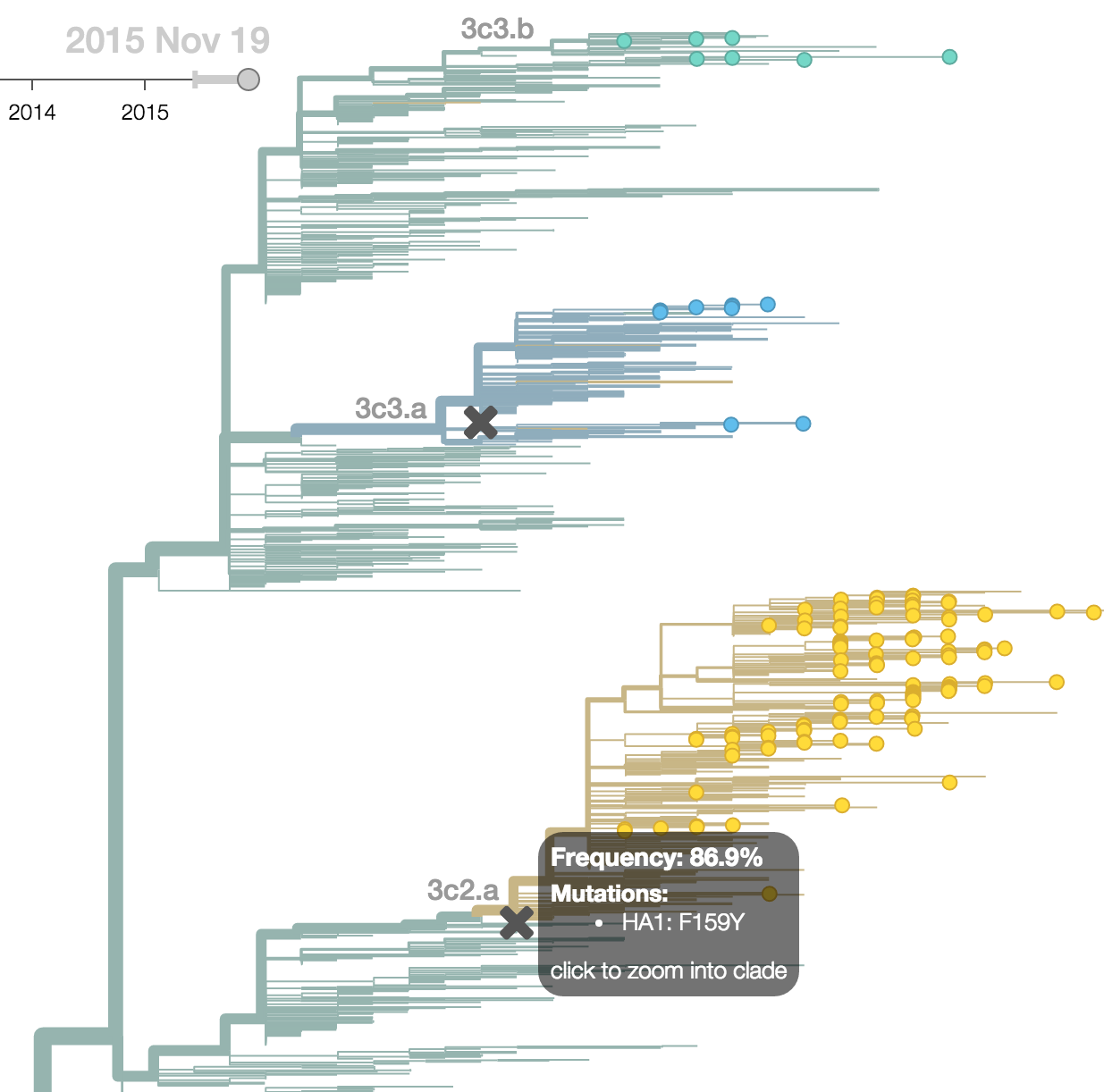
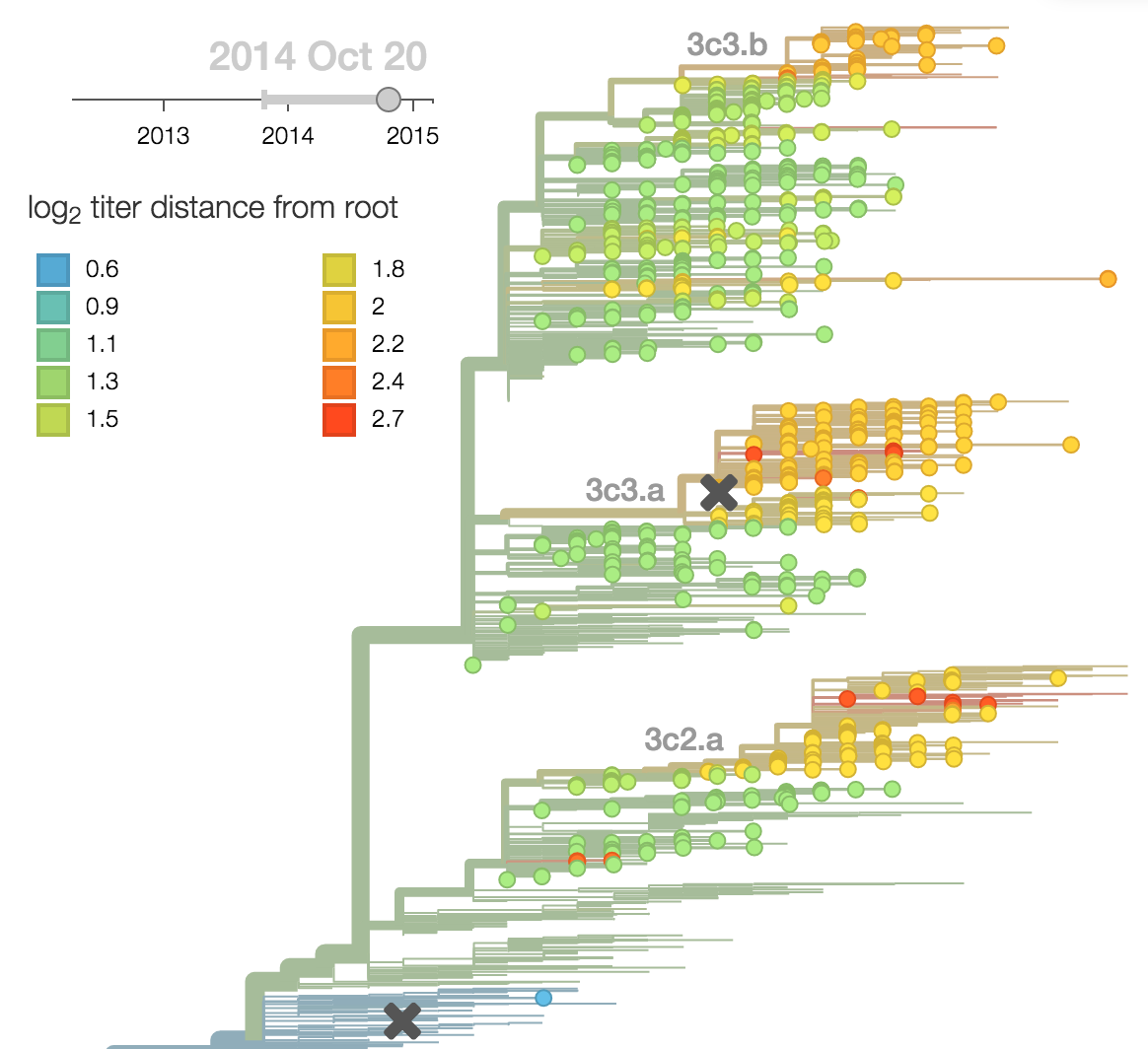
H3N2 phylogeny showing antigenic drift
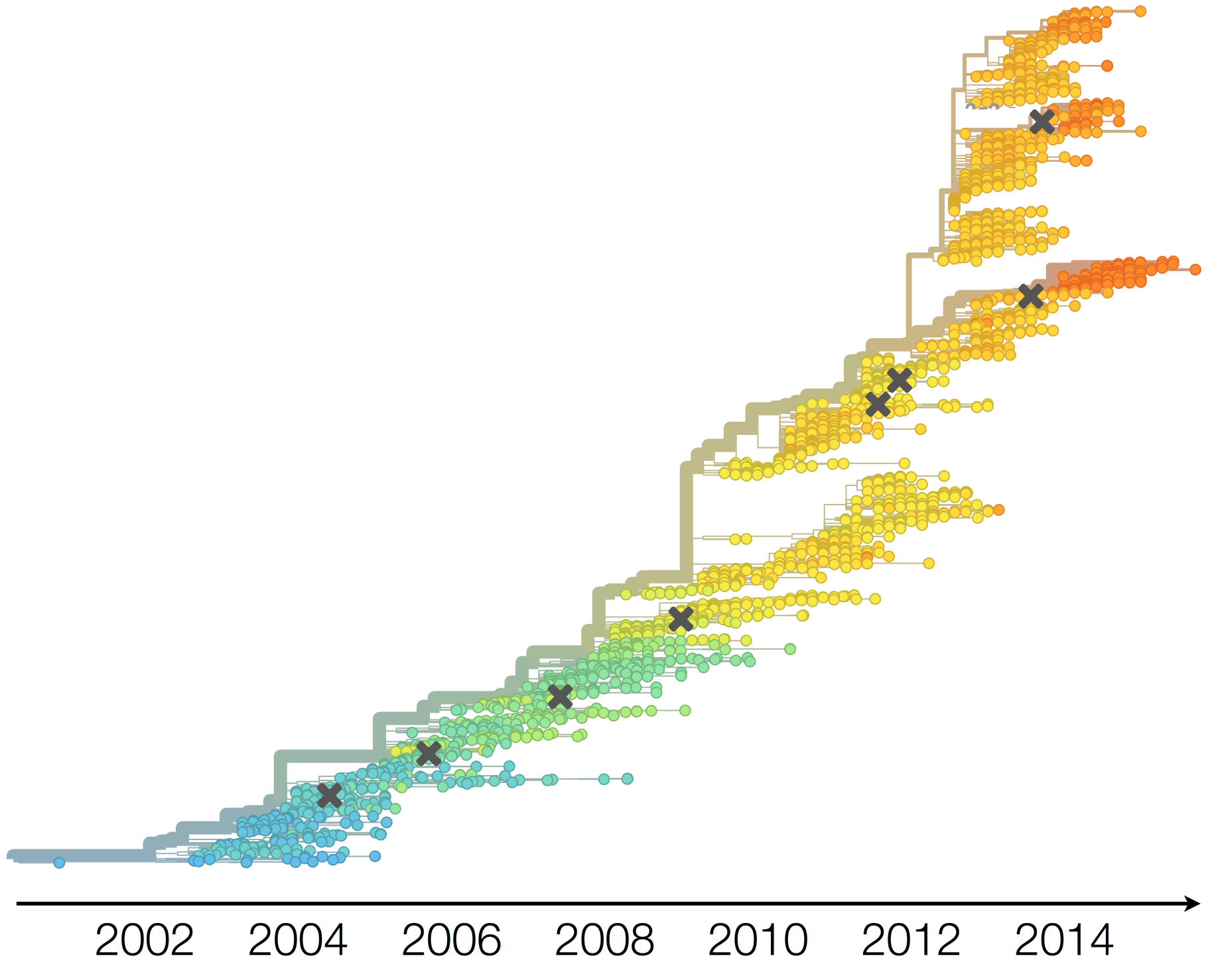
H3N2 phylogeny showing antigenic drift
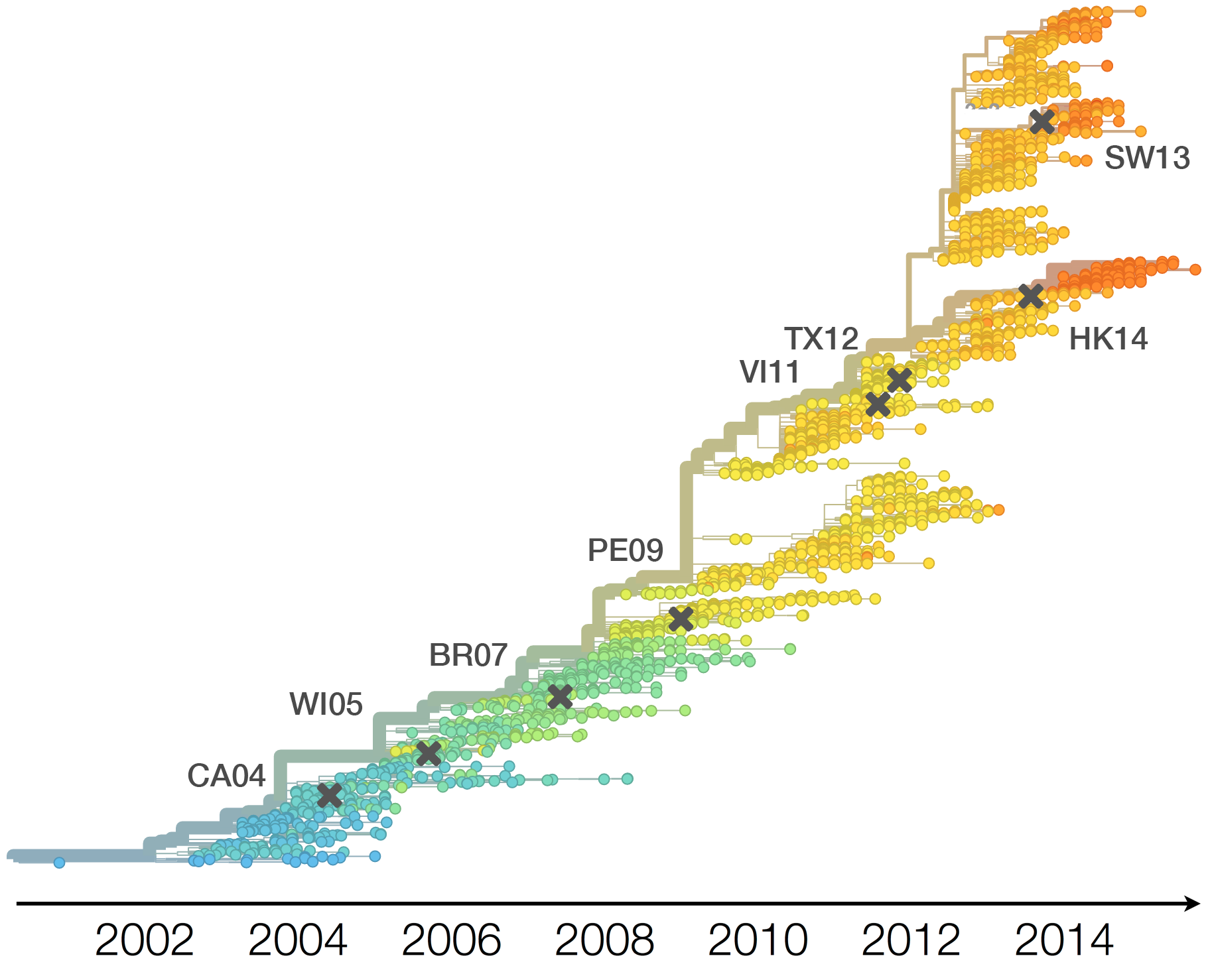
Drift variants rapidly take over the virus population
Timely surveillance and rapid analysis essential to understand ongoing influenza evolution
nextflu
Project to provide a real-time view of the evolving influenza population
All in collaboration with Richard Neher

nextflu pipeline
- Download all recent HA sequences from GISAID
- Filter to remove outliers
- Subsample across time and space
- Align sequences
- Build tree
- Estimate frequencies
- Export for visualization
Up-to-date analysis publicly available at:
nextflu.org
Antigenic evolution
Influenza hemagglutination inhibition (HI) assay
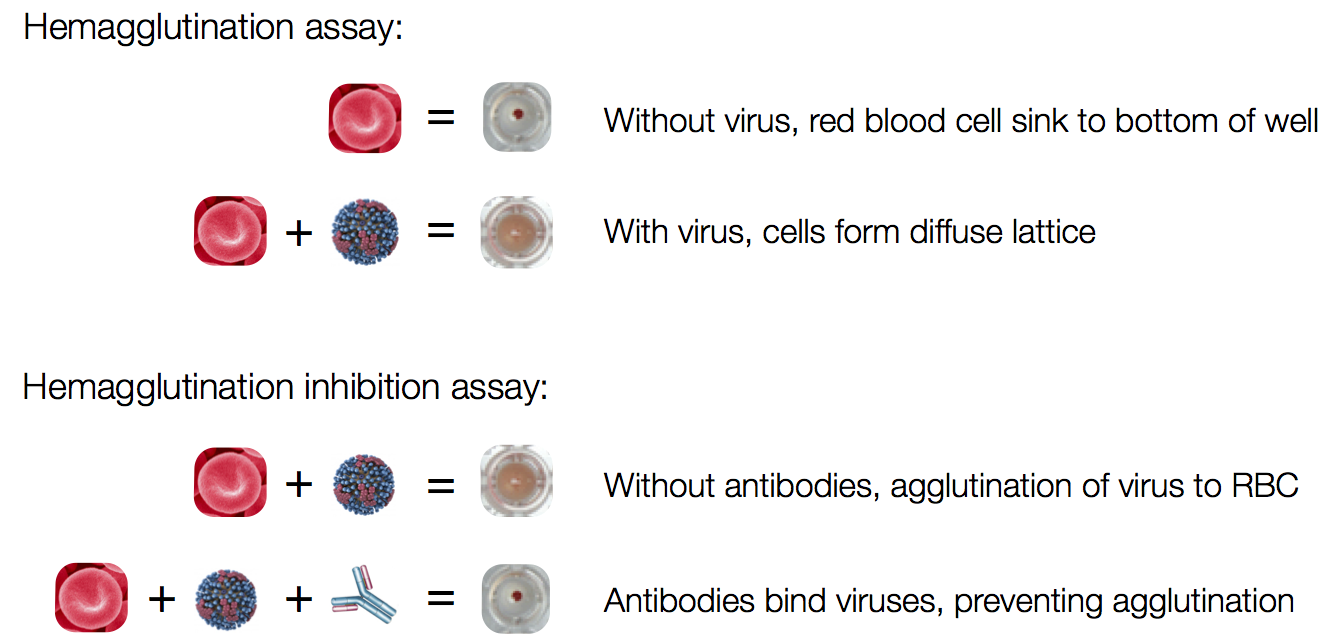
HI measures cross-reactivity across viruses
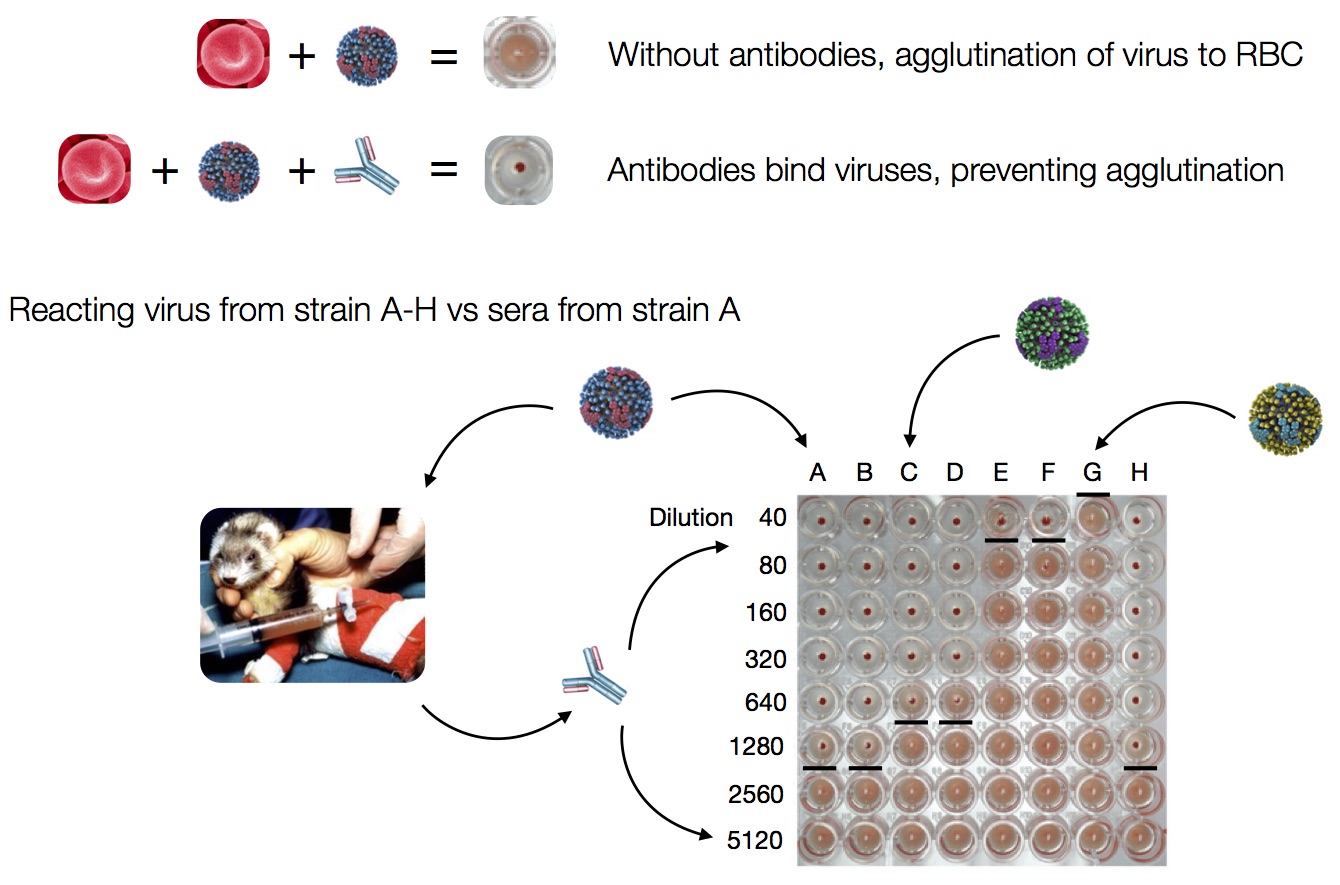
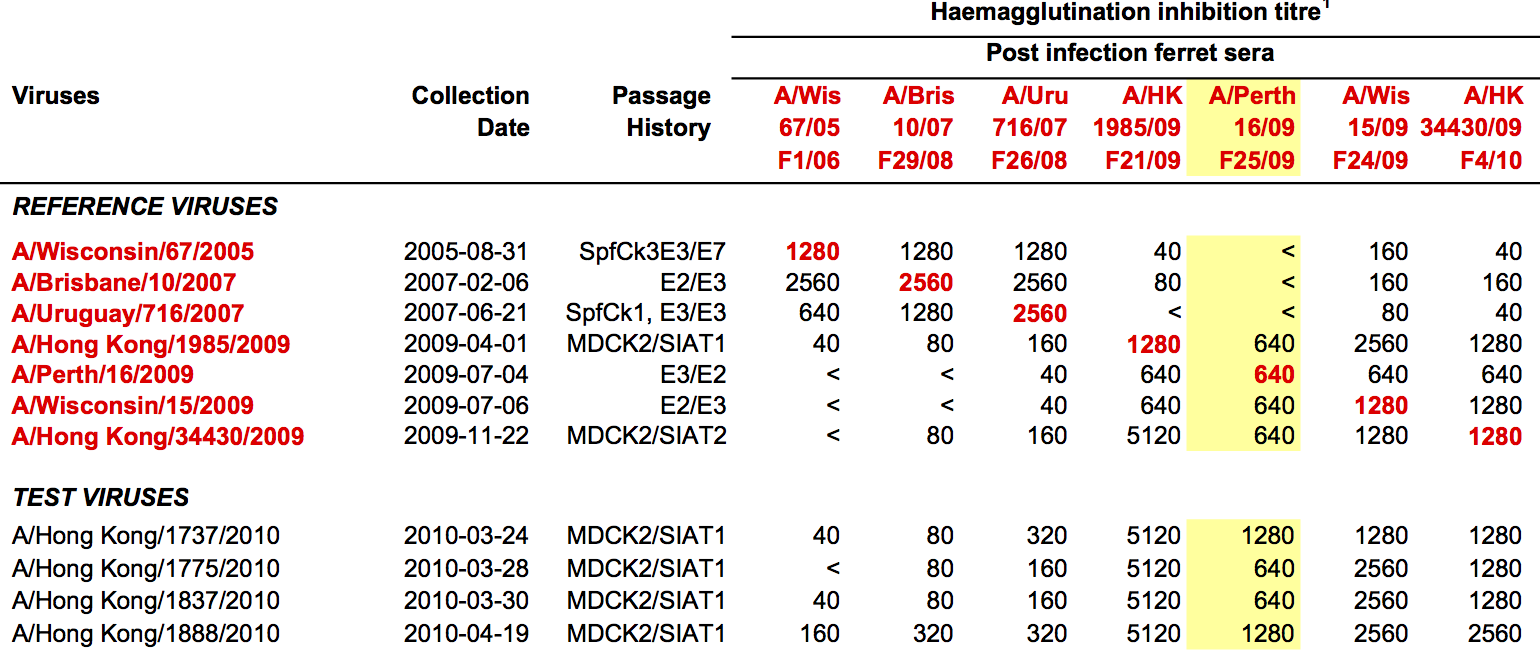
Data in the form of table of maximum inhibitory titers
Fit HI titer drops to phylogeny branches
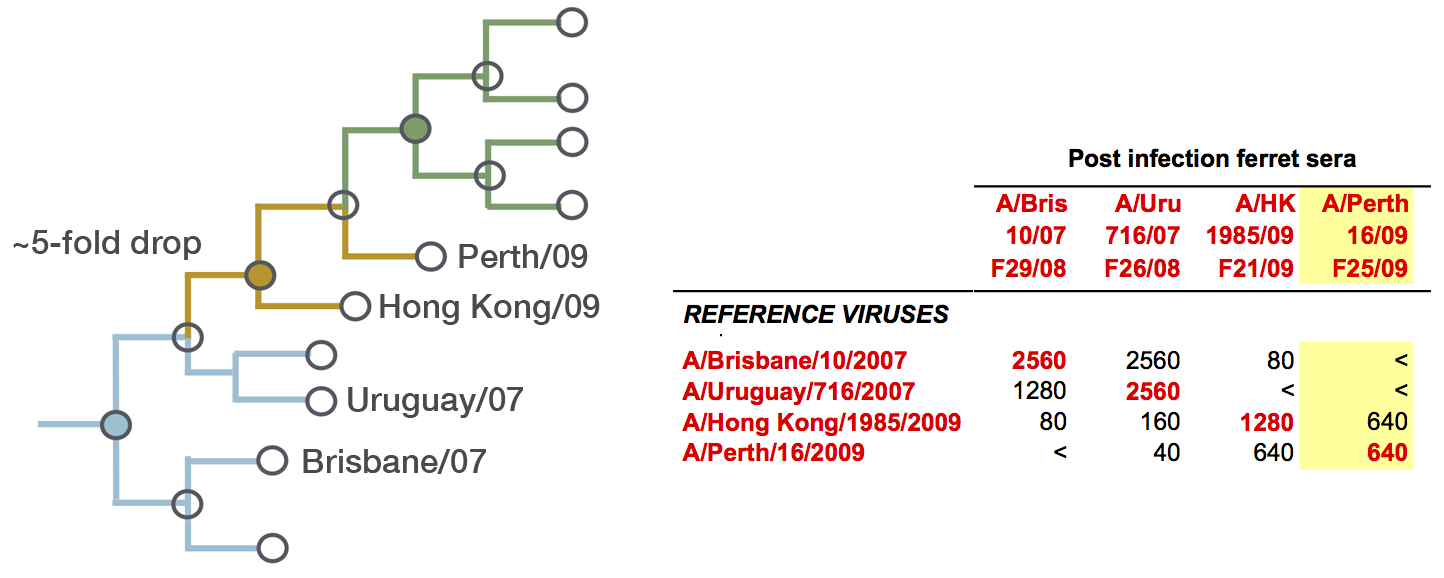
Model is highly predictive of missing titer values
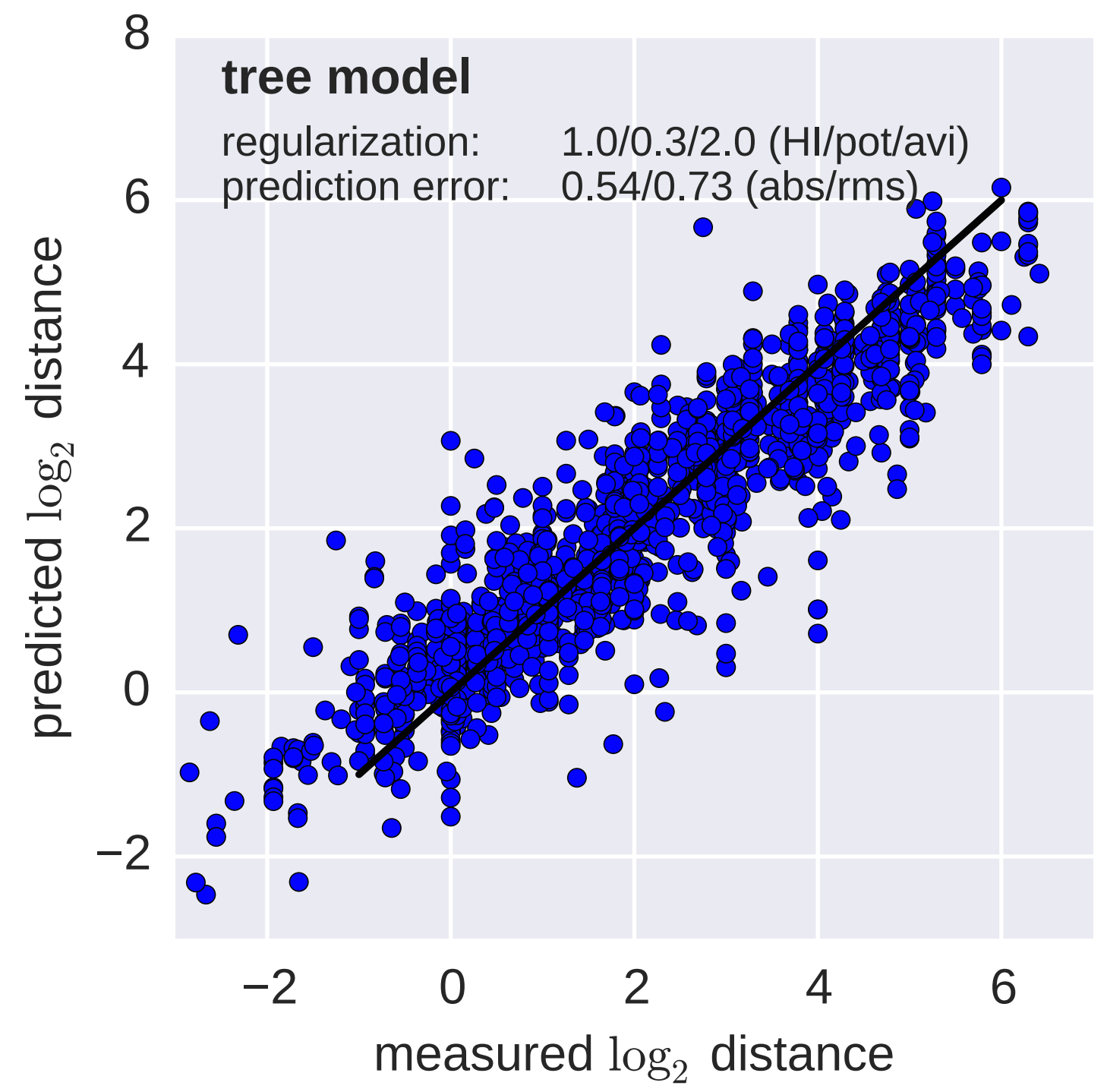
Recent HI data from WHO CC London annual and interim reports
Up-to-date analysis at:
nextflu.org
Forecasting
The future is here, it's just not evenly distributed yet
— William Gibson
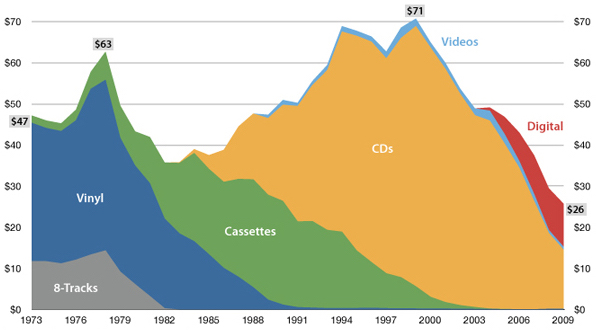
USA music industry, 2011 dollars per capita
Influenza population turnover
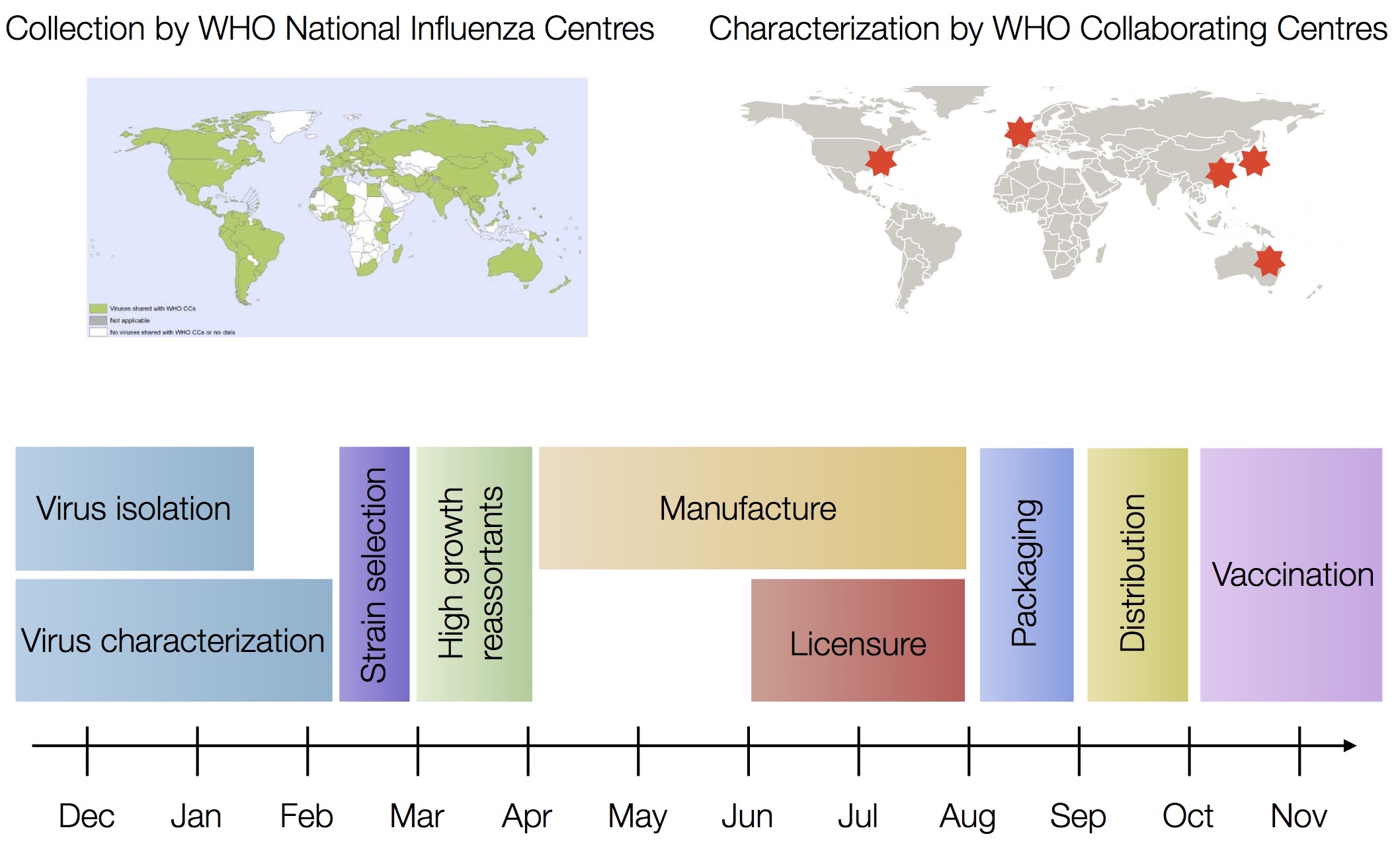
Vaccine strain selection timeline
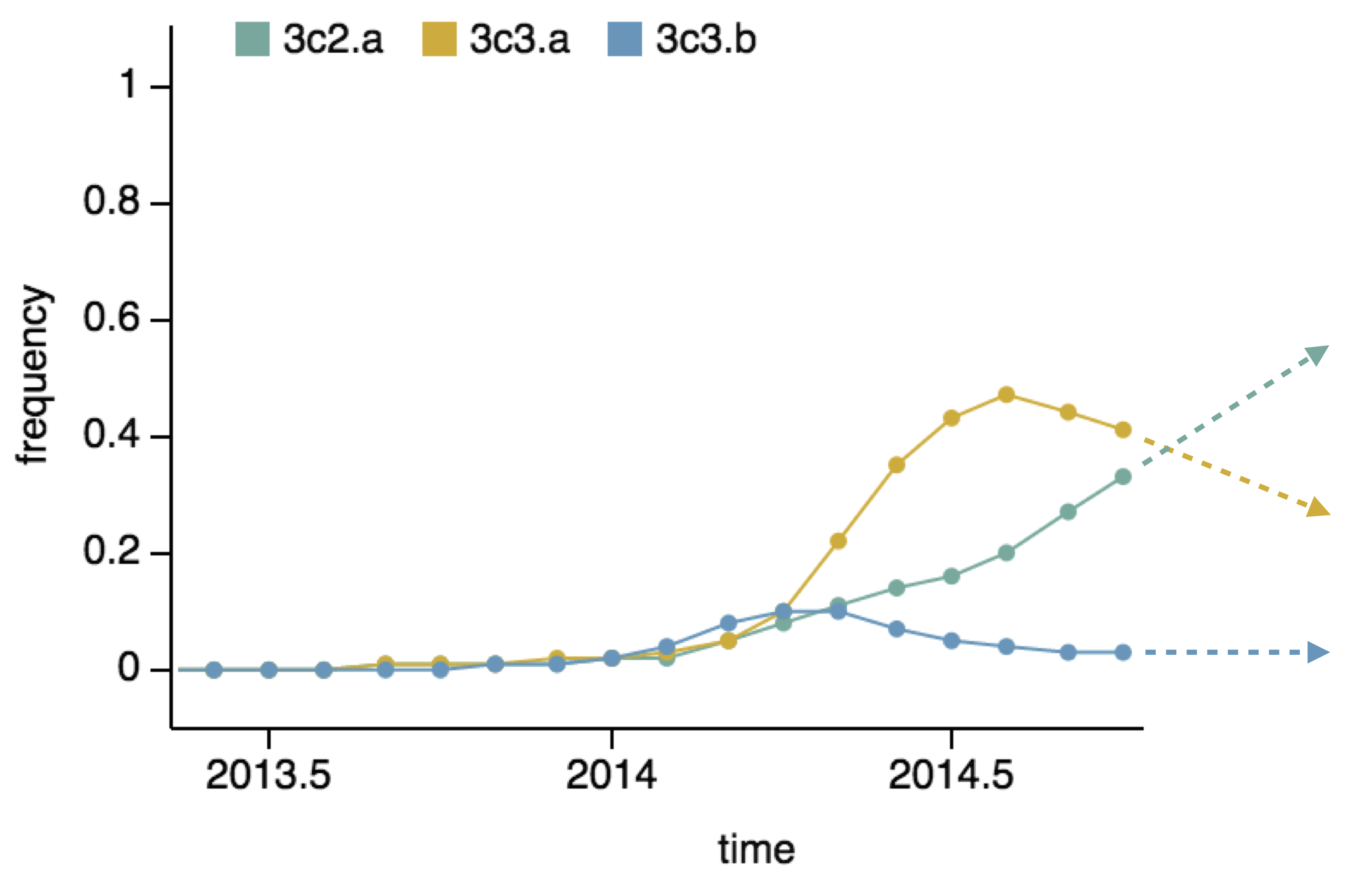
Seek to explain change in clade frequencies over 1 year
Fitness models can project clade frequencies
Clade frequencies $X$ derive from the fitnesses $f$ and frequencies $x$ of constituent viruses, such that
$$\hat{X}_v(t+\Delta t) = \sum_{i:v} x_i(t) \, \mathrm{exp}(f_i \, \Delta t)$$
This captures clonal interference between competing lineages
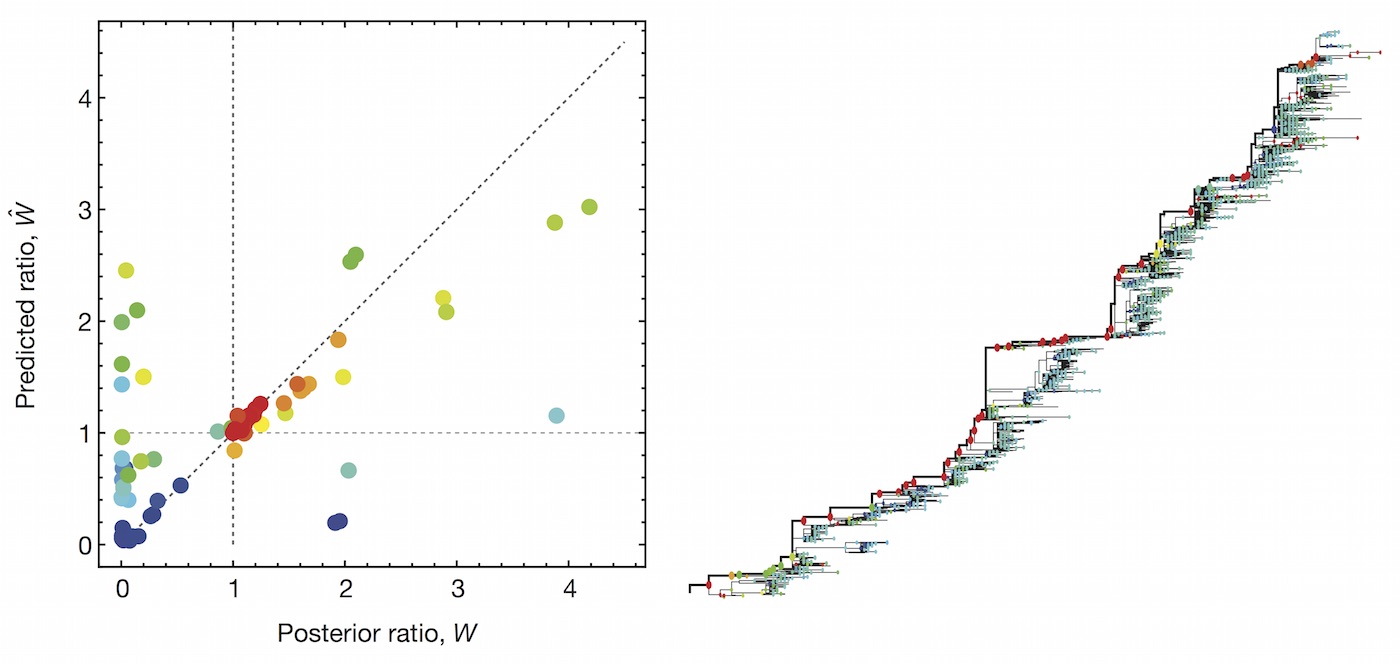
Predictive fitness models
A simple predictive model estimates the fitness $f$ of virus $i$ as
$$\hat{f}_i = \beta^\mathrm{ep} \, f_i^\mathrm{ep} + \beta^\mathrm{ne} \, f_i^\mathrm{ne}$$
where $f_i^\mathrm{ep}$ measures cross-immunity via substitutions at epitope sites and $f_i^\mathrm{ep}$ measures mutational load via substitutions at non-epitope sites
We implement a similar model based on two predictors
- Clade frequency change
- Antigenic advancement
Project frequencies forward,
growing clades have high fitness
Calculate HI drop from ancestor,
drifted clades have high fitness
Fitness model parameterization
Our predictive model estimates the fitness $f$ of virus $i$ as
$$\hat{f}_i = \beta^\mathrm{freq} \, f_i^\mathrm{freq} + \beta^\mathrm{HI} \, f_i^\mathrm{HI}$$
We learn coefficients and validate model based on previous 15 H3N2 seasons
Clade growth rate is well predicted
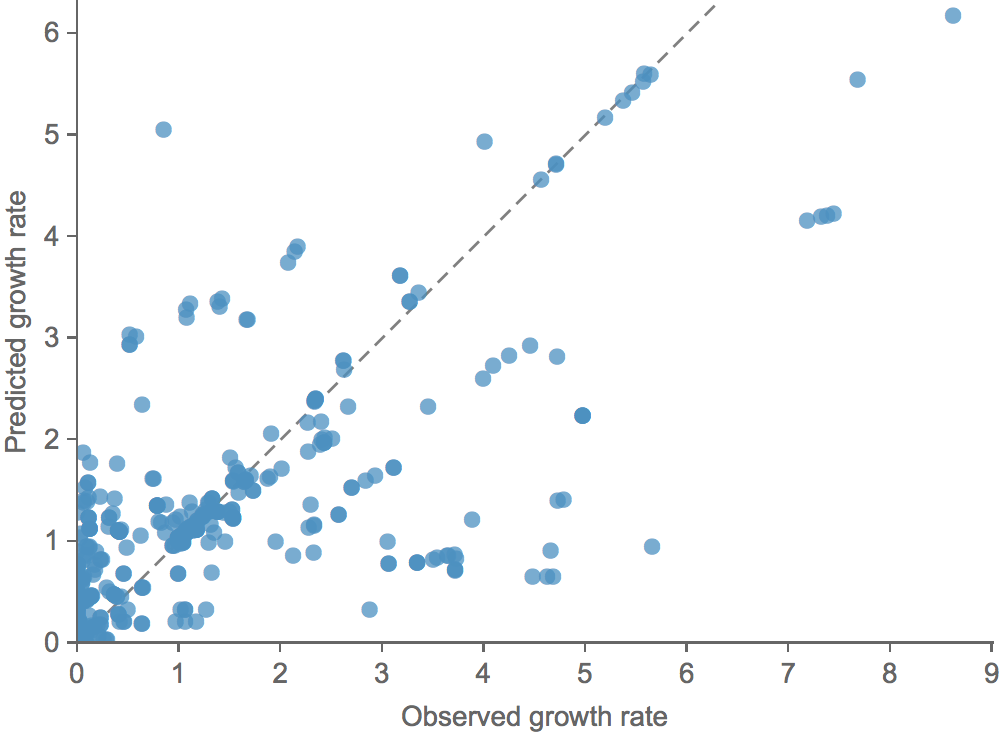
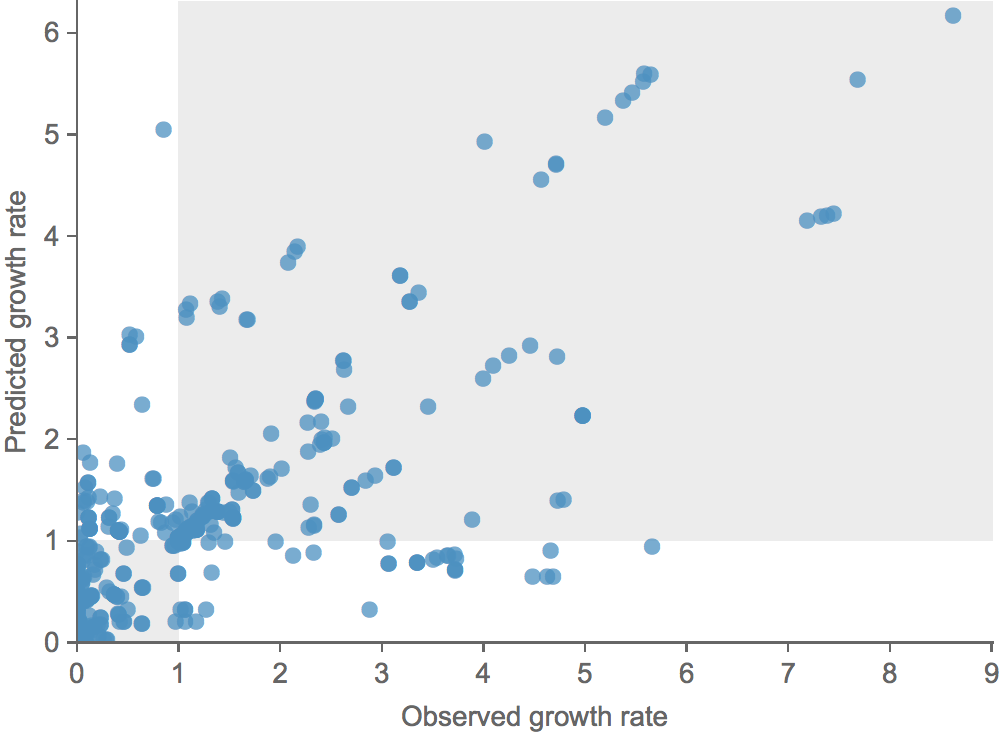
Growth vs decline correct in 83% of cases
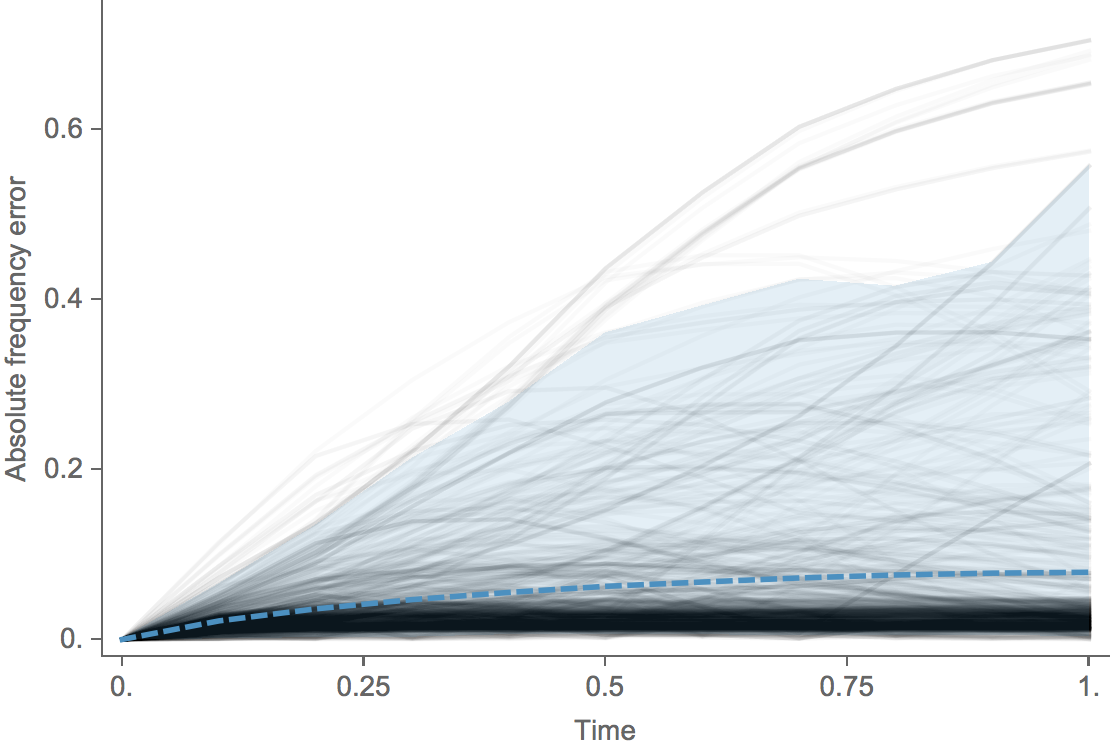
Clade error increases steadily over time
Trajectories show more detailed congruence
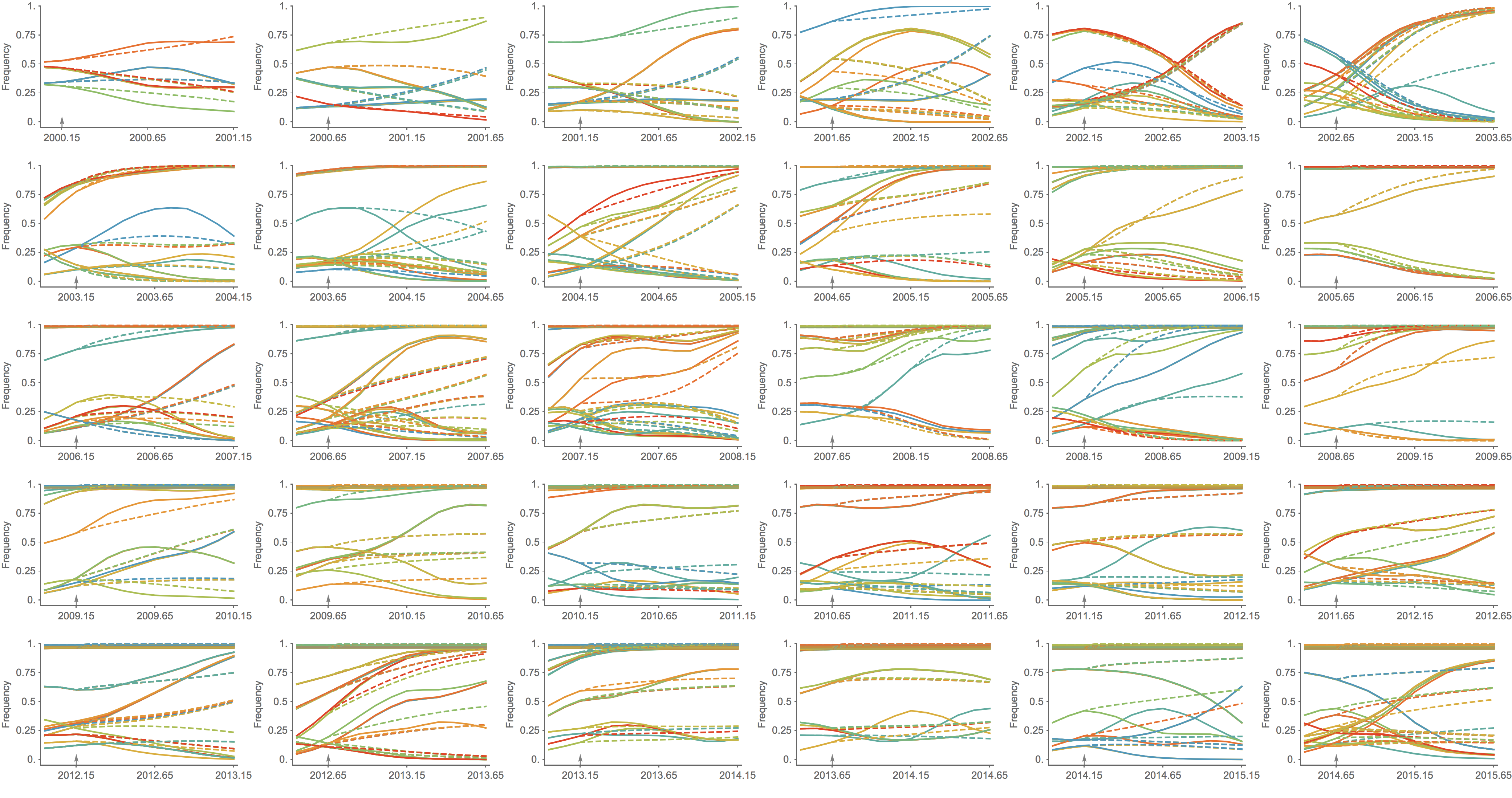
Formalizes intuition about drivers of influenza dynamics
| Model | Ep coefficient | HI coefficient | Freq error | Growth corr |
|---|---|---|---|---|
| Epitope only | 2.36 | -- | 0.10 | 0.57 |
| HI only | -- | 2.05 | 0.08 | 0.63 |
| Epitope + HI | -0.11 | 2.15 | 0.08 | 0.67 |
Further work on predictive modeling
- Integrate data predictors and data sources, e.g. plan to investigate a geographic predictor
- Possible to build predictive models for H1N1 and B and to forecast NA evolution
Evolutionary analyses can inform influenza vaccine strain selection
Analyses must be rapid and widely available
Predictive models can flag clades for experimental follow-up and creation of vaccine candidates
Ebola
Epidemic nearly contained, but resulted in >28,000 confirmed cases and >11,000 deaths
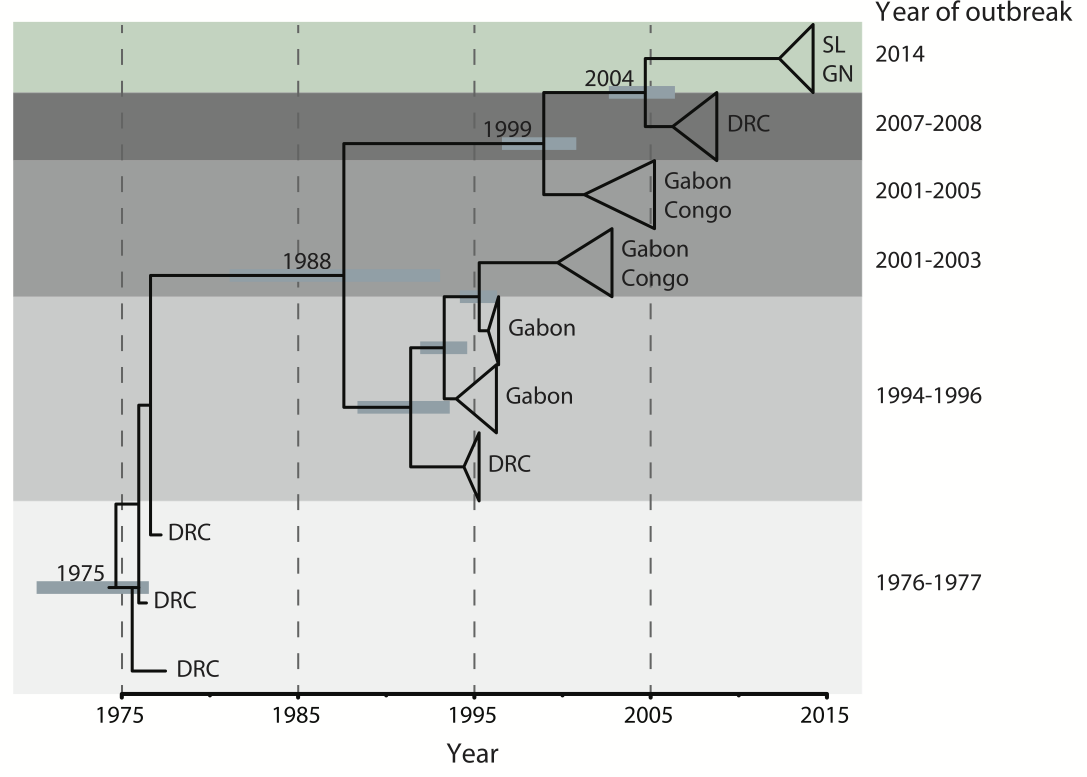
Outbreaks are independent spillovers from the animal reservoir
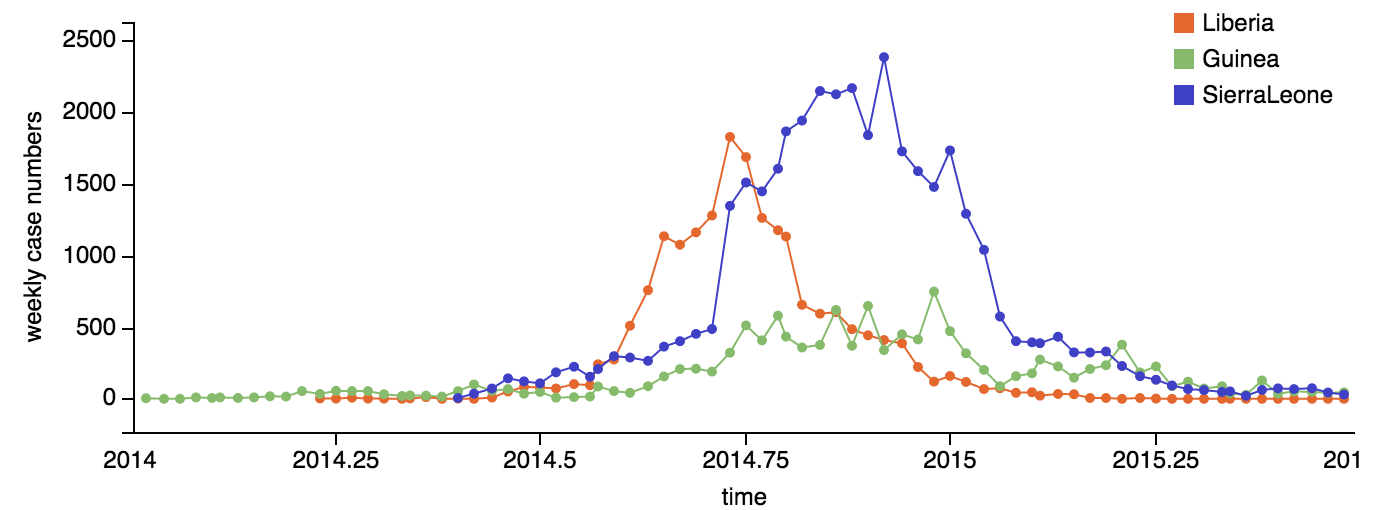
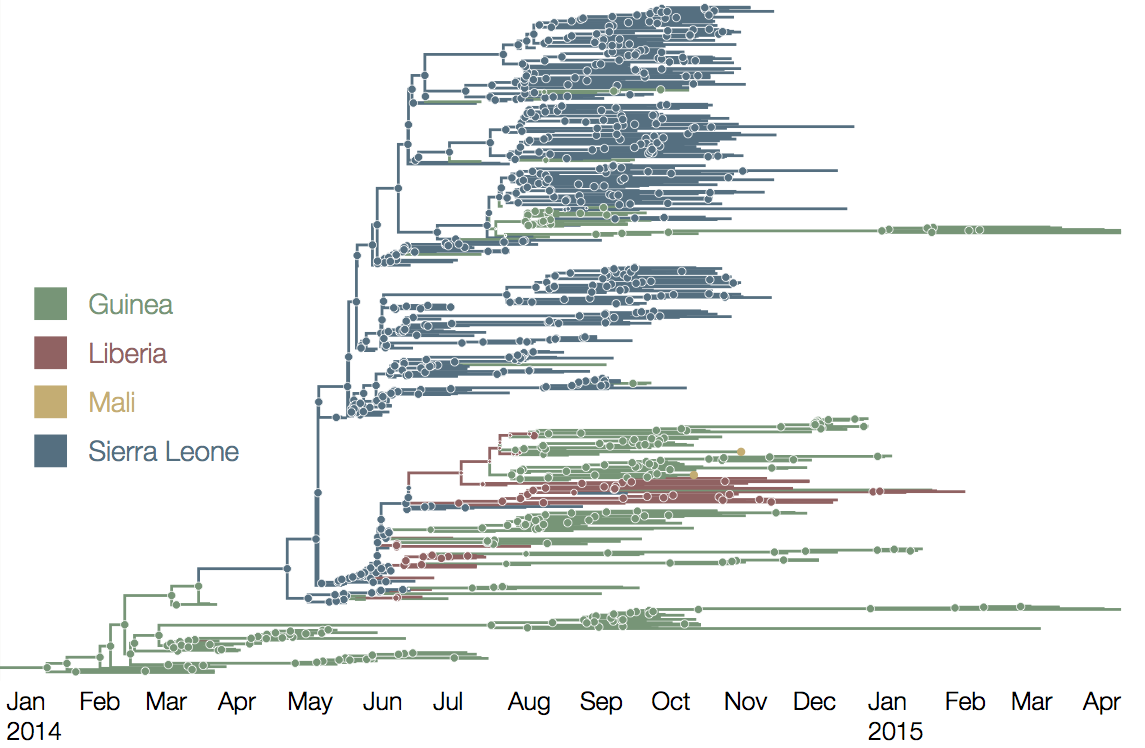
Person-to-person spread in the early West African outbreak
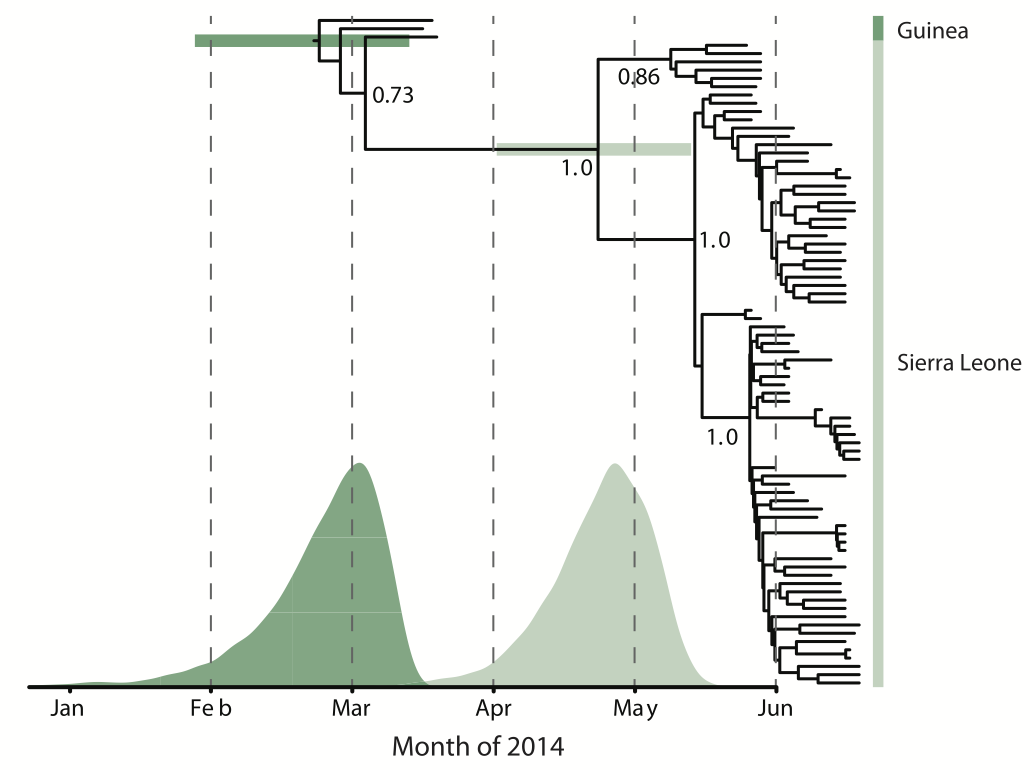
Continued spread through Dec 2014
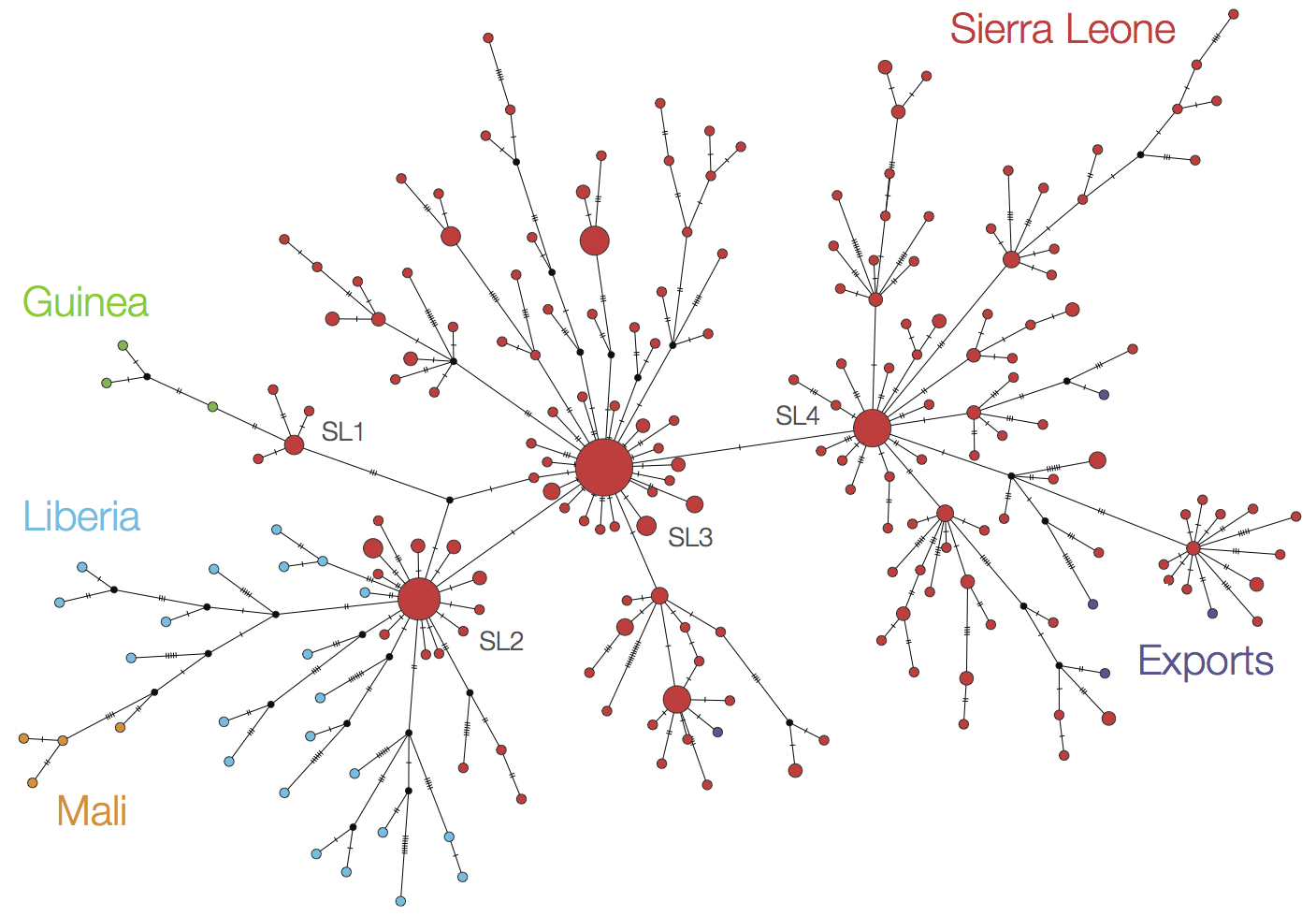
At epidemic height, geographic spread of particular interest
Later on, tracking transmission clusters of primary importance
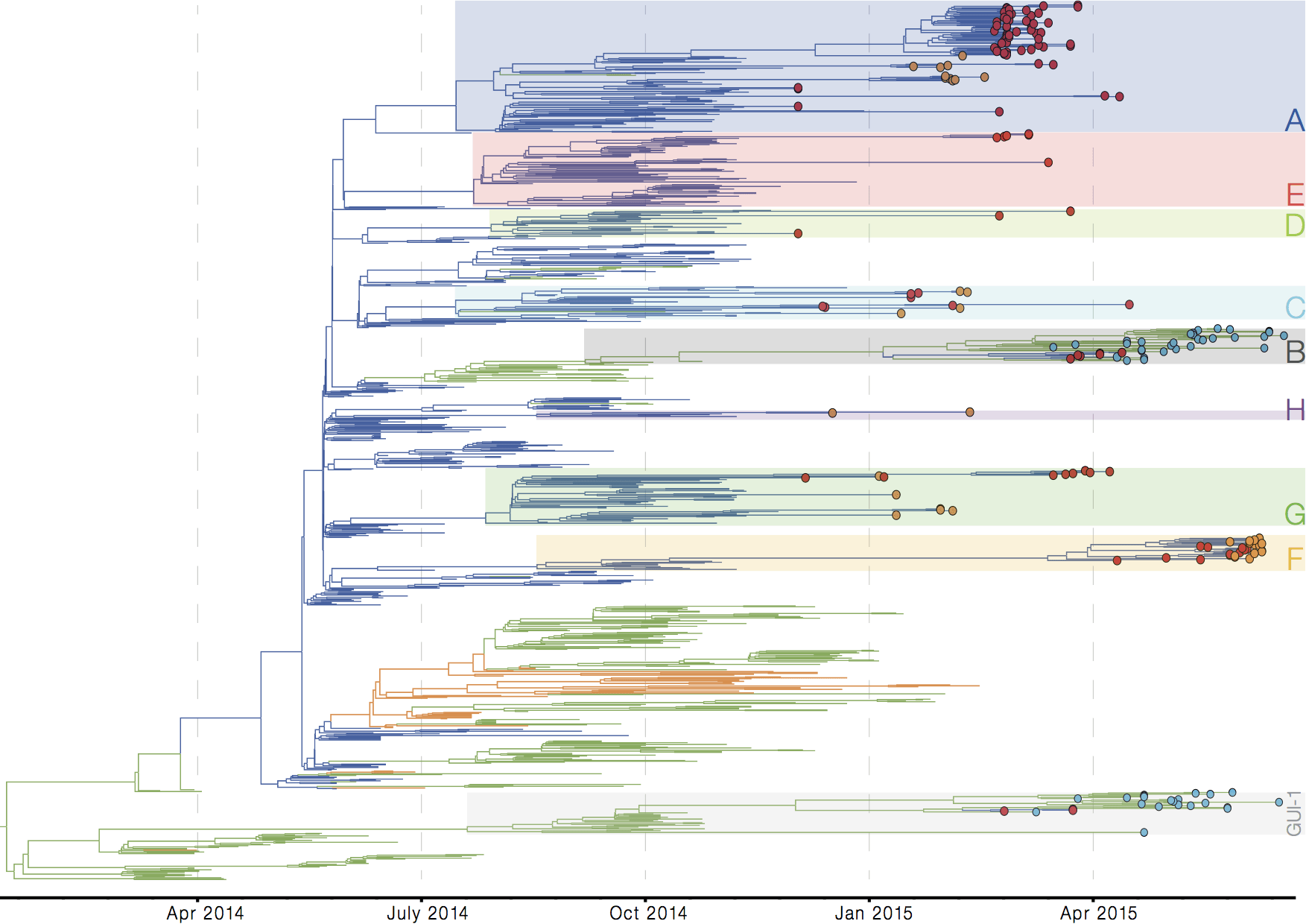
Tracking epidemic spread in real-time:
ebola.nextstrain.org
Middle East respiratory syndrome coronavirus (MERS-CoV)
Cases concentrated in the Arabian Peninsula with occasional exports
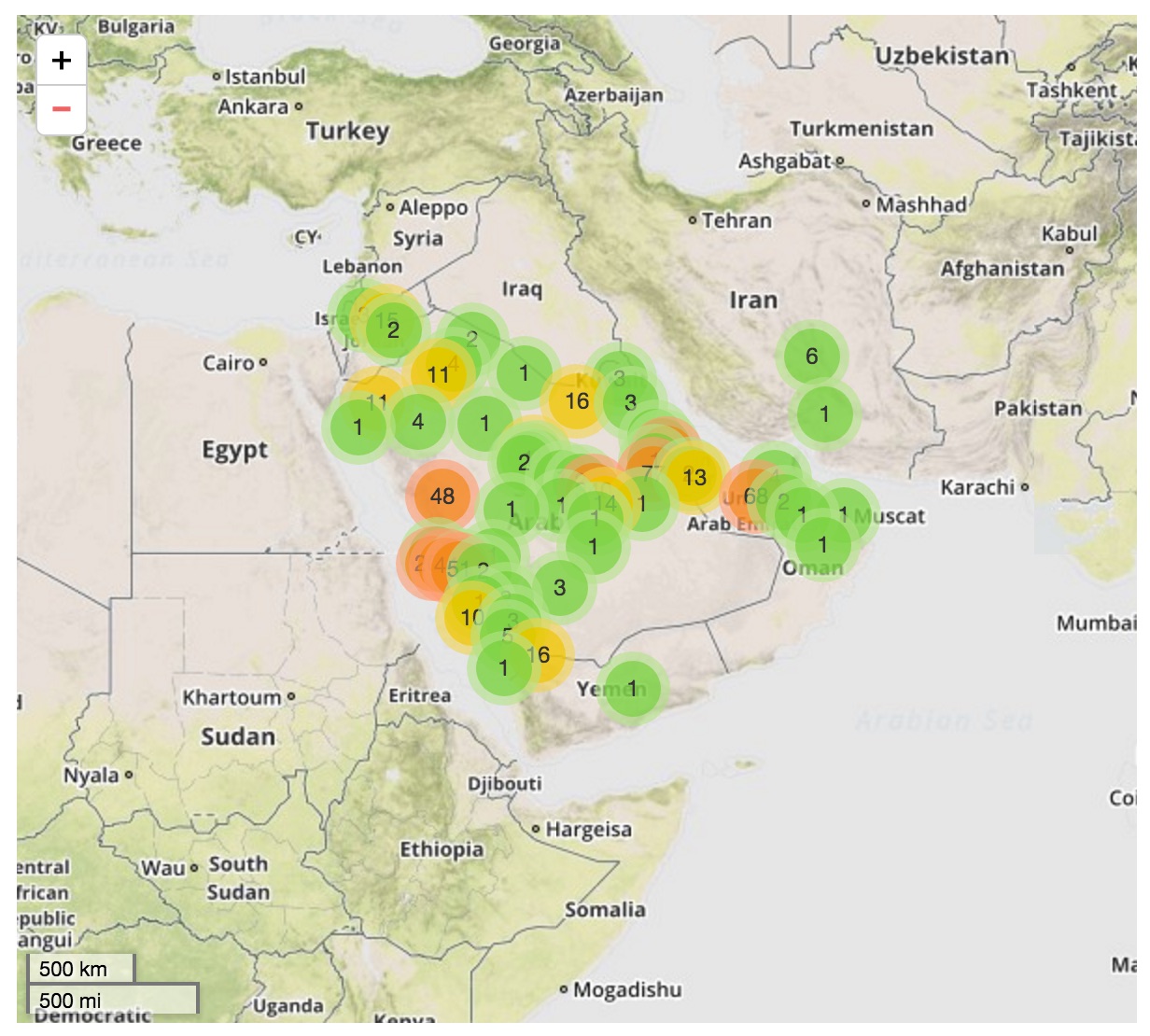
No evidence of epidemic growth, spill-over transmission clusters
Bats ➞ Camels ➞ Humans
Tracking spillover events in real-time:
mers.nextstrain.org
Moving forward, genetically-informed outbreak response requires:
- Rapid sharing of sequence data, genetic context critical
- Technologies to rapidly conduct phylogenetic inference
- Technologies to explore genetic relationships and inform epidemiological investigation
Acknowledgements
Richard Neher (Max Planck Tübingen), Andrew Rambaut (University of Edinburgh), Colin Russell (Cambridge University), Michael Lässig (University of Cologne), Marta Łuksza (Institute for Advanced Study), Gytis Dudas (University of Edinburgh), Pardis Sabeti (Harvard University), Danny Park (Harvard University), Nick Loman (University of Birmingham) Matthew Cotten (Sanger Institute), Paul Kellam (Sanger Institute), WHO Global Influenza Surveillance Network, GISAID



Contact
- Website: bedford.io
- Twitter: @trvrb
- Slides: bedford.io/talks/real-time-tracking-combi/